Table 3.
Results From Univariable Meta-regression on Log Odds of Sustained Virologic Responsea,b
| Variable | No. ofStudies | CoefficientEstimate | 95% CI | P Valuec | PearsonCorrelationCoefficient | HeterogeneityParameter (I2)d | PValue |
|---|---|---|---|---|---|---|---|
| Treatment of addiction during HCV therapy | 26 | −0.0426 | −.9784, .8932 | .9289 | −0.0468 | 0.2014 (0.7020) | <.0001 |
| Genotype 1 or 4 | 26 | −1.7062 | −2.7413, −.6712 | .0012* | −0.7058 | 0.1349 (0.7031) | <.0001 |
| Human immunodeficiency virus | 22 | −1.5767 | −2.8753, −.2781 | .0173* | −0.5921 | 0.1418 (0.6794) | <.0001 |
| Location in United States | 32 | −0.5847 | −1.1294, −.0401 | .0354* | −0.4929 | 0.2077 (0.7777) | <.0001 |
| Support services offered | 31 | −0.0245 | −.4556, .4067 | .9114 | −0.0967 | 0.2343 (0.7909) | <.0001 |
| Multidisciplinary team involved | 32 | 0.0626 | −.3506, .4759 | .7665 | 0.0830 | 0.2265 (0.7800) | <.0001 |
| Methadone maintenance during HCV treatment | 20 | −0.4157 | −1.1916, .3602 | .2937 | −0.4871 | 0.163 (0.6029) | .0002 |
| Study design (randomized or matched control vs other) | 32 | −0.1484 | −.7701, .4733 | .6399 | 0.0747 | 0.2133 (0.7847) | <.0001 |
| Age | 20 | −0.0088 | −.0486, .0311 | .6656 | −0.3288 | 0.1191 (0.5187) | .0025 |
| Male | 22 | 0.1513 | −1.3726, 1.6752 | .8457 | 0.0123 | 0.2541 (0.7993) | <.0001 |
| Caucasians from US studies | 6 | 0.0424 | −1.1812, 1.2659 | .9459 | 0.0987 | 0.1405 | .1386 |
| African American from US studies | 4 | −4.8407 | −11.1615, 1.4801 | .1333 | −0.8544 | 0.039 | .2711 |
| Psychiatric comorbidities | 14 | −0.151 | −1.1111, .8092 | .758 | −0.3007 | 0.1704 (0.7117) | <.0001 |
| Substitution therapy at baselinee | 24 | −0.246 | −1.2266, .7345 | .6229 | −0.1652 | 0.2163 (0.7224) | <.0001 |
| Drug use during HCV treatment | 15 | 0.346 | −.1833, .8754 | .2001 | 0.3777 | 0.0462 | .0887 |
| Biopsy performed | 14 | −0.2575 | −.9557, .4407 | .4697 | −0.1952 | 0.071 (0.4498) | .0228 |
Abbreviations: CI, confidence interval; HCV, hepatitis C virus.
a Results listed in order as presented in the text.
b Results from the univariable meta-regression expressed as 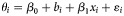 , where θi is the log odds for achieving a sustained virologic response from study i, β0 is intercept, bi is random effect for study i, xi is the value of the covariate from study i, and εi is the within study error. The covariates are expressed either as proportions (treatment of addiction during HCV therapy, substitution therapy at baseline, drug use during HCV treatment, genotype 1 or 4, human immunodeficiency virus infection, male, African American from US studies, Caucasians from US studies, psychiatric comorbidities, biopsy performed, methadone maintenance during HCV treatment), as categorical variables (study design, location in United States, multidisciplinary team involved, support services offered) or as continuous variables (median/mean age).
, where θi is the log odds for achieving a sustained virologic response from study i, β0 is intercept, bi is random effect for study i, xi is the value of the covariate from study i, and εi is the within study error. The covariates are expressed either as proportions (treatment of addiction during HCV therapy, substitution therapy at baseline, drug use during HCV treatment, genotype 1 or 4, human immunodeficiency virus infection, male, African American from US studies, Caucasians from US studies, psychiatric comorbidities, biopsy performed, methadone maintenance during HCV treatment), as categorical variables (study design, location in United States, multidisciplinary team involved, support services offered) or as continuous variables (median/mean age).
c Significant P values are indicated with an asterisk.
d I2 added if heterogeneity is present.
e Substitution therapy included patients on either methadone or buprenorphine.
