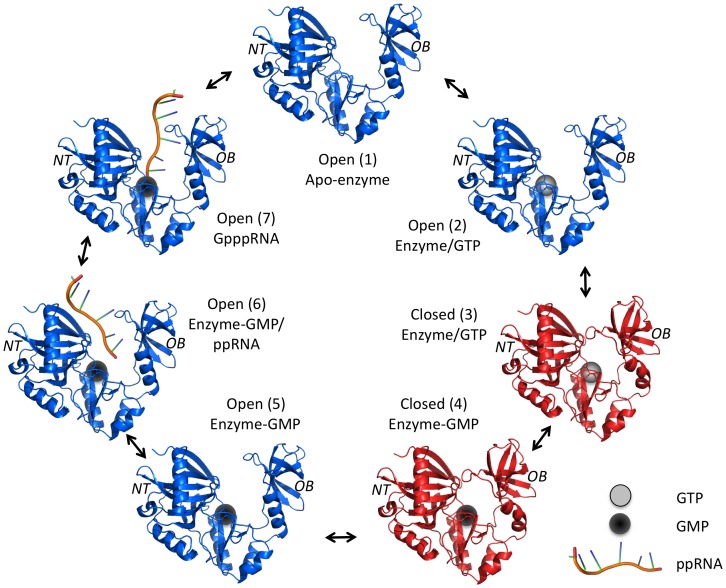
Figure 1. RNA guanylyltransferase mechanistic pathway and associated structures.
The phosphoryltransfer catalysis requires conformational changes between the open (blue) and close (red) form of the RNA guanylyltransferase enzyme. The apo-enzyme (structure 1) first binds GTP (grey sphere, structure 2) which promotes the closure of the OB fold domain toward the NT domain (structure 3). In the catalytically active close conformation, the enzyme hydrolyzes the GTP to form the hallmark enzyme-GMP covalent intermediate complex (black sphere, structure 4). The lost of interactions between the bound guanylate and the OB fold domain, upon GTP hydrolysis, destabilizes the close conformation of the enzyme and leads to its reopening (structure 5) concomitant with the release of the pyrophosphate product. This exposes the RNA-binding site of the enzyme (exact location unknown), thereby allowing 5′-diphosphate RNA binding (structure 6) and subsequent GMP moiety transfer onto the acceptor RNA (structure 7). The capped RNA is then released and the apo-enzyme (structure 1) is regenerated allowing reinitiation of the pathway. (PDB: 1CKN).