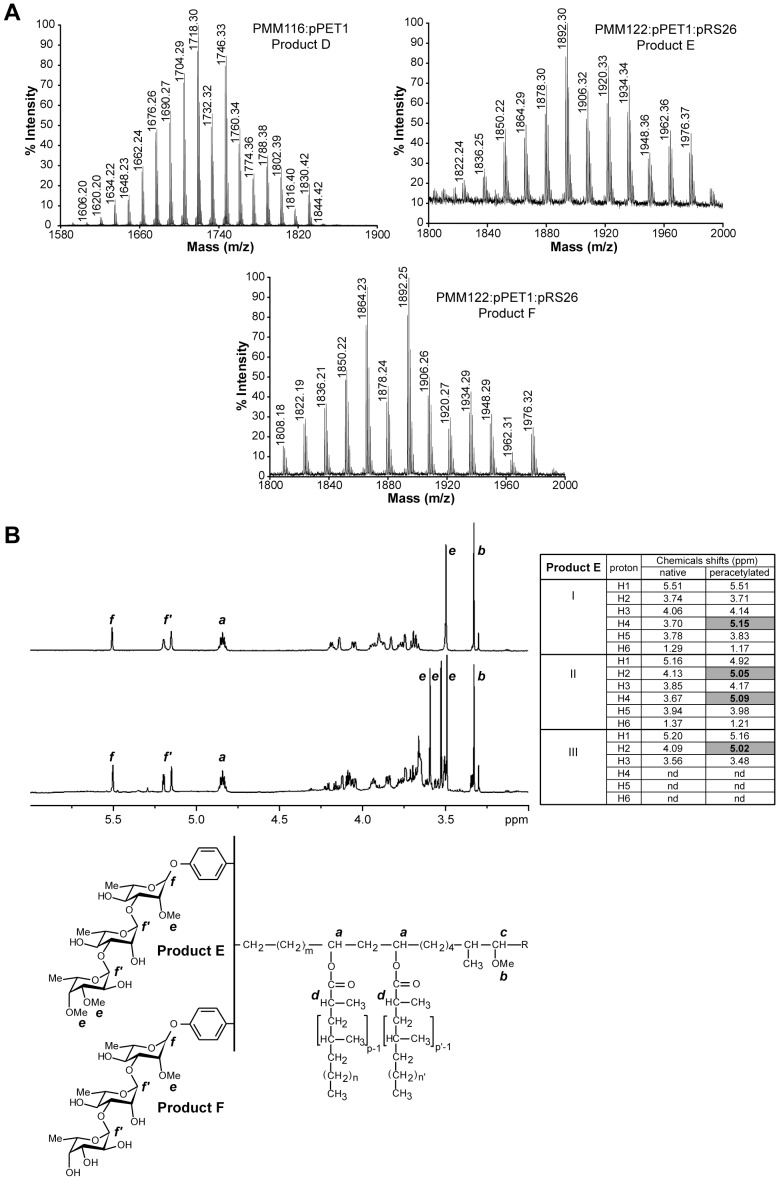
Figure 6. Biochemical analysis of products D, E and F.
(A) MALDI-TOF mass spectra of purified product D from the PMM116:pPET1 mutant strain and of purified products E and F from the PMM122:pPET1:pRS26 mutant strain. (B) NMR analysis of products E and F. 1D 1H-NMR spectra (3.0–6.0 ppm) of native glycolipids E (bottom) and F (upper) (600 MHz, in CDCl3). The structures of the analyzed compounds are shown below the spectrum and the protons corresponding to the main signals are indicated. The table summarizes the assignments of resonances on the basis of chemical shift correlations deduced from the 2D-COSY spectra of native and per-O-acetylated compound E. Proton resonances shifted by acetylation are written in bold (grey cells). The first column of the table indicates the sugar residue: I, first rhamnosyl residue; II, second rhamnosyl residue; III, terminal fucosyl residue.