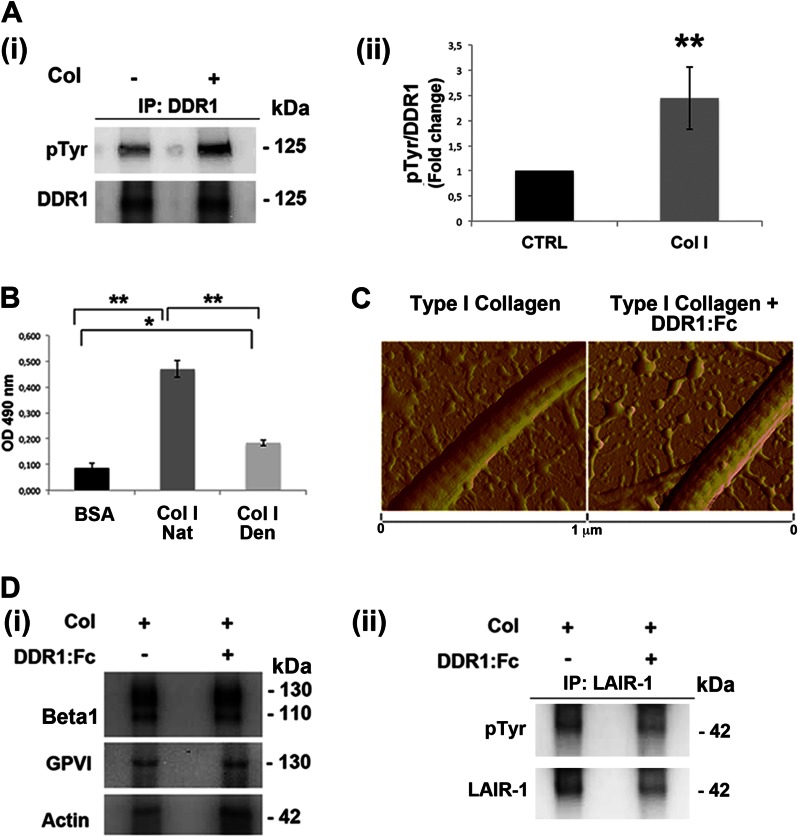
FIGURE 2.
Type I collagen-dependent DDR1 activation. A, panel i, lysates of MKs, plated on tissue culture plastic or on type I collagen (Col) for 16 h, were immunoprecipitated (IP) with an anti-DDR1 antibody and subjected to Western blotting. Membranes were stained with a monoclonal antibody against phosphotyrosine and with anti-DDR1 antibody. Panel ii, densitometry analysis of the Western blots of 125-KDa phospho-Tyr (pTyr) band on DDR1 immunoprecipitates. B, binding assay to evaluate the DDR1-Fc binding affinity on native type I collagen (Nat Col I) with respect to denatured type I collagen (Den Col I). BSA was used as negative control. Optical density (OD) was measured at 490 nm. C, atomic force microscopy images of dehydrated collagen-coated coverslips in the presence or absence of DDR1-Fc showing similar banded fibrils. D, panel i, MKs were plated, for 2 h, on type I collagen, in the presence or absence of DDR1-Fc. Lysates were subjected to Western blot analysis of active β1 integrin (HUTS-4) and dimeric GPVI. Actin was probed to show equal loading. Panel ii, lysates of megakaryocyte treated as above described were immunoprecipitated with anti-LAIR-1 antibody and subjected to Western blot. Membranes were stained with antibodies anti-phospho-Tyr (pTyr) and anti-LAIR-1. Shown here are representative Western blots out of three independent experiments. Data are presented as means ± S.D. (n = 5 and 4 independent experiments). *, p < 0.05. **, p < 0.01.