Figure 1.
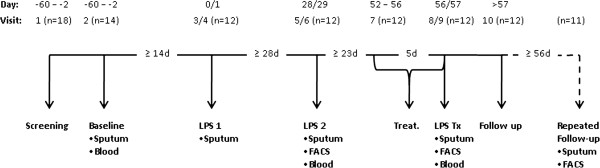
Study design. LPS: inhalation of 2 μg (20,000 E.U.) nebulized Lipopolysaccharide. Treat: Oral administration (500 μg/day) of the PDE-4 inhibitor Roflumilast. FACS: Flow cytometry of sputum cells was performed. In a separate study performed >56 days after the end of the LPS challenge trial, 11 subjects underwent a follow-up sputum induction. (Visits 4, 6, and 9 refer to phone calls done 24 h after the respective challenges).
