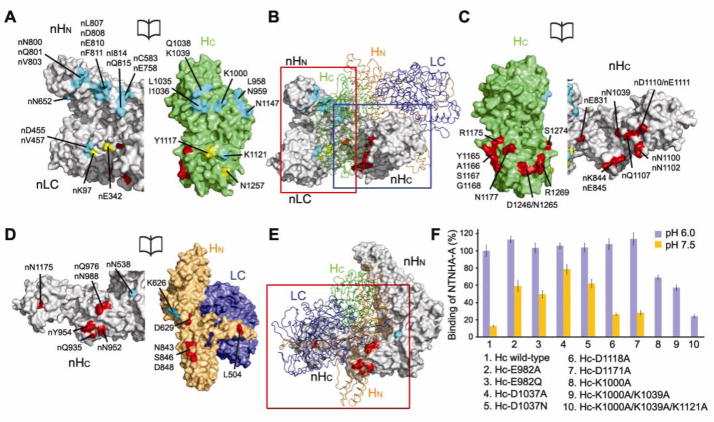
Fig. 3.
The M-PTC is stabilized by extensive intermolecular interactions. (A–C) HC interacts with all three domains of NTNHA-A. The overall structure of the M-PTC is shown in (B) where NTNHA-A is in surface representation (gray) and BoNT/Ai is blue, orange, and green for LC, HN, and HC, respectively. Open-book views of the interfaces highlighted in the red or blue boxes are shown in (A) and (C), respectively. Residues in HC that form hydrogen bonds or salt bridges with nLC, nHN, or nHC are yellow, cyan, and red, respectively. (D) Open-book view of the interface highlighted in the red box of panel E. HN directly contacts nHC (interacting residues shown in red) and nHN (cyan), but not nLC. (F) Pull-down assays between full-length NTNHA-A (prey) and HC variants (baits). Bar graph shows mean ± SD, n=3. Binding of samples 8–10 at pH 7.5 was not detectable.