Figure 5.
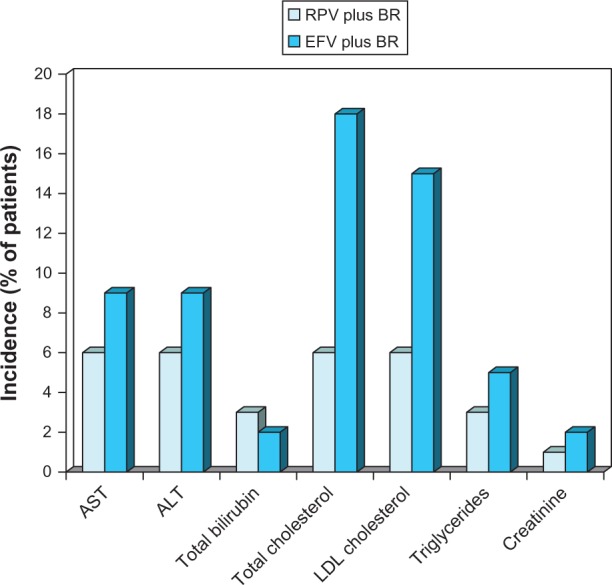
Laboratory abnormalities and changes in blood lipids associated with oral rilpivirine as a component of combination therapy in antiretroviral-naïve patients with HIV infection.
Notes: Pooled descriptive data10 from the ECHO24 and THRIVE trials25 are for severity-level grades 2–4 laboratory abnormalities or changes in blood lipids occurring in ≥2% of patients in at least one group after 48 weeks of treatment. Values shown as <1 at a particular grade level of severity in the original reference were rounded up to 1 when summing to obtain totals for grade 2–4 levels.
Abbreviations: BR, background regimen; EFV, efavirenz; LDL, low-density lipoprotein; RPV, rilpivirine; AST, aspartate aminotransferase; ALT, alanine aminotransferase.
