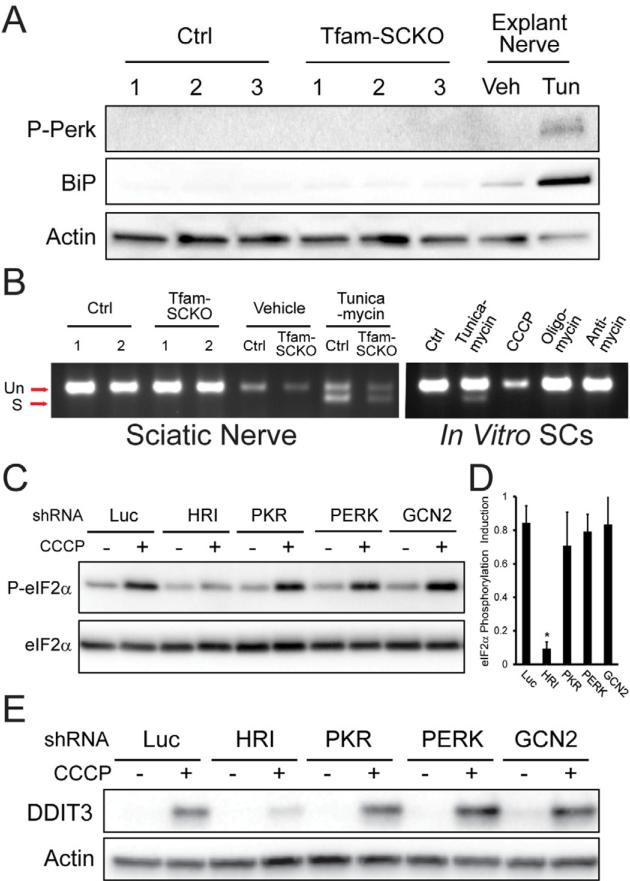
Figure 3. Mitochondrial dysfunction-induced ISR activation in SCs is mediated by heme-regulated inhibitor (HRI) kinase independent of ER-stress.
(A) Immunoblot analysis of the phosphorylation (activation) status of the ER-stress sensor Perk and the UPR-induced molecular chaperone BiP/Grp78 in 2-month-old Ctrl and Tfam-SCKO nerves (3 independent mice per genotype) shows no differences, indicating that ISR activation following SC mitochondrial deficits does not involve ER-stress. Tunicamycin (Tun) treatment of sciatic nerves cultured as explants, serves as a positive control for ER-stress. Veh, vehicle. (B) Gel showing the absence of Xbp-1 splicing downstream of the activation of the ER-stress sensor Ire-1 in 2-month-old Tfam-SCKO nerves, or SCs treated with mitochondrial inhibitors, confirms that ISR activation induced by mitochondrial derangement is independent of ER-Stress. Tunicamycin treatment of cultured SCs or sciatic nerves cultured as explants serve as positive controls for ER-stress. (C) Immunoblot analysis of eIF2α phosphorylation in 3T3 cells expressing shRNA to the indicated eIF2α kinases (HRI, PKR, PERK, GCN2) that were treated for three hrs with 5 μM CCCP to inhibit mitochondrial respiration. Knockdown of HRI (but not of GCN2, PKR or PERK) is sufficient to prevent eIF2α phosphorylation following inhibition of mitochondrial respiration, indicating the specific role of HRI in this process. (D) Quantification of the immunoblot as shown in (C) by densitometry. N=3 independent experiments. (E) Immunoblot analysis of the induction the ISR mediator DDIT3/CHOP 6 hrs after inhibition of mitochondrial respiration with 5 μM CCCP in 3T3 cells in which expression of indicated eIF2a kinase (HRI, PKR, PERK, GCN2) was knocked down using shRNA. Knockdown of HRI (but not of GCN2, PKR or PERK) is sufficient to prevent DDIT3/CHOP induction downstream of eIF2α phosphorylation following inhibition of mitochondrial respiration.