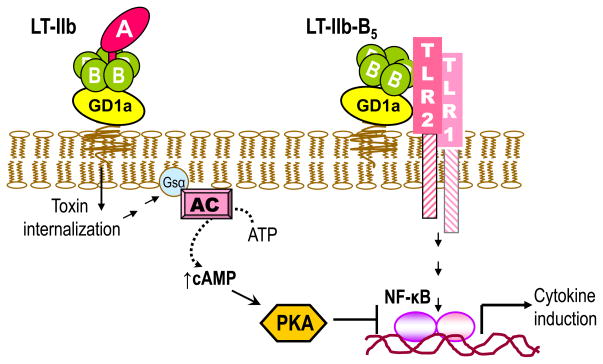
Figure 2. Differential and antagonistic effects on NF-κB activation by LT-IIb holotoxin and its B pentamer.
LT-IIb-B5 activates the TLR2/TLR1 heterodimer and induces NF-κB-dependent production of proinflammatorry cytokines (Liang et al., 2009a; Liang et al., 2007b). In contrast, the holotoxin does not interact with TLR2/TLR1 due to A subunit-dependent steric hindrance (Liang et al., 2009a; Liang et al., 2007b). However, upon GD1a binding and internalization of the holotoxin, the ADP-ribosyltransferase activity of its A subunit activates the Gsα component of adenylate cyclase (AC). This leads to elevation of intracellular cAMP, activation of cAMP-dependent protein kinase A (PKA), and inhibition of NF-κB-dependent transcription of proinflammatory cytokines (e.g., TNF-α) (Liang et al., 2007b).