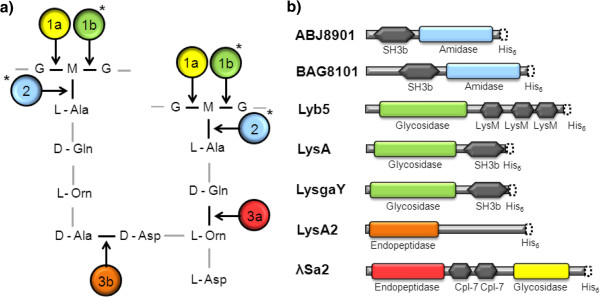
Figure 1.
Schematic representation of PG, putative lysin catalytic sites and domain structures of bacteriophage lysins. a) Fragment of the repeat structure of Lactobacillus fermentum PG with a D-Asp interpeptide bridge with known enzymatic cut sites: (1a) λSa2 endolysin; (3a) λSa2 endolysin; (3b) LysA2, and predicted lysin catalytic sites: (1b*) Lyb5, LysgaY, and LysA; (2*) ABJ8901 [GenBank: ABJ63875] and BAG8101 [GenBank:BAG27815]. * Predicted catalytic sites are based on amino acid homologies to other biochemically characterized enzymes. b) Lysin architecture consisting of an enzymatically active domain(s) (square box) and cell wall binding domain(s) (hexagon box) drawn nearly to scale predicted using the NCBI Conserved Domain Database. A His6-tag (dot box; not to scale) was fused on the C-terminal for metal ion affinity chromatography purification.