Figure 6. Enhanced presentation of iFt persists in the presence of anti-iFt mAb.
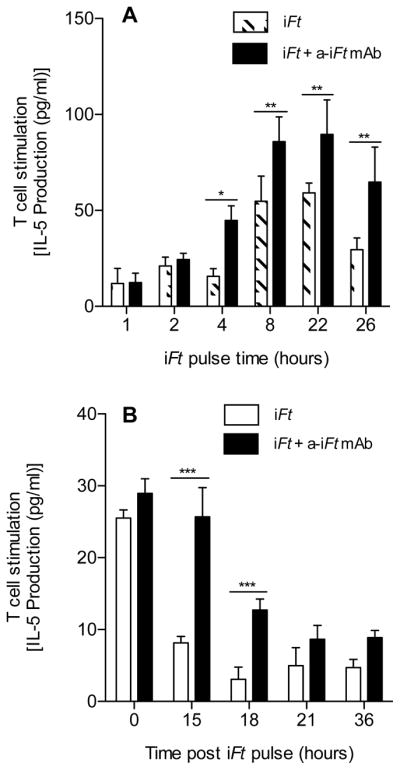
Balb/c MØs (2×105 cells) were cultured in media alone or media containing 1×106 iFt organisms in the presence or absence of 1 μg/ml of anti-iFt mAb at 37°C in 5% CO2 for 0, 1, 2, 4, 8, 22 or 26 hours. At each time point the cells were then fixed for 15 minutes at room temperature in 1% paraformaldehyde, washed, and stored at 4°C until all time points were completed. Cells were then counted and adjusted to 2×106 cells/ml, 100 μl of the latter were then added to the wells of a 96 well plate containing the Ft-specific T cell hybridoma at 1×105 cells/well. The plate was then incubated at 37°C in 5% CO2 in a humidity chamber for 24 hours, the supernatant was collected, and assayed for IL-5 using CBA. The average of three independent experiments is presented (A). Balb/c MØs (2×105 cells) were cultured in media alone or media containing 1×106 iFt organisms in the presence or absence of 1 μg/ml of anti-iFt mAb for 8 hours at 37°C in 5% CO2. After the incubation, the cells were washed 3 times with PBS and further incubated at 37°C in 5% CO2 for 0, 15, 18, 21 or 36 hours, at which point they were fixed with 1% paraformaldehyde, washed with PBS and then stored at 4°C until all time points were completed. Subsequently, cells were counted and adjusted to 2×106 cells/ml and 100 μl of the latter were then added to the wells of a 96 well plate containing the Ft-specific T cell hybridoma at 1×105 cells/well. The plate was incubated at 37°C in 5% CO2 in a humidity chamber for 24 hours and the supernatant was collected. The supernatants were assayed for IL-5 using CBA (B). Results are representative of three independent experiments. (*) p value < 0.05, (**) p value < 0.01, (***) p value < 0.001.
