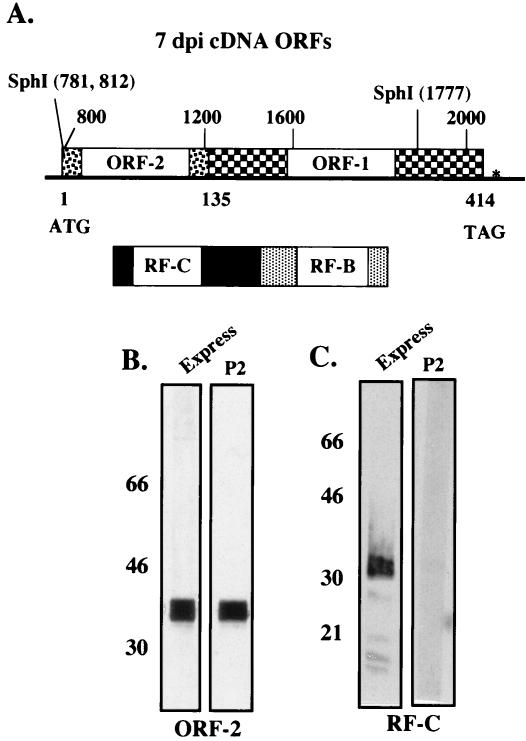
FIG. 2.
Expression of LR ORFs in baculovirus expression vectors. (A) An alternatively spliced LR transcript that was identified in trigeminal ganglia of infected calves at 7 days after infection (dpi) (6) was cloned into pcDNA3.1− (5). This cDNA is spliced at nucleotides 1180 and 1363 such that ORF-2 is fused with ORF-1. The numbers above the ORF-2 schematic are nucleotide numbers of the LR gene, and the numbers below ORF-2 are amino acid numbers. Splicing also generates a fusion protein containing a portion of the RF-C product fused to the RF-B product. The LR cDNA begins at nucleotide 765 and ends at nucleotide 2182. The details of cloning and characterizing the properties of this cDNA were described elsewhere (5). To construct a baculovirus that expressed ORF-2, the SphI fragment containing ORF-2 was cloned into the pGEM-11Zf(−) vector (Promega, Madison, Wis.) into the unique SphI site such that the SphI site at LR nucleotide 810 was adjacent to the unique BamHI site in the vector. This plasmid was digested with HindIII, treated with the Klenow fragment, and then digested with XhoI to release the LR fragment. This LR fragment was cloned into pGEX(GST-5X) (Pharmacia Biotech, Piscataway, N.J.) at unique SmaI and XhoI sites. The resulting fragment was digested to completion with XhoI and then partially digested with BamHI. The resulting BamHI-XhoI fragment was then cloned into pBlueBacHis2 at the unique BamHI-XhoI sites. This method fused ORF-2 in frame with the His fusion sequences and allowed a recombinant baculovirus that expressed the ORF-2 fusion protein to be constructed. (B and C) Recombinant baculovirus strains were constructed and the viruses were grown in SF9 insect cells using procedures described by Invitrogen. Recombinants were characterized by testing for LR protein expression using the Express antibody that recognizes the His tag at the 5′ terminus of LR protein sequences (R910-25; Invitrogen). Large-scale expression was carried out in 2-liter flasks seeded with SF9 cells at a density of 2 × 106/ml in a total volume of 1,000 ml. At 4 days after infection, cells were pelleted by centrifuging for 30 min at 8,000 rpm (Beckman J2-21 centrifuge, JA-10 rotor), suspended in 20 ml of guanidinium lysis buffer (6 M guanidine hydrochloride, 20 mM sodium phosphate, 500 mM NaCl; pH 7.8). Suspended cells were then passed through an 18-gauge needle four times to lyse the cells and shear cellular DNA. (B) His fusion protein purified from SF9 cells infected with the ORF-2 fusion; (C) RF-C fusion protein. The respective protein was loaded at 2 μg/lane, Western blots were performed, and the respective lanes were cut and incubated with the designated antibodies. The P2 antibody was diluted 1:500 for these studies. Western blot analysis was performed with the designated antibodies as previously described (18).