Abstract
Jamun or Indian blackberry (Syzygium cumini L.) is a minor and highly perishable fruit enriched with flavonoids, essential oils, anthocyanins phenolic compounds and other antioxidants. The quantitative and qualitative losses in this seasonal fruit are tremendous and can be reduced by appropriate packaging and storage techniques which have not been applied hitherto. This study was undertaken to extend the shelf-life as well as to assess the biochemical, microbiological and physiological changes in jamun fruit under perforated and non-perforated modified atmosphere (MA) conditions. Fruits were stored under differential MA in macro-perforated (1 and 2 perforations, 0.3 mm dia. each) and non-perforated polypropylene (PP) film packages (Thickness: 35 μm, bag area: 0.036 m2) at 5 °C and 75% relative humidity (RH) for 23 days. Sachets containing white silica gel beads were placed inside all the packages to check water accumulation, if any. Different physiological, biochemical and microbiological characteristics which generally affect the post-harvest life of the produce were monitored during the storage period. Results of the study suggested most of the subjectively and objectively determined qualitative parameters to be retained satisfactorily under macro-perforated packaging treatments. Further, the microbiological analysis, surmised that the fruits could be stored for long term using packages with 1 macro-perforation.
Keywords: Antioxidants, Jamun, Modified atmosphere packaging, Headspace, Pigments, Microbial load
Indian blackberry (Syzygium cumini L.), commonly known as jamun is a minor and under-utilised tree-fruit of Indian subcontinent. The plant belongs to Myrtaceae family and grows naturally in clayey loamy soil in tropical as well as in sub-tropical zones of Indo-gangetic plains. These species are reported to be very rich in tannins, flavonoids, essential oils, anthocyanins and other phenolic constituents (Sharma et al. 2003; Reynertson et al. 2008). The purple coloured fruits are available abundantly during summer season for a short span of time, causing a seasonal glut. Jamun contains 19.7% carbohydrates, 0.7% proteins, 0.02% calcium, 0.1% fat, 0.01% phosphorous, 0.4% mineral matter, 0.1% iron and 0.9% fiber (Singh et al. 1967; Benherlal and Arumughan 2007). Besides, they contain anthocyanins which are the most important and widespread groups of colouring matters in plants. These intensely coloured water soluble pigments are responsible for nearly all colours in leaves, petals and fruits of higher plants. Jamun fruits and seeds are sweet, acrid and sour and they are used for treatment and control of diabetes, diarrhoea and ringworm. It is also reported to be a blood pressure regulator (Bhargava et al. 1968). Jamun trees are available around parks, on roadsides, on avenues and are good windbreakers. On account of its unorganised farming, consolidated information regarding its area and production in India is apparently not available. The fruits are highly perishable and are sold by the roadside vendors, under the unhygienic environment, which cuts short its shelf-life drastically.
Modified atmosphere packaging (MAP) along with low temperature storage extends the shelf-life and maintains qualitative parameters of perishable produce by creating of appropriate headspace (O2 and CO2) around a produce packaged in plastic films (Paull 1999). MAP is used with various types of products, where the equilibrated gaseous atmosphere inside a package containing fruits or vegetables depends on the rate of respiration and weight of the produce; permeability characteristics of the packaging material; storage temperature; relative humidity; and free volume (head-space) of the package. The interaction of the packaging material with the packaged produce at the respective storage temperature results in establishment of an equilibrium modified atmosphere (EMA) which is usually low in O2 and high in CO2. These low O2 and high CO2 concentrations which do not cause physiological stress to tissues can reduce respiration and ethylene production, and retard compositional changes associated with colour, firmness, flavor and nutritional quality of various horticultural produce (Kader et al. 1989). Further, the ability of modified atmosphere to reduce the respiratory activity inside the packages not only controls the respiration rate of the fruit, but also decreases the intensity of catabolic activity and degradation processes (Lougheed 1987). Modified atmosphere packaging is commercially used to increase the shelf life of packaged produce by reducing the produce respiration rate, delaying senescence, and inhibiting the growth of many spoilage organisms, ultimately increasing product shelf life (Al-Ati and Hitchkiss 2003). If the storage temperature increases during storage, it enhances the physiological disorders in the stored produce as the rate of biological reactions increases with increase in temperature (Tano et al. 1999).
Studies related to fermentation of jamun fruits to prepare the red wine are available in literature (Chaudhary and Ray 2007). However, for such type of highly perishable and highly respiring produce, micro and macro-perforated films having high gas transmission rates for O2 and CO2 can be used to extend their shelf-life. Further, it is also necessary to evaluate the effect of package headspace on the different biochemical, microbiological and physiological changes in fruit and to access the most suitable headspace for its prospective long term storage under MAP. The specific information related to effect of different in pack gaseous environments under MAP over the qualitative changes in jamun is apparently lacking. During preliminary laboratory investigations, it was observed that jamun has high rates of O2 consumption and CO2 evolution (data not shown). Therefore, this study was undertaken to evaluate the effect of MAP on qualitative and microbiological changes in jamun during its storage for 23 days at 5 °C.
Materials and methods
Fresh jamun were procured from a local wholesaler in Ludhiana. Bright purple coloured firm fruits (22–25 mm long), free from blemishes, apparent disease, insect and mechanical damage; were first hydro-cooled for 60 s using ice-cooled water containing citric acid (0.5%); centrifuged to remove surface water using a basket centrifuge (CIPHET, India) for removal of excess moisture and were then air-cooled for 2 h at 5 °C in a cold room (Sheel Biotech Ltd, India), before the start of the experiment. Air cooling helped in surface drying as well as prepared the fruit for further experimentation. The fruit were packaged in cold rooms itself after they were surmised to be sufficiently acclimatised to the surrounding environment in the cold room.
Packaging and storage
Packages (bag area: 0.036 m2) made from polypropylene (PP) film (Thickness: 35μm, Gas permeability coefficients: 1.54 × 10−6 ml.mm−2 h−1 kPa−1 for O2 and 5.96 × 10−6 ml.mm−2 h−1 kPa−1 for CO2 at 5 °C and 75% RH) were selected for the storage studies. The gas transmission rates of the packaging film for O2 and CO2 at 5 °C, were measured by a gas permeability tester (Labthink, model BTY-B1P, China). The fruits (250 g each) were finally packaged in macro-perforated (1 and 2 perforations, perforation diameter: 0.3 mm each) as well as in non-perforated PP film packages. During preliminary experimentation utilising different desiccants (data not shown), it was observed that packaging of jamun in PP film packages resulted in excessive accumulation of water inside the packages which could be prevented best with the use of white silica gel beads. Therefore, sachets prepared from non-woven cloth and containing 15 g of white silica gel beads were placed inside all the packages. The packaged samples were stored for 23 days at 75% relative humidity (RH) at 5 °C. Jamun kept in the unsealed PP bags were taken as control samples. Weight loss and headspace gaseous concentrations of O2 and CO2 were evaluated daily. Three packages from each type of packaging treatments were analyzed on 3rd, 15th, and 23rd day of storage for pigments, ascorbic acid, anthocyanin, phenolic content, flavonoids and antioxidant capacity. Total count was recorded with standard plate count technique.
Weight loss and headspace gas analysis
During the entire storage period, the weight loss was determined by weighing the individual package on the day of observation using a laboratory level weighing scale having 0.01 g accuracy (Scaltec Instruments, Germany). Package headspace was monitored by means of a portable Gas Analyzer (Model 902 D Dualtrak, Quantek, USA). The instrument evaluated the headspace by means of an electrochemical and an infrared sensor (sensitivity: 0.1% O2; 0.1% CO2, accuracy: 0.1% O2; 0.2% CO2) for O2 and CO2 concentrations, respectively. The instrument was calibrated with the standard O2 and CO2 gases; before the actual observation. A sampling probe containing a particulate filter and a removable needle having dual side-port holes were used to draw the sample from the package headspace with the help of an electronically controlled miniature pump. The drawn samples were fed simultaneously to the O2 and CO2 sensors and concentrations of O2 and CO2 were directly read on the digital display panel of the instrument.
Total chlorophyll
Total chlorophyll was determined by homogenizing 1 g of fruit with 10 ml of acetone and n-hexane (4:6); using a tissue homogenizer (Labco, New Delhi, India) over ice to prevent any biochemical changes during extraction. The extraction was carried out till the whole tissue was extracted leaving white precipitate behind .The homogenized solution was allowed to stand for 1 min in an amber colored glass tube placed inside the flaked ice and was protected from direct exposure to the light by covering it from outside by aluminium foil. One ml of the supernatant was taken and was diluted with 9 ml of the extract solution. The resulting solution was analysed spectrophotometrically with the help of a UV-Vis spectrophotometer (Spectroscan 80DV, Biotech Engineering Management Company Limited, UK). Optical density of the solution was measured at different wavelengths namely 663, 645, 505, 453 nm using acetone and n-hexane (4:6) as blank. Total chlorophyll (mg/100 g fresh weight of fruit) was quantified using relationships as described by Nagata and Yamashita (1992).
Total Anthocyanin content
The total anthocyanin content was determined as per the method of Swain and Hillis (1959). 1 g of fruit was extracted with 10 ml of methanol. The extract was centrifuged at 3,000 g in a cold centrifuge (MP 400-R, Eltek Limited, India) at 3 °C for 15 min. 1 ml of supernatant was mixed with 3 ml of HCl in aqueous methanol (0.5N HCl in 80–85% methanol) and 1 ml of anthocyanin reagent [1 ml of 30% H2O2 in 9 ml of methanolic HCl (5:1, 3N)]. After 15 min of incubation in the dark, optical density of the reaction mixture was measured against blank at 525 nm with the help of UV-Vis spectrophotometer (Spectroscan 80DV, Biotech Engineering Management Company Limited, UK). Quantification of anthocyanins was carried out as per the standard curve prepared from cyanin hydrochloride.
Ascorbic acid
Ascorbic acid was determined quantitatively as per the modified 2,6-dichlorophenolindophenol (DIP) method (Klein and Perry 1982). Standard curve for the ascorbic acid was prepared within the linear range of 0–500 mg ascorbic acid/ml. For extraction, 1 g of fruit pulp from different packaging treatments was homogenized using a tissue homogenizer (Labco, New Delhi, India) for 1 min with 10 ml of 1% metaphosphoric acid (v/v) over ice. The extract was centrifuged at 3,000 g in a cold centrifuge (MP 400-R, Eltek Limited, India) at 3 °C for 15 min. 1 ml of the supernatant was mixed with 9 ml of 0.05 mM of DIP using a vortex shaker (Labco, New Delhi, India) for 15 s and absorbance of solution was measured against the blank at 515 nm with the help of a UV-V spectrophotometer (Spectroscan 80DV, Biotech Engineering Management Company Limited, UK).
Total phenolic content
Total phenols were determined as per McDonald et al. (2001), using Folin Ciocalteu reagent. 1 g of jamun was extracted with 10 ml of methanol: water (50:50, v/v). 0.5 ml of the diluted (1:10) extract or the standard phenolic compound (Gallic acid) was mixed with 5 ml of Folin Ciocalteu reagent (1:10 diluted with distilled water) and 4 ml of aqueous Na2CO3 (1M). The mixture was allowed to stand for 15 min and optical density of the mixture was determined against the blank at 765 nm with the help of UV-Vis spectrophotometer (Spectroscan 80DV, Biotech Engineering Management Company Limited, UK).The standard curve was prepared using 0,50,100, 150, 200, 250 μg solutions of gallic acid per ml of methanol: water (50:50, v/v). Total phenol values were expressed in terms of the standard reference compound as gallic acid equivalent (g/100 g fresh weight of fruit).
Total flavonoids determination
Total flavonoid content was determined Chang et al. (2002). 1 g of fruit was extracted with 10 ml of pure methanol. 0.5 ml of extract solution was mixed with taken 1.5 ml of methanol, 0.1 ml of 10% aluminium chloride, 0.1 ml of 1 M potassium acetate and 2.8 ml of distilled water. The solution was then incubated for 30 min. The optical density of the reaction mixture was measured against the blank at 415 nm with the help of UV-Vis spectrophotometer (Spectroscan 80DV, Biotech Engineering Management Company Limited, UK). The standard calibration curve was prepared by using quercetin solutions at concentrations 12.5 to 100 μg/ml in methanol.
Antioxidant capacity
Antioxidant capacity was determined through assessment of free radical-scavenging effect on 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical (HiMedia Laboratories, India) as per the method of De Ancos et al. (2000). 1 g of fruit was extracted with 5 ml of methanol. The extract solution was centrifuged at 6,000 g for 15 min at 4 °C, using a cold centrifuge (MP 400-R, Eltek Limited, India). Aliquots of 0.01 ml of supernatant so obtained were mixed with 3.9 ml of methanolic DPPH (0.025 g/l) and 0.090 ml of distilled water. The resulting mixture was shaken on a vortex shaker (Labco, New Delhi, India) and was then incubated in dark for 30 min. Absorbance of the mixture was measured against the blank at 515 nm using UV-Vis spectrophotometer (Spectroscan 80DV, Biotech Engineering Management Company Limited, UK). The results were obtained as the percentage decrease with respect to the absorbance of a reference DPPH solution.
Assessment of microbial load
Fruits obtained from the market as well as the ones stored under MAP were evaluated for microbial contamination as per the standard plate count method. 25 g of jamun was first homogenized with 225 ml of peptone water (0.1M, pH 7.0) using a sterilized kitchen blender (Singer India Ltd., India) and was then further diluted to get serial dilutions. Each set of serially diluted sample was spread on to respective agar plates. Mesophilic count was determined using plate count agar (HiMedia Laboratories, India) by spread plate technique followed by incubation at 35 °C for 72 h. Yeast and mould count were determined with Rose Bengal medium followed by incubation at 28 °C for 3–5 days.
Sensory and visual appearance assessment
The sensory and visual assessment for fruits was carried out by a five member trained panel by suitably modifying the procedure of Carvalho and Clemente (2004). Aroma was evaluated as per the scale: 5 = no off-odour, 4 = very light off-odour, 3 = light off-odour, 2 = medium off-odour and 1 = strong off-odour. Texture evaluation was carried out using: 5 = very firm, 4 = firm, 3 = slightly wilted, but acceptable, 2 = wilted, 1 = very much wilted. Visual appearance was scored as per Deza et al. (2003) using: 5 = very good, 4 = good, 3 = normal, 2 = limited quality and 1 = not acceptable. Disease severity was assessed as per the method of Burton et al. (1987), as utilised with appropriate modifications for this work: 5 = severely diseased, 4 = diseased, 3 = moderately diseased, 2 = slightly diseased and 1 = no disease. Another nine point scale; 0 = fruits completely wet and water accumulation; 1 = fruits and film moderately wet; 3 = fruits moderately wet; 5 = fruits and film slightly wet; 7 = fruits slightly wet; 9 = no water accumulation was used to represent the water vapour accumulation inside the film packages (Rai et al. 2009). During scoring, the intermediate scores were also given to the samples depending upon the perceived condition of the samples.
Statistical analysis
The statistical analysis of data obtained was carried out to establish the difference among the samples. All the experiments were performed in triplicate. One-way analysis of variance (ANOVA) and multiple comparisons (Fisher’s least significant-difference test) were used to evaluate the significant difference of the data at p < 0.05 using a statistical package (Statgraphics Plus, Statpoint Inc., USA) and the means were plotted to draw out inferences.
Results and discussion
Weight loss and headspace atmospheric composition
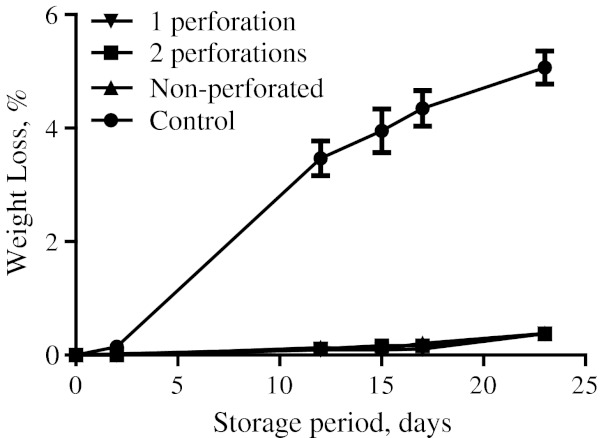
Among all the packaging treatments, no significant weight loss was observed (Fig. 1) under MAP treatments (both for macro-perforated as well as non-perforated ones), due to the effect of altered headspace prevailing in them. Further, the apparent increase in in-pack relative humidity also prevented the weight loss in conformation with the earlier reported results of Tomkins and Cumming (1988). However, the control samples, being under normal atmosphere lost 4.80% of their initial weight towards the 23 days.
Fig. 1.
Weight loss in jamun fruits under modified atmosphere packaging for 23 days of storage at 5 °C under different packaging treatments (1 perforation, 2 perforations, non-perforated, control) (n = 3)
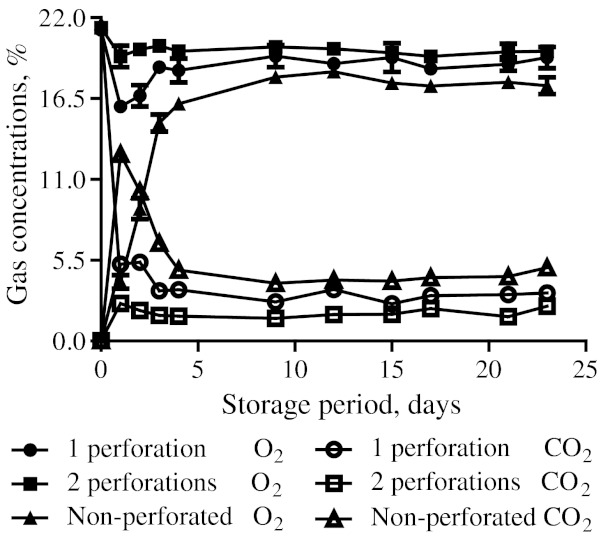
Package headspace compositions of O2 and CO2 for different packaging treatments are plotted in Fig. 2. Under all the packaging treatments, initially a rapid decrease in O2 and a corresponding increase in CO2 concentrations respectively were observed on 1st day which could be attributed to the initial adjustment and high respiratory behaviour of fruits in the transient state of stabilisation and equilibration. Subsequently, with the progress of storage under low temperature, the respiration of fruits decreased suddenly which led to in-pack accumulation of O2 under all the treatments. The O2 concentration inside the macro-perforated film packages containing 1 and 2 perforations arrived at 18.3 and 19.7%, respectively on the 4th day of storage itself and did not change much thereafter till 23rd day of storage, which indicated that the gaseous equilibrium was achieved on 4th day itself. Whereas, for non-perforated packages, the O2 levels stabilized around 16.5%. Also, CO2 concentrations inside the macro-perforated packages were observed to be 3.5% and 1.7% for packages having 1 and 2 perforations, respectively. The non-perforated packages had CO2 concentration to the tune of 4.8% on the 4th day of storage and it remained almost at the similar level till the end of storage period.
Fig. 2.
Changes in concentrations of O2 and CO2 inside the polypropylene (PP) film bags containing jamun fruits under modified atmosphere packaging at 5 °C and 75% RH (n = 3)
Biochemical constituents
Quality deterioration (colour, flavour and texture) is attributed to the effect of endogenous enzyme, enhanced respiration (Gil et al. 1998; Kim et al. 1993), physical abuse and environmental factors (Chau and Talasila 1994). Table 1 depicts the changes in the different biochemical constituents, as observed after 23 days of storage under different packaging treatments. Practically, the matured fruits are considered to be devoid of green coloured chloroplasts as basically they are purple in colour. However, this study indicated the initial presence of chlorophyll in jamun fruit, which was later observed to decrease under all the packaging treatments; indicating the continuous depletion of the green-colour during the storage. The total chlorophyll initial content in jamun was observed to be 13.88 mg/100 g of fruit. At the end of the storage period, the total chlorophyll content arrived at 2.24, 3.62 and 6.06 mg/100 g of fruit in 1, non-perforated and control samples, respectively. However, only a slight decrease in the chlorophyll content was observed under 2 perforation samples, largely due to the prevailing headspace atmosphere which might have helped to retain the chlorophyll, in these packages.
Table 1.
Changes in the biochemical constituents of jamun fruit after storage for 23 days under modified atmosphere packaging at 5 °C and 75% RH
| Chlorophyll mg/100 g fw | Ascorbic acid mg/100 g fw | Phenol mg/100 g fw | Flavonoid mg/100 g fw | DPPH inhibition % | Anthocyanin mg/100 g fw | |
|---|---|---|---|---|---|---|
| At harvest | 13.88 ± 1.05a | 55.48 ± 4.32a | 1175.17 ± 87.45a | 115.11 ± 13.32a | 61.56 ± 8.96a | 7.25 ± 0.96a |
| 1 Perforation | 2.24 ± 0.23b | 44.51 ± 4.02b | 1495.07 ± 77.34b | 159.88 ± 13.87b | 89.27 ± 10.13b | 4.64 ± 0.43b |
| 2 Perforation | 12.18 ± 1.34a | 36.70 ± 2.23c | 2097.82 ± 112.32c | 141.05 ± 15.22c | 92.10 ± 12.65c | 0.64 ± 0.17c |
| Non-Perforated | 3.62 ± 0.45b,c | 21.06 ± 1.98d | 1565.78 ± 110.32b,d | 143.98 ± 17.77d | 56.68 ± 7.49d | 0.27 ± 0.09c,d |
| Control | 6.06 ± 0.65d | ND | 1478.23 ± 98.54b,e | 92.52 ± 11.32e | 96.55 ± 8.43e | 1.26 ± 0.29c,e |
Means±SD within the same column with different superscripts are significantly different at p < 0.05; n = 3
Anthocyanins has many positive health benefits which includes reduction of risk of coronary heart disease, improved visual acuity, antioxidant activities and anticancer activities (Timberlake and Henry 1988; Tamura and Yamagami 1994). Further, its accumulation in plant tissues is influenced by many environmental factors (Grisebach 1982). In the present study, the total initial anthocyanin content of jamun fruits at the beginning of storage was observed to be 7,250 μg/100 g of fruit (Table 1) which decreased sharply under all the packaging treatments with the progress of storage. This is in agreement with earlier reported observations on fruits and vegetables (Gil et al. 1996). Among all the packaging treatments, the decrease was statistically significant at 95% confidence level with p-value < 0.05 which indicated a major change in anthocyanin content during storage. For the perforated packages (1 and 2 macro-perforations), the anthocyanin content could be retained up to 64% on 23rd day of storage in packages having 1 macro-perforation. On the other hand, non-perforated and control samples could retain only 4 and 17% of anthocyanin at the end of storage, which was indicative of the appropriateness of macro-perforated MAP for this fruit under low-temperature.
Tissue damage and bruising leads to a decrease in the ascorbic acid content on account of increased water loss and exposure of ascorbic acid to oxidation (Klein 1987; Loewus and Loewus 1987). During this study, although the ascorbic acid content decreased under all the packaging treatments (Table 1), it was largely undetectable in control samples after 23rd days because of tissue rupture. However, the ascorbic acid could be retained in macro-perforated packages and its retention was found to be significantly higher (80%) at 95% confidence level with p-value < 0.05 for fruits packaged in packages having 1 macro-perforation which indicated that the in-pack gaseous atmosphere was just suitable for maintenance of the ascorbic acid content of fruits. On the other hand, the non-perforated samples could retain only 37% of their initial concentration towards the end of the storage period.
Induction of phenolic compounds is a physiological response to infections or injuries (Amanatidous et al. 2000). Wounding stimulates the adjacent cells to produce more phenolics in an attempt to initate the repair process. The increased production of phenols has an antimicrobial effect which helps in reduction of microbial load (Dixon and Paiva 1995). One of the detrimental changes induced by wounding (cutting, cracking and breaking) is the induction of phenylpropanoid metabolism which results in the accumulation of phenolic compounds (Amiri and Bompeix 2004). The total initial phenolic content of jamun fruits was 1,175 mg/100 g fresh weight of fruit. MAP increased the phenolic content of fruits during storage, both for perforated and non-perforated treatments which may be due to an increase in the activity of the phenylpropanoid pathway under stressful conditions and as evident from synthesis and accumulation of phenolics compounds (Kang and Saltveit 2002). For the perforated samples, the increase was observed to be more for 2 macro-perforation samples which could be attributed to the higher microbial invasion as well as the lower levels of headspace CO2 prevailing in these packages which is also reported to have an inhibitory effect on micro-organisms (Luck and Jager 1997). On the other hand, increase in the phenolic content of non-perforated and control samples could also be solely attributed to the wound response mechanism as the damage to the stored fruits was visibly apparent on 23rd day of storage.
Flavonoids are known to reduce risks of cancer, heart disease and other age related degenerative diseases as well as for providing free radical scavenging activity to the body fluids such as blood. Recent studies have shown that coronary heart disease in elderly males was inversely correlated with their intake of flavonoids (Rice-Evants et al. 1996). Initially, the total flavonoid content of fruits was observed to be 115 mg/100 g fresh weight of jamun fruit. After 23rd day of storage under MAP in non-perforated and perforated samples, the flavonoid content was found to be increasing significantly at 95% confidence level with p-value < 0.05 for fruits under MAP. The increase was seen to be more prominent in 1 perforation treatment. The wound induced ethylene concentrations are also reported (Del Caro et al. 2004) to increase the flavonoid content which could have been the case in packaged fruits only, in this study too. On the other hand, unsealed control samples lost a significant amount of their initial flavonoid content and at the end of 23rd day; they could retain only 80% of their initial concentration as the generated ethylene could easily escape to surrounding environment, leading to dilution in its concentration.
The antioxidant capacity of fruits and vegetables largely depends upon a plethora of individual or due to combined effect of individual antioxidants. Additionally, it has been determined that the antioxidant effect of plant products is mainly due to radical scavenging activity of phenolic compounds such as flavonoids, polyphenols, tannins, and phenolic terpenes (Matsukawa et al. 1997; Rahman and Moon 2007). The antioxidant activity of phenolic compounds is mainly due to their redox properties, which play an important role in adsorbing and neutralising free radicals, quenching singlet and triplet oxygen, or decomposing peroxides (Hasan et al. 2008). In this study, the free radical scavenging activity of jamun fruits vis-a-vis DPPH radical was evaluated on the days of observation (Table 1). It was observed that the scavenging activity increased at the end of storage period in perforated and control samples, which could largely be attributed to the observed increase in antioxidant content generated through wound-response mechanism (Lana and Tijskens 2006). On the other hand, the radical scavenging activity decreased slightly for non-perforated samples due to the highest level of headspace CO2 observed in these packages, which might have prevented the action of antioxidants.
Changes in aroma, texture, visual colour, disease severity and water accumulation
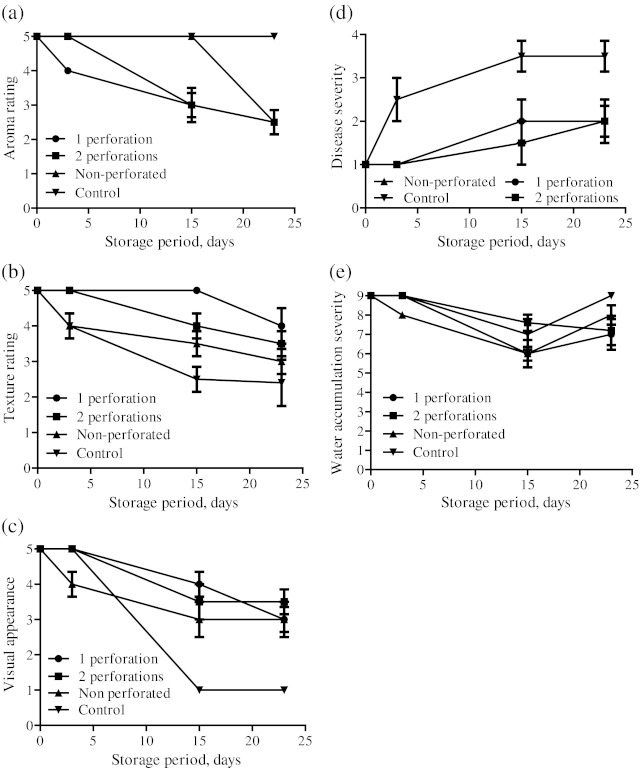
The aroma, texture, visual colour, disease severity and water accumulation ratings after 23 days of storage for different packaging treatments are presented in Fig. 3(a-e). Basically, aroma is a function of package headspace as different gaseous conditions have different effects on biochemical constituents and thus headspace aroma. The headspace aroma was not observed to be statistically significant among all the perforated packaging treatments (Fig. 3a). The 1 and 2 holes treatments had the aroma rating as “light off odour” which led to the conclusion that the equilibrated in-pack headspace gaseous concentration did not allow much synthesis and accumulation of off odour. The rating for non-perforated treatment arrived at “medium off odour” mainly due to deterioration of some fruits and thus synthesis of off odours under non-perforated conditions. For unsealed samples, as observed by various other researchers (Toivonen 1997; Rai et al. 2009), no off odours could accumulated inside the packages as they could escape easily to the storage environment.
Fig. 3.
The variation in sensory and visual ratings for aroma(a); texture (b); visual colour (c); disease severity (d); and water accumulation (e) inside the packages containing jamun for 23 days of storage at 5 °C under different packaging treatments(1 perforation, 2 perforation, non-perforated, control) (n = 3)
A significant difference in the texture was observed among the packaged and control samples (Fig. 3b). 1 perforation sample was observed to be firm after 23 days of storage, whereas slight softness took place under 2 perforations. While the non-perforated samples had an acceptable texture on 23rd day, the control samples became wilted and shrivelled and were totally unacceptable on 23rd day. Visually, the control samples (Fig. 3c) lost their bright purple colour at a faster rate and at the end of 15th day; the skin surface became very patchy and abnormal. While for perforated and non-perforated packaging treatments, the visual colour ratings at the end of storage were although observed to be very slightly different from each other, but on the whole they were acceptable.
A difference in the disease severity ratings was observed among the packaged and control samples (Fig. 3d). The control sample fruits had slight to moderate disease on the apical portion which later on became severe but remained confined to same area during progression of storage period. Whereas, the disease symptoms within the acceptable range appeared for all the packaging treatments only around 15th day and subsequently they were not significantly different on 23rd day for perforated and non-perforated treatments.
As the product respires, accumulation of water inside the film packages takes place which either goes out of the package or condenses, depending upon the water vapour transmission rate of the packaging film used. Our preliminary investigations showed that storage of jamun fruits under MAP leads to substantive condensation and thus accumulation of moisture inside the packages. In this study, water accumulation among the packaging treatments and control samples remained within acceptable limits largely due to its absorption by silica gel pouches. An interesting phenomenon was observed on 15th day, as a drop in water accumulation rating was observed under all treatments which could be due to the saturation of the silica gel pouches (Fig. 3e). However, the rating was observed to be better on 23rd day as later on, the accumulated water diffused out of packages slowly.
Total microbial load
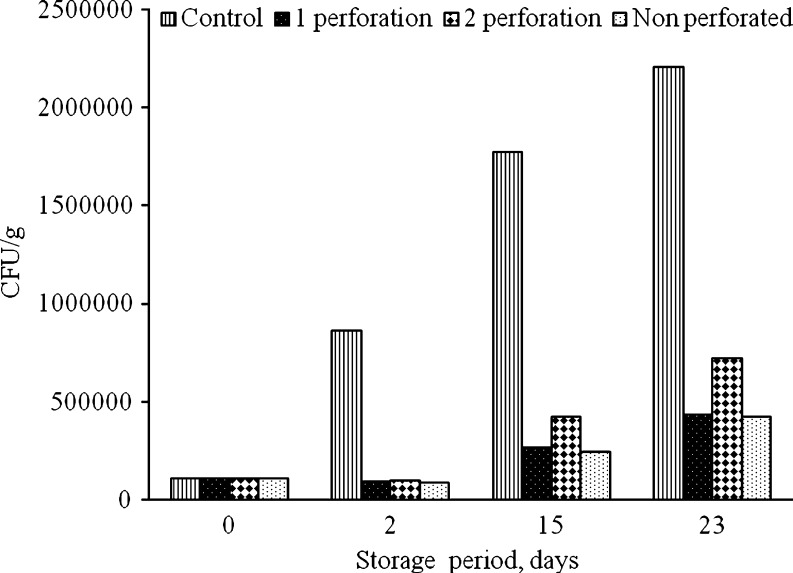
The microbial load on fresh and stored jamun was evaluated during storage under modified atmosphere packaging. Microbiological analyses were carried out on fresh as well as citric acid (0.5%) treated fruits. Initially, the total microbial count was observed to be 18 × 104, which consisted of 51.7% bacteria, 46.4% yeast and 1.7% mold. However, treatment with citric acid resulted in 45% reduction in microbial count. The inhibitory effect of citric acid was observed to be more prominent on yeast and mold probably due to pH lowering effect of citric acid treatment. In this study, the microbial population was found to be lower in packaged samples as compared to non packaged control samples. On 3rd day of storage, both perforated (1 and 2 perforations) and non perforated samples showed a decline in microbial count as compared to control samples (unsealed packages).The microbial count in control sample was found to be 86 × 104, which was higher than the initial microbial count as well as that observed under all packaging treatments (Fig. 4). Also, till 3rd day, the microbial load of packaged samples was found to be lower than the total microbial count recorded for fresh samples. Further, while the fruits packaged in macro-perforated packages (1 and 2 perforation(s)) showed similar trends on 15th day, the non-perforated film packages had the minimum microbial load, followed by 1 and 2 perforations in the increasing order, respectively. Quantitatively, the increase for non-perforated, 1 and 2 perforations was observed to be 7, 6.5 and 4 folds lower than the control samples, respectively. No molds were detected till 15th day of storage both in control and packaged samples. Although, bacteriostatic effect of CO2 is not fully understood, exclusion of O2 by CO2 is reported to create an acidification effect leading to a decrease in microbial count. On 23rd day, control sample showed highest microbial load which indicated that fruits packed in PP film (with and without perforation) harboured less microbes as compared to control largely due to accumulation of CO2 in these packages. While, in control samples, the fruits were exposed to the normal atmosphere for a longer period of time which led to survival and growth of micro-organisms (Daniels et al. 1985; Babic et al. 1996).
Fig. 4.
Total microbial count (cfu/g) of packaged and unpackaged samples during storage of jamun fruits for 23 days of at 5 °C (n = 3)
Conclusion
Modified atmosphere had a beneficial effect on the shelf-life, biochemical and microbiological content of otherwise highly perishable jamun fruit stored for 23 days at 5 °C. Among the macro-perforated treatments (1 and 2 macro-perforations), selection of particular headspace on the basis of biochemical constituents was difficult as they behaved differently under slightly different package headspace generated in macro-perforated packages used in this study. While the ascorbic acid content, flavonoids and anthocyanin content were retained better under 1 perforation treatment, the chlorophyll and phenolic content were either maintained or had a nominal increase in 2 perforations treatment. On the other hand, the free radical scavenging activity, which is basically a combined effect of a plethora of antioxidants, was not significantly different for macro-perforated treatments. It is for this very reason (action of antioxidants) that the fruits under non-perforated and control treatments had their radical scavenging activity intact even when most of the other physico-chemical constituents such as ascorbic acid and anthocyanin content showed a downward trend. Subjective analysis of the samples under different treatments showed light off odour, acceptable visual color, firm texture, disease and water accumulation ratings within the limits of acceptability for macro-perforated pouches. Further, the microbiological analysis throughout the course of experiment were not significantly different for non-perforated and packages with one perforation till the end of storage. Hence packaging of jamun fruits in film with one macro-perforation (0.3 mm diameter) was best suited for its long term storage at 5 °C.
References
- Al-Ati T, Hitchkiss JH. The role of packaging film permselectivity in modified atmosphere packaging. J Agric Food Chem. 2003;51(14):4133–4138. doi: 10.1021/jf034191b. [DOI] [PubMed] [Google Scholar]
- Amanatidous A, Slump RA, Gorris LGM, Smid EJ. High oxygen and high carbon dioxide modified atmospheres for shelf-life extension of minimally processed carrots. J Food Sci. 2000;65:61–66. doi: 10.1111/j.1365-2621.2000.tb15956.x. [DOI] [Google Scholar]
- Amiri A, Bompeix G. Micro-wound detection on apple and pear fruit surfaces using sulphur dioxide. Postharvest Biol Technol. 2004;36:51–59. doi: 10.1016/j.postharvbio.2004.10.010. [DOI] [Google Scholar]
- Babic I, Roy S, Watada E, Wergin WP. Changes in microbial populations on fresh cut spinach. Int J Food Microbiol. 1996;31:107–119. doi: 10.1016/0168-1605(96)00969-5. [DOI] [PubMed] [Google Scholar]
- Benherlal PS, Arumughan C. Chemical composition and in vitro antioxidant studies on Syzygium cumini fruit. J Sci Food Agric. 2007;87:2560–2569. doi: 10.1002/jsfa.2957. [DOI] [PubMed] [Google Scholar]
- Bhargava UC, Westfall AB, Siehr DJ. Prelimnary pharmacology of ellagic acid from juglens nigrra (black walnut) J Pharmaceutical Sci. 1968;57:1728–1732. doi: 10.1002/jps.2600571021. [DOI] [PubMed] [Google Scholar]
- Burton KS, Frost CE, Nichols R. A combination of plastic permeable film system for controlling post harvest mushroom quality. Biotechnol Lett. 1987;9:529–534. doi: 10.1007/BF01026655. [DOI] [Google Scholar]
- Carvalho PT, Clemente E. The influence of broccoli (Brassica oleracea var. italica) fill weight on postharvest quality. Ciência e. Tecnologia. Alimentos. Campinas. 2004;24:646–651. [Google Scholar]
- Chang CC, Yang MH, Wen HM, Chern JC. Estimation of total flavonoid content in propolis by two complementary colorimetric methods. J Food Drug Analysis. 2002;10:178–182. [Google Scholar]
- Chau K, Talasila P. Design of modified atmosphere packages for fresh fruits and vegetables. In: Singh R, Oliveria F, editors. Minimal processing of foods and process optimization. Boca Raton, FL: CRC Press; 1994. pp. 407–416. [Google Scholar]
- Chaudhary P, Ray RC. Fermentation of jamun (Syzgium cumini L.) fruits to form red wine. Asean Food J. 2007;14:15–23. [Google Scholar]
- Daniels JA, Krishhnamurthi R, Rizvi SSH. Effects of carbon dioxide on microbial growth and food quality: A review. J Food Prot. 1985;48:532–537. doi: 10.4315/0362-028X-48.6.532. [DOI] [PubMed] [Google Scholar]
- De Ancos B, Gonzalez E, Cano MP. Effect of high-pressure treatment on the carotenoid composition and the radical scavenging activity of persimmon fruit purees. J Agri Food Chem. 2000;48:3542–3548. doi: 10.1021/jf990911w. [DOI] [PubMed] [Google Scholar]
- Del Caro A, Piga A, Vacca V, Agabbio M. Changes of flavonoids, Vitamin C and antioxidant capacity in minimally processed citrus segments and juices during storage. Food Chem. 2004;84:99–105. doi: 10.1016/S0308-8146(03)00180-8. [DOI] [Google Scholar]
- Deza MA, Araujo M, Garrido MJ. Inactivation of Escherichia coli 0157:H7, Salmonella enteritidis and Listeria monocytogenes on the surface of tomatoes by neutral electrolyzed water. Lett Appl Microbial. 2003;37:482–487. doi: 10.1046/j.1472-765X.2003.01433.x. [DOI] [PubMed] [Google Scholar]
- Dixon RA, Paiva NL. Stress-induced phenylpropanoid metabolism. The Plant Cell. 1995;7:1085–1097. doi: 10.2307/3870059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gil MI, Artes F, Tomas-Barberan FA. Minimal processing and modified atmosphere packaging effects on pigments of pomegranate seeds. J Food Sci. 1996;61:161–164. doi: 10.1111/j.1365-2621.1996.tb14749.x. [DOI] [Google Scholar]
- Gil M, Gorny J, Kader A. Response of ‘Fuji’ apples slices to ascorbic acid treatments and low-oxygen atmospheres. Hort Science. 1998;33(2):305–309. [Google Scholar]
- Grisebach H. Biosynthesis of anthocyanins. In: Markakis P, editor. Anthocyanins as food colors. New York: Academic Press; 1982. pp. 69–92. [Google Scholar]
- Hasan SM, Hossain MM, Faruque A, Mazumder MEH, Rana MS, Akter R, Alam MA. Comparison of antioxidant potential of different fractions of commelina benghalensis Linn. Bang. J Life Sci. 2008;20(2):9–16. [Google Scholar]
- Kader AA, Zagory D, Kerbel EL. Modified atmosphere packaging of fruits and vegetables. Crit Rev Food Sci Nutr. 1989;28:1–30. doi: 10.1080/10408398909527490. [DOI] [PubMed] [Google Scholar]
- Kang H, Saltveit ME. Antioxidant capacity of lettuce leaf tissue increases after wounding. J Agric Food Chem. 2002;50:7536–7541. doi: 10.1021/jf020721c. [DOI] [PubMed] [Google Scholar]
- Kim D, Smith N, Lee C. Apple cultivar variations in response to heat treatment and minimal processing. J Food Sci. 1993;58(5):1115–1118. doi: 10.1111/j.1365-2621.1993.tb06127.x. [DOI] [Google Scholar]
- Klein BP. Nutritional consequences of minimal processing of fruits and vegetables. J Food Qual. 1987;10:795–798. doi: 10.1111/j.1745-4557.1987.tb00857.x. [DOI] [Google Scholar]
- Klein BP, Perry AK. Ascorbic-acid and vitamin-A activity in selected vegetables from different geographical areas of the United States. J Food Sci. 1982;47(3):941–945. doi: 10.1111/j.1365-2621.1982.tb12750.x. [DOI] [Google Scholar]
- Lana MM, Tijskens LMM. Effect of cutting and maturity on antioxidant activity of fresh-cut tomatoes. Food Chem. 2006;97(2):203–211. doi: 10.1016/j.foodchem.2005.03.037. [DOI] [Google Scholar]
- Loewus FA, Loewus MW. Biosynthesis and metabolism of ascorbic acid in plants. Crit Rev Plant Sci. 1987;5:101–119. doi: 10.1080/07352688709382235. [DOI] [Google Scholar]
- Lougheed EC. Interactions of oxygen, carbon dioxide, temperature, and ethylene that may induce injuries in vegetables. Hort Sci. 1987;22:791–794. [Google Scholar]
- Luck E, Jager M. Antimicrobial food additives. 2. New York: Springer; 1997. [Google Scholar]
- Matsukawa R, Dubinsky Z, Kishimoto E, Masak K, Masuda Y, Takeuchi T. A comparison of screening methods for antioxidant activity in seaweeds. J Appl Phy. 1997;9:29–35. doi: 10.1023/A:1007935218120. [DOI] [Google Scholar]
- McDonald S, Prenzler PD, Antolovich M, Robards K. Phenolic content and antioxidant activity of olive extracts. Food Chem. 2001;73:73–84. doi: 10.1016/S0308-8146(00)00288-0. [DOI] [Google Scholar]
- Nagata M, Yamashita I. Simple method for simultaneous determination of chlorophyll and carotenoids in tomato fruit. J Japan Soc Food Sci Technol. 1992;39:925–928. [Google Scholar]
- Paull RE. Effect of temperature and relative humidity on fresh commodity quality. Postharv Bio Technol. 1999;15:263–277. doi: 10.1016/S0925-5214(98)00090-8. [DOI] [Google Scholar]
- Rahman MAA, Moon SS. Antioxidant polyphenol glycosides from the plant draba nemorosa. Bull Korean Chem Soc. 2007;28(5):827–831. doi: 10.5012/bkcs.2007.28.5.827. [DOI] [Google Scholar]
- Rai DR, Jha SN, Wanjari OD, Patil RT. Chromatic changes in broccoli under modified atmospheres in perforated film packages. Food Sci Tech Int. 2009;15(4):0387–395. doi: 10.1177/1082013209346587. [DOI] [Google Scholar]
- Reynertson KA, Yang H, Jiang B, Basile MJ, Kennelly MEJ. Quantitative analysis of antiradical phenolic constituents from fourteen edible myrtaceae fruits. Food Chem. 2008;109(4):883–890. doi: 10.1016/j.foodchem.2008.01.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rice-Evants CA, Miller NJ, Paganga G. Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Radic Biol Med. 1996;20:933–956. doi: 10.1016/0891-5849(95)02227-9. [DOI] [PubMed] [Google Scholar]
- Sharma SB, Nasir A, Prabhu KM, Murthy PS, Dev G. Hypoglycaemic and hypolipidemic effect of ethanolic extract of seeds of Eugenia jambolana in alloxan-induced diabetic rabbits. J Ethnopharmacol. 2003;85(2–3):201–206. doi: 10.1016/S0378-8741(02)00366-5. [DOI] [PubMed] [Google Scholar]
- Singh S, Krishnamurthi S, Katyal SL (1967) Fruit Culture in India, ICAR, New Delhi, p 225
- Swain T, Hillis WE. The phenolic constituents of prunus domestica 1. The quantitative analysis of phenolic constituents. J Sci Food Agri. 1959;10:63–68. doi: 10.1002/jsfa.2740100110. [DOI] [Google Scholar]
- Tamura H, Yamagami A. Antioxidative activity of monoacylated anthocyanins isolated from muscat bailey a grape. J Agri Food Chem. 1994;42:1612–1615. doi: 10.1021/jf00044a005. [DOI] [Google Scholar]
- Tano K, Arul J, Doyon G, Castaigne F. Atmospheric composition and quality of fresh mushrooms in modified atmosphere packages as affected by storage temperature abuse. J Food Sci. 1999;64:1073–1077. doi: 10.1111/j.1365-2621.1999.tb12285.x. [DOI] [Google Scholar]
- Timberlake CF, Henry BS. Anthocyanins as natural food colourants, plant flavonoids in biology and medicine II: Biochemical, cellular and medicinal properties. New York: Liss; 1988. pp. 107–121. [Google Scholar]
- Toivonen PMA. The effects of storage temperature, storage duration, hydro-cooling, and micro-perforated wrap on shelf-life of broccoli (Brassica oleracea L., Italica Group) Postharvest Biol Technol. 1997;10:59–65. doi: 10.1016/S0925-5214(97)87275-4. [DOI] [Google Scholar]
- Tomkins RB, Cumming BA. Effect of pre-packaging on asparagus quality after simulated transportation and marketing. Sci Horti. 1988;36:25–35. doi: 10.1016/0304-4238(88)90004-0. [DOI] [Google Scholar]