Table 1.
The fluorophore-substituted compounds photophysical properties and abilities to inhibit pili- and curli-dependent biofilm formation. 
| ID | R1 (C8) | R2 (C7) | X | EC50[a] Pili [μM] | EC50[a] Curli [μM] | λabs[b,c] [nm] | λfl[b,d] [nm] | Quantum Yield[b] ΦF [%] (λex nm) |
|---|---|---|---|---|---|---|---|---|
| 11 a |
|
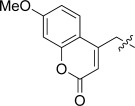 |
-S- | >200 | NA[e] | 328 | 394 | 5 (330)[f] |
| 11 b | 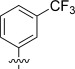 |
 |
-S- | >200 | NA[e] | 330 | 420 | 1 (330)[f] |
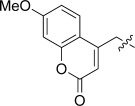
| 11 c |  |
-S- | >200 | NA[e] | 328 | 396 | 0.7 (330)[f] | |
| 11 d | 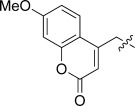 |
-S- | >200 | NA[e] | 328 | 413 | 0.4 (330)[f] | |
| 16 a | 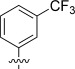 |
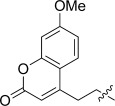 |
-S- | 65 | 175 | 329 | 430 | 0.6 (355)[f] |
| 16 b | 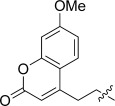 |
-S- | 156 | NA[e] | 327 | 393 | 0.5 (330)[f] | |
| 16 c | 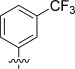 |
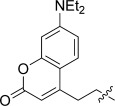 |
-S- | 18 | 25 | 393 | 474 | 15 (390)[g] |
| 16 d | 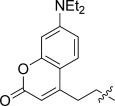 |
-S- | 12 | NA[e] | 392 | 484 | 6 (330)[f] | |
| 23 | 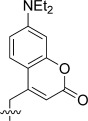 |
 |
-S- | 5 | 17 | 396 | 478 | 11 (346)[f] |
| 28 | 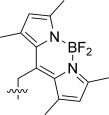 |
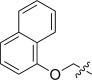 |
-S- | 4 | 14 | 506 | 524 | 10 (470)[h] |
| 31 | 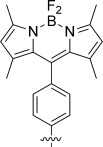 |
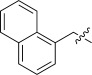 |
-S- | 14 | 12 | 502 | 514 | 67 (470)[h] |
| 34 | 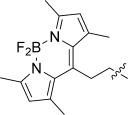 |
-S- | 10 | 24 | 498 | 509 | 11 (480)[h] | |
| 37 | 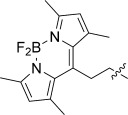 |
-O- | 13 | 14 | 497 | 531 | 27 (480)[h] | |
| 38 | 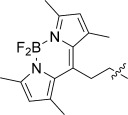 |
-S(O)- | 29 | 40 | 498 | 516 | 71 (480)[h] | |
| 3 | 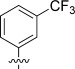 |
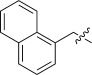 |
-S- | 17 | 38 | – | – | – |
[a] Estimated from 16–32 data points on every concentration. [b] All substances were dissolved in DMSO and subsequently diluted in phosphate buffer at pH 7.0. The samples DMSO concentrations never exceed 5 wt %. The sample concentrations in the DMSO stock solutions are adjusted so that the final samples never have a peak absorbance higher than 0.1. [c] Wavelengths of the peak absorption. [d] The peak fluorescence. [e] Not active. [f] Reference: POPOP in MeOH. [g] Reference: Perylene in cyclohexane. [h] Reference: Rhodamine 6G in water.
