Summary
Background and objectives
Candidates with AKI including hepatorenal syndrome often recover renal function after successful liver transplantation (LT). This study examined the incidence and risk factors associated with renal nonrecovery within 6 months of LT alone among those receiving acute renal replacement therapy (RRT) before LT.
Design, setting, participants, & measurements
Scientific Registry of Transplant Recipients data were linked with Centers for Medicare and Medicaid Services ESRD data for 2112 adult deceased-donor LT-alone recipients who received acute RRT for ≤90 days before LT (February 28, 2002 to August 31, 2010). Primary outcome was renal nonrecovery (post-LT ESRD), defined as transition to chronic dialysis or waitlisting or receipt of kidney transplant within 6 months of LT. Cumulative incidence of renal nonrecovery was calculated using competing risk analysis. Cox regression identified recipient and donor predictors of renal nonrecovery.
Results
The cumulative incidence of renal nonrecovery after LT alone among those receiving the pre-LT acute RRT was 8.9%. Adjusted renal nonrecovery risk increased by 3.6% per day of pre-LT RRT (P<0.001). Age at LT per 5 years (P=0.02), previous-LT (P=0.01), and pre-LT diabetes (P<0.001) were significant risk factors of renal nonrecovery. Twenty-one percent of recipients died within 6 months of LT. Duration of pretransplant RRT did not predict 6-month post-transplant mortality.
Conclusions
Among recipients on acute RRT before LT who survived after LT alone, the majority recovered their renal function within 6 months of LT. Longer pre-LT RRT duration, advanced age, diabetes, and re-LT were significantly associated with increased risk of renal nonrecovery.
Introduction
Liver transplantation (LT) candidates with severe renal dysfunction, and especially those receiving pre-LT renal replacement therapy (RRT), were previously thought to be too sick to be transplanted. Adoption of Model for End-Stage Liver Disease (MELD)–based allocation in the United States in 2002 for deceased donor livers has led to the reconsideration of LT for candidates with renal dysfunction. Since then, the proportion of candidates with renal dysfunction or on RRT at LT has increased by 25% (1). The incidence of simultaneous liver and kidney transplantation (SLKT) has almost doubled since the implementation of MELD-based liver allocation policy, resulting in taking away deceased donor kidneys from candidates awaiting kidney transplant (2).
The spectrum of renal dysfunction among LT candidates varies from slight elevation in serum creatinine due to prerenal or renal disease to full-blown renal failure from hepatorenal syndrome (HRS) requiring RRT. In the absence of LT, the outcomes of these candidates are poor, and are associated with very high mortality rates (1). Pre-LT medical management to reduce portal pressure and the need for RRT are not very successful, but are often used to bridge patients to LT (3–8).
Although candidates with AKI including HRS may recover renal function after LT (7–9), the rate and timing of renal recovery have not been very well elucidated. One study showed the renal nonrecovery to be as high as 32% among LT recipients who were on pre-LTRRT and received LT alone (9). However, this study did not distinguish between the candidates receiving acute and chronic RRT before LT. Our primary and secondary objectives were to assess the reversibility of AKI among those who were on acute RRT for ≤90 days before LT and to determine the minimum threshold of the duration of pre-LT RRT above which the risk of renal nonrecovery was substantially increased. Therefore, we aimed to determine the incidence and risk factors for renal nonrecovery (ESRD) within 6 months of LT alone among those who were on acute RRT for ≤90 days before LT.
Materials and Methods
Data Sources and Study Population
This study used data from the Scientific Registry of Transplant Recipients (SRTR), the Centers for Medicare and Medicaid Services (CMS) ESRD program, and the Social Security Death Master File (SSDMF) (10). The SRTR maintains a database of all candidates listed for and recipients of solid-organ transplants in the United States. Candidates on the waiting list and recipients of solid-organ transplants are tracked on a periodic basis; data are submitted to the Organ Procurement and Transplantation Network (OPTN). The SRTR supplements information on vital status with data on deaths from the Social Security Death Master File and the Medicare Beneficiary Database. The latter is maintained by CMS and contains data on all patients treated for ESRD in the United States (Medicare beneficiaries and other patients with ESRD who have received maintenance RRT), including demographics, treatment, hospitalization, and costs. The CMS ESRD database also includes records of any changes in vital status or method of renal replacement, including kidney transplantation.
Once listed for LT, a candidate’s MELD score must be re-certified periodically, and may be updated sooner than required by OPTN due to a change in clinical condition. Each transplant center is mandated to submit MELD laboratory results, including serum creatinine, serum bilirubin, international normalized ratio of prothrombin time, dialysis status (acute RRT at least twice or continuous RRT [CRRT] in the previous week), serum sodium, and diagnosis at each re-certification of MELD score (11). Failure to do so in accordance with the schedule would cause the candidate to be re-assigned to his or her previous lower MELD score.
The study population included candidates aged ≥18 years who were receiving acute RRT (≤90 days) and received LT alone between February 28, 2002 and August 31, 2010. Recipients of living-donor LT or SLKT and those on chronic RRT (>90 days) before LT were excluded. The linked data were obtained from SRTR under a data use agreement.
Statistical Analyses
Continuous and categorical variables were expressed as medians (quartile 1 [Q1] to quartile 3 [Q3]) and as percentages, respectively. Primary outcome was renal nonrecovery (post-LT ESRD) defined as transition to chronic dialysis (designation on a CMS-2728 form that the patient began treatment for ESRD ascertained by the first service date on CMS-2728) or listing or receipt of a kidney transplant within 6 months of LT. The CMS-2728 is a medical evidence form completed by the patient’s dialysis facility within 45 days of initiation of chronic dialysis treatments or by the kidney transplant program in the case of pre-emptive kidney transplant. We allowed for at least 3 months of reporting lag time after the end of the 6-month follow-up period to avoid the under-reporting of the primary outcome. The CMS-2728 form is not completed if the patient is in ARF or expected to recover renal function. Therefore, a patient who receives CRRT or a few courses of RRT in the immediate pre-, peri-, or post-transplant phase for some period of time after LT but recovers renal function would not be included in the CMS ESRD database. Because renal nonrecovery was the primary outcome of interest, and listing of kidney transplant is a surrogate for RRT, we accepted the earlier of transition to chronic dialysis treatments or listing for kidney transplant within 6 months of LT as evidence of renal nonrecovery.
The SRTR database captures presence or absence of acute RRT or CRRT in the previous week on each re-certification of MELD score (status update form). If a candidate was not on RRT on the previous status update and the new status update indicated RRT, then the candidate was deemed to have started RRT 7 days before the status update date. For candidates who were on dialysis at listing, 7 days were added to RRT duration because the SRTR does not capture the RRT start date. Patients were followed from the date of LT to the earliest of post-LT ESRD, death, or end of the follow-up period. The incidence rate was calculated as the number of ESRD events divided by total patient time, expressed as patient-years.
Given the nature of the data structure and our objectives, death is a competing risk, because post-LT death before the onset of ESRD precludes subsequent ESRD. Therefore, using standard competing risks methods, we modeled the cause-specific ESRD hazard (12). This can be thought of as the rate of ESRD incidence among patients while they are alive and ESRD free.
Covariate-adjusted Cox regression models were used to identify recipient and donor predictors of renal nonrecovery and post-LT mortality. Potential adjustment covariates included the following: recipient age, sex, race, diagnosis, MELD, pre-LT hypertension, diabetes, pre-LT RRT duration, hospitalization status at LT, donor age and sex, cold ischemia time, local versus shared organ (regionally, nationally), and donation after cardiac death.
Parsimonious models for each outcome were developed by choosing a subset of the covariates listed above based on clinical relevance and statistical significance; covariates with P<0.10 were retained in the final model. In addition to duration of pre-LT RRT, the renal nonrecovery model was adjusted for the following: recipient age, re-LT, pre-LT diabetes, donor age, cause of death, and donor sex. The post-LT mortality model was also adjusted for recipient age (linear term), race, hypertension, and cold ischemia time. Because we were interested specifically in the effect of pre-LT RRT duration, it was forced into the post-LT mortality model. All statistical analyses were conducted using SAS software (version 9.2; SAS Institute, Cary, NC).
Results
Patient Characteristics
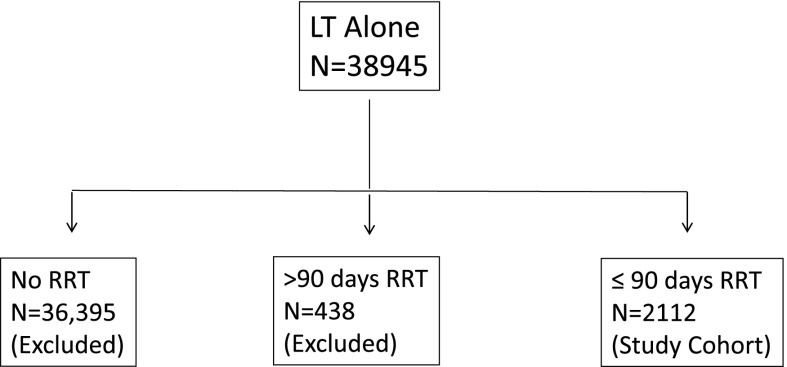
A total of 38,945 LTs alone were performed during the study period. To define the cohort of interest, we excluded 36,833 recipients for the absence of pre-LT RRT (n=36,394) and already receiving chronic RRT before LT (n=439). Our cohort consisted of 2112 LT recipients who were on acute RRT (≤90 days) before LT (Figure 1).
Figure 1.
Selection of cohort.
Table 1 shows the baseline characteristics of the cohort. This was a very sick group of LT recipients with a median MELD score of 39 at LT; 40% were on life support and 59% were hospitalized in the intensive care unit setting. The median pre-LT RRT duration was 11 days. Very few candidates (1.3%) had RRT for >41 days (Figure 2).
Table 1.
Baseline characteristics of recipients on acute RRT (≤90 days) at LT
| Factor | Median (Q1–Q3) or n (%) (N=2112) |
|---|---|
| Age at LT (yr) | 53 (46–58) |
| Male sex | 1275 (60) |
| Race | |
| Caucasian | 1475 (70) |
| African American | 160 (8) |
| Hispanic | 376 (18) |
| Other | 101 (5) |
| Hepatitis C | 768 (36) |
| Status-1a | 373 (18) |
| Hospitalized | |
| ICU | 1256 (59) |
| Non-ICU | 679 (32) |
| On life support | 854 (40) |
| Re-LT | 335 (16) |
| Pre-LT diabetes | 265 (13) |
| Laboratory MELD at LT | 39 (35–40) |
RRT, renal replacement therapy; LT, liver transplantation; ICU, intensive care unit; MELD, Model for End-Stage Liver Disease.
Status-1 is the highest priority on the waiting list category for the liver transplant candidates with fulminant liver failure with a life expectancy without a liver transplant of <7 days.
Figure 2.
Distribution of the duration of pre-LT RRT. LT, liver transplantation; RRT, renal replacement therapy.
Primary and Secondary Outcomes
Incidence of Renal Nonrecovery after LT.
There were 189 patients on pre-LT acute RRT who did not recover renal function after LT. The unadjusted crude rate of renal nonrecovery was 229 per 1000 patient-years. Using competing risk methods, the cumulative incidence of renal nonrecovery among those who survived after 6 months of LT alone was 8.9%. Table 2 shows the percentage of patients with renal nonrecovery and death within 6 months of LT, categorized by pre-LT RRT duration.
Table 2.
Percentage of recipients with renal nonrecovery and death within 6 months of LT by duration of pre-LT RRT
| Duration of Pre-LT RRT, d | Patients, n | Nonrecovery, n (%) | Died within 6 mo of LT, n (%) |
|---|---|---|---|
| Entire cohort | 2112 | 189 (8.9) | 443 (21.0) |
| <10 | 619 | 36 (5.8) | 157 (25.4) |
| 10–13 | 756 | 63 (8.3) | 152 (20.1) |
| 14–27 | 614 | 71 (11.6) | 110 (17.9) |
| 28–41 | 95 | 11 (11.6) | 18 (18.9) |
| >41 | 28 | 8 (28.6) | 6 (21.4) |
LT, liver transplantation; RRT, renal replacement therapy.
Recipient and Donor Factors Associated with Renal Nonrecovery after LT.
Most baseline recipient and donor characteristics were similar for LT recipients with and without renal recovery after LT. Age at LT (hazard ratio [HR], 1.10 per 5 years; 95% confidence interval [95% CI], 1.02 to 1.18; P=0.02), duration of pre-LT RRT (HR, 1.04 per day; 95% CI, 1.02 to 1.05; P<0. 001), re-LT (HR, 1.60; 95% CI, 1.10 to 2.33; P=0.01), and pre-LT diabetes (HR, 1.80; 95% CI, 1.27 to 2.56; P<0.001) were significant independent predictors of renal nonrecovery (Table 3). The duration of serum creatinine >1.5 mg/dl before pre-LT RRT was not associated with renal nonrecovery, and was not included in the final multivariable model.
Table 3.
Predictors of renal nonrecovery within 6 months of LT from a multivariable Cox regression model for LT recipients who were on RRT at LT
| Factor | HR (95% CI) | P |
|---|---|---|
| Recipient factors | ||
| Duration of pre-LT RRT (per day) | 1.04 (1.02 to 1.05) | <0.001 |
| Age at LT (per 5 yr) | 1.10 (1.02 to 1.18) | 0.02 |
| Re-LT | 1.60 (1.10 to 2.33) | 0.01 |
| Type 2 diabetes | 1.80 (1.27 to 2.56) | <0.001 |
| Donor factors | ||
| Cause of death | ||
| Head trauma | 1.00 | Reference |
| Stroke | 1.22 (0.87 to 1.70) | 0.25 |
| Anoxia | 0.86 (0.55 to 1.34) | 0.49 |
| Other | 2.57 (1.28 to 5.17) | 0.01 |
| Male donor | 1.41 (1.03 to 1.93) | 0.03 |
LT, liver transplantation; RRT, renal replacement therapy; HR, hazard ratio; 95% CI, 95% confidence interval.
We examined the functional form for the duration of pre-LT RRT in the nonrecovery model to evaluate a threshold at which the risk of nonrecovery is substantially increased. We plotted the parameter estimates for pre-LT RRT duration into various quantiles (tertiles, quartiles, deciles) and looked for patterns in Martingale residuals by pre-LT RRT duration. None of these methods indicated a threshold effect. The risk of renal nonrecovery increased in a dose-dependent manner with the increase in the pre-LT RRT duration. The cumulative incidence of renal nonrecovery within 6 months of LT ranged from 5.8% in the lowest tertile to 12.2% in the highest tertile of pre-LT RRT duration (Figure 3).
Figure 3.
Cumulative incidence of post-LT ESRD (renal nonrecovery) within 6 months of LT among patients on acute RRT at LT, stratified by the tertiles of duration of pre-LT RRT. LT, liver transplantation; RRT, renal replacement therapy.
Organs accepted from male donors were associated with a 41% higher risk of post-LT ESRD (Table 3). Donor-recipient sex mismatch in the renal nonrecovery model was not significant and was excluded from the final model.
Renal Function and Mortality at 6 Months after LT.
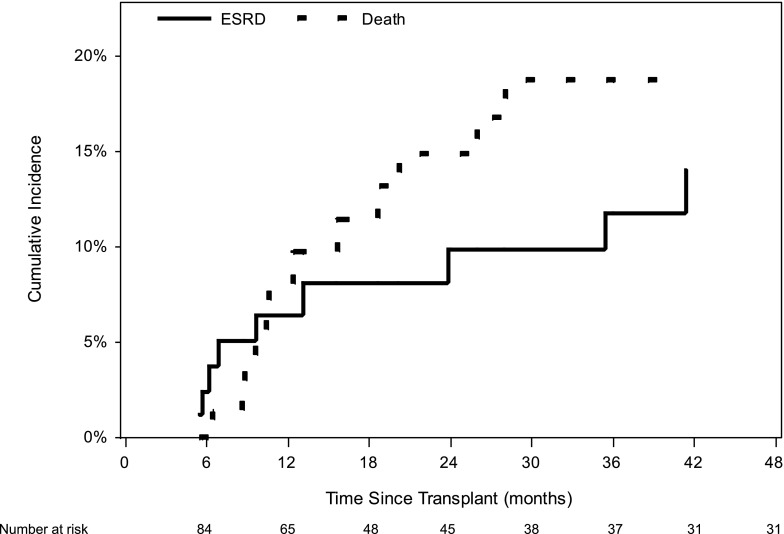
Table 4 shows recipient status at 6 months after LT. Only 4.0% of patients had stage 4 CKD at 6 months after LT. Another clinically relevant unanswered question was whether the majority of stage 4 CKD patients at 6 months progress to ESRD over time, given the 6-month follow-up time. The subset analysis restricted to these patients showed a post-LT ESRD cumulative incidence of 6.4% at 1 year (Figure 4).
Table 4.
Recipient status at 6 months post-LT
| Status at 6 mo after LT | Recipients, n (%) |
|---|---|
| CKD stage (eGFR, ml/min per 1.73 m2) | |
| 1 (90≤ eGFR) | 182 (8.6) |
| 2 (60≤ eGFR <90) | 445 (21.1) |
| 3 (30≤ eGFR <60) | 691 (32.7) |
| 4 (15≤ eGFR<30) | 84 (4.0) |
| 5 (nonrecovery) | 163 (7.7) |
| Re-transplant ≤6 mo after LTa | 46 (2.2) |
| Death ≤6 mo after LTa | 443 (21.0) |
| Unknown renal function | 58 (2.7) |
| Total | 2112 (100.0) |
The definition of CKD is based upon the Kidney Disease Outcomes Quality Initiative Clinical Practice Guidelines. LT, liver transplantation; eGFR, estimated GFR based upon the Modification of Diet in Renal Disease four-variable equation.
Participants who died or were re-transplanted within 6 months of the index liver transplant were not evaluable for CKD stage or ESRD status.
Figure 4.
Cumulative incidence of post-LT ESRD (renal nonrecovery) and death after 6 months of LT among patients with stage 4 CKD at 6 months after LT alone. LT, liver transplantation.
Our results showed that 443 recipients (21%) died and 46 required re-LT within 6 months of LT. The median time to death, among those that died within 6 months of LT, was 30 days (Q1=5 days, Q3=82 days). The recipient and donor factors independently associated with high 6-month post-LT mortality were recipient age at LT >60 years (HR, 1.07 per year >60 years; 95% CI, 1.03 to 1.11; P<0.001), life support at LT (HR, 1.78; 95% CI, 1.47 to 2.16; P<0.001), re-LT (HR, 2.12; 95% CI, 1.71 to 2.62; P<0.001), initial immunosuppression without induction therapy (HR, 1.35; 95% CI, 1.07 to 1.71; P=0.01), donor age >40 years (HR, 1.09 per 5 years >40; 95% CI, 1.04 to 1.15; P<0.001), and other donor cause of death (versus head trauma) (HR, 1.85; 95% CI, 1.09 to 3.11; P=0.02).
The duration of pre-LT RRT was not predictive of 6-month post-LT mortality (HR, 0.99; 95% CI, 0.98 to 1.01; P=0.24). Moreover, the presence of renal nonrecovery did not affect the 6-month post-LT mortality (HR, 0.93; 95% CI, 0.60 to 1.42; P=0.73).
Discussion
This is the largest study reporting the outcomes and evaluating the epidemiology of renal dysfunction after LT among liver-alone recipients with AKI who were on acute RRT before LT. Our results demonstrate that among LT recipients who required acute RRT pretransplant and survived after LT, only 9% had renal nonrecovery and needed chronic RRT within 6 months after LT. An even smaller proportion had stage 4 CKD at 6 months after LT. These results are based upon prevailing practice patterns and listing characteristics and validate the soundness of clinical decision making and judgments regarding LT alone among those on acute RRT in the current MELD-based allocation system.
A retrospective study consisting of a heterogeneous population of LT candidates with CKD and AKI showed that >12 weeks of renal dysfunction defined as serum creatinine >1.5 mg/dl was associated with post-LT CKD at 1 year (13). In contrast, our cohort consisted of homogeneous LT candidates with AKI on acute RRT. Our analysis did not show an association between the duration of serum creatinine >1.5 mg/dl before pre-LT RRT and renal nonrecovery.
One of our secondary objectives was to identify the minimum threshold effect of pre-LT RRT duration associated with renal nonrecovery so that such candidates could be listed for SLKT. Like several other studies (9,13–18), we also found a strong effect of duration of pre-LT RRT on renal nonrecovery. Each additional day of pre-LT RRT was associated with a 3.6% higher risk of renal nonrecovery, and we did not find a minimal threshold of pre-LT RRT duration above which the risk of renal recovery was especially increased.
There were very few patients with >41 days of pre-LT RRT in our cohort. The paucity of patients on RRT in excess of 41 days who received solitary LT is reflective of variation in clinical practice regarding the practice patterns of SLKT listing. A recent survey of US transplant centers demonstrated significant variation in the criteria used for SLKT among transplant centers (19). Approximately 73% of transplant centers took dialysis duration into account when selecting candidates with AKI for SLKT; however, pre-LT RRT duration varied among the transplant centers (>4, >6, >8 weeks of RRT) (20).
The decision of SLKT for candidates on acute RRT is driven by the concern over the likelihood of renal nonrecovery after LT alone and associated morbidity and mortality (18). The rising incidence of SLKT since the implementation of MELD-based allocation plateaued after the 2008 SLKT consensus conference (21). However, the minimum duration of AKI required for listing for SLKT is evolving. In 2006, Davis et al. recommended patients with AKI and/or HRS on RRT ≥6 weeks should be considered for SLKT (22). These recommendations were pushed back to ≥8 weeks’ duration of AKI/HRS based upon the retrospective analysis by Ruiz et al. and analysis of SRTR data (21,23). In 2009, OPTN proposed that sustained AKI (defined as GFR ≤25 ml/min) with or without RRT for ≥6 consecutive weeks should be considered for SLKT (24). Most recently, this duration is further reduced to ≥4 weeks AKI with or without RRT. Although only one-half of the patients who underwent SLKT had radionuclide scans to assess the recovery of native kidney, a higher proportion of native kidney recovery was seen in SKLT recipients who did not meet the OPTN criteria for SLKT compared with those who met the OPTN criteria (25).
A previous study demonstrated a 32% rate of renal nonrecovery (n=1041) (9). We expected a similar or higher rate of nonrecovery because of inclusion of re-LT candidates who may be at increased risk of renal nonrecovery and post-LT ESRD due to exposure to calcineurin inhibitors, but observed only a 8.9% nonrecovery rate among >2000 recipients who were on acute RRT before LT. It is likely that many patients on acute RRT before LT had reversible HRS, contributing to the low renal nonrecovery rate.
The objective of our study was to assess the reversibility of AKI secondary to decompensated liver failure and identification of the candidate characteristics associated with renal nonrecovery so that these candidates may be pre-emptively listed for SLKT. Therefore, we defined renal nonrecovery as transition to chronic RRT within 6 months of LT. Northup et al. defined renal recovery as freedom from dialysis after LT during the entire follow-up period (7). For example, a patient who recovered renal function shortly after LT alone and developed post-LT ESRD 3 years later due to the long-term contribution of other factors such as calcineurin inhibitor toxicity, hypertension, or diabetes-associated CKD would have been counted as having renal nonrecovery, as would a patient who transitioned to chronic dialysis shortly after LT without an intervening period of RRT-free survival (9). Finally, the authors did not address the post-LT mortality in their cohort. Despite these contrasting features, these two studies are complementary because they answer two different questions and add to our understanding of this complex area.
Our study found a small subset of patients with stage 4 CKD at 6 months after LT. Our speculation was that the majority of these patients might be on the verge of ESRD and may have high incidence of post-LT ESRD at 1 year. In contrast, analysis limited to this subgroup showed a low cumulative incidence of post-LT ESRD at 1 year. Furthermore, the cumulative incidence of death was higher than the cumulative incidence of post-LT ESRD in this subset of LT recipients.
Diabetes and older age, risk factors for ESRD in the general population, were also the significant predictors of renal nonrecovery. Among donor factors, male sex was associated with a higher risk of renal nonrecovery. We do not have a biologically plausible explanation for this new association. To further tease out the causality of this association, we evaluated the effect of donor and recipient sex mismatch in the renal nonrecovery model and it was not significant.
The post-LT mortality was high in our cohort. Significant predictors of post-LT mortality included previously reported factors among the overall recipient population, and donor factors (older donor, prolonged cold ischemia time, and cause of donor death other than head trauma, stroke, or anoxia) (26–29). These three donor factors are components of the donor risk index (29).
The limitations of our study are the retrospective observational design, which may result in the potential for bias due to patient selection and unmeasured patient characteristics, underestimation of pre-LT RRT duration among those who were on RRT at listing, and the lack of peri-transplant data including the specific date of discontinuation of acute RRT that could contribute inaccurate estimation of the risk of renal nonrecovery. Despite these limitations, our study identified the incidence and predictors of renal nonrecovery after LT among those who were on acute RRT before LT. These results have implications for patient counseling, management, listing behavior, and post-LT care.
In conclusion, under current waiting list and transplant practices in the United States, most candidates who are on acute RRT for <90 days before LT and survive after LT alone would recover renal function, are unlikely to progress to ESRD within 6 months after LT, and should not be routinely listed for SLKT. The renal nonrecovery risk is higher among older recipients, and those with a longer duration of pre-LT RRT, diabetes, and prior LT.
Disclosures
None.
Acknowledgments
The authors thank Ms. Shauna Leighton, Medical Editor, Arbor Research Collaborative for Health, Ann Arbor, Michigan, for providing editorial assistance.
P.S. is supported by National Institutes of Health (NIH) Grant KO8 DK-088946 and a research award from the American College of Gastroenterology. The statistical methodology development and analysis for this investigation was supported in part by NIH Grant 5R01 DK-70869 to D.E.S.
These data were presented orally at the American Transplant Congress, held June 2–6, 2012, in Boston, Massachusetts.
Footnotes
Published online ahead of print. Publication date available at www.cjasn.org.
References
- 1.Gonwa TA, McBride MA, Anderson K, Mai ML, Wadei H, Ahsan N: Continued influence of preoperative renal function on outcome of orthotopic liver transplant (OLTX) in the US: Where will MELD lead us? Am J Transplant 6: 2651–2659, 2006 [DOI] [PubMed] [Google Scholar]
- 2.Eason JD, Gonwa TA, Davis CL, Sung RS, Gerber D, Bloom RD: Proceedings of Concensus Conference on Simultaneous Liver Kidney Transplantation (SLK). Am J Transplant 8: 2243–2251, 2000 [DOI] [PubMed] [Google Scholar]
- 3.Brensing KA, Textor J, Perz J, Schiedermaier P, Raab P, Strunk H, Klehr HU, Kramer HJ, Spengler U, Schild H, Sauerbruch T: Long term outcome after transjugular intrahepatic portosystemic stent-shunt in non-transplant cirrhotics with hepatorenal syndrome: A phase II study. Gut 47: 288–295, 2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Martín-Llahí M, Pépin MN, Guevara M, Díaz F, Torre A, Monescillo A, Soriano G, Terra C, Fábrega E, Arroyo V, Rodés J, Ginès P, TAHRS Investigators : Terlipressin and albumin vs albumin in patients with cirrhosis and hepatorenal syndrome: A randomized study. Gastroenterology 134: 1352–1359, 2008 [DOI] [PubMed] [Google Scholar]
- 5.Sanyal AJ, Boyer T, Garcia-Tsao G, Regenstein F, Rossaro L, Appenrodt B, Blei A, Gülberg V, Sigal S, Teuber P, Terlipressin Study Group : A randomized, prospective, double-blind, placebo-controlled trial of terlipressin for type 1 hepatorenal syndrome. Gastroenterology 134: 1360–1368, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Angeli P, Volpin R, Gerunda G, Craighero R, Roner P, Merenda R, Amodio P, Sticca A, Caregaro L, Maffei-Faccioli A, Gatta A: Reversal of type 1 hepatorenal syndrome with the administration of midodrine and octreotide. Hepatology 29: 1690–1697, 1999 [DOI] [PubMed] [Google Scholar]
- 7.Runyon BA, AASLD Practice Guidelines Committee : Management of adult patients with ascites due to cirrhosis: An update. Hepatology 49: 2087–2107, 2009 [DOI] [PubMed] [Google Scholar]
- 8.Moreau R, Lebrec D: Acute renal failure in patients with cirrhosis: Perspectives in the age of MELD. Hepatology 37: 233–243, 2003 [DOI] [PubMed] [Google Scholar]
- 9.Northup PG, Argo CK, Bakhru MR, Schmitt TM, Berg CL, Rosner MH: Pretransplant predictors of recovery of renal function after liver transplantation. Liver Transpl 16: 440–446, 2010 [DOI] [PubMed] [Google Scholar]
- 10.Levine GN, McCullough KP, Rodgers AM, Dickinson DM, Ashby VB, Schaubel DE: Analytical methods and database design: Implications for transplant researchers, 2005. Am J Transplant 6: 1228–1242, 2006 [DOI] [PubMed] [Google Scholar]
- 11.United Network for Organ Sharing: 3.6 Allocation of Livers, 2005. Available at: http://optn.transplant.hrsa.gov/PoliciesandBylaws2/policies/pdfs/policy_8.pdf Accessed on November 30, 2012
- 12.Kalbfleisch JD, Prentice RL: The Statistical Analysis of Failure Time Data, 2nd Ed., New York, Wiley, 2002 [Google Scholar]
- 13.Bahirwani R, Campbell MS, Siropaides T, Markmann J, Olthoff K, Shaked A, Bloom RD, Reddy KR: Transplantation: Impact of pretransplant renal insufficiency. Liver Transpl 14: 665–671, 2008 [DOI] [PubMed] [Google Scholar]
- 14.Gonwa TA, Klintmalm GB, Levy M, Jennings LS, Goldstein RM, Husberg BS: Impact of pretransplant renal function on survival after liver transplantation. Transplantation 59: 361–365, 1995 [PubMed] [Google Scholar]
- 15.Nair S, Verma S, Thuluvath PJ: Pretransplant renal function predicts survival in patients undergoing orthotopic liver transplantation. Hepatology 35: 1179–1185, 2002 [DOI] [PubMed] [Google Scholar]
- 16.Ojo AO, Held PJ, Port FK, Wolfe RA, Leichtman AB, Young EW, Arndorfer J, Christensen L, Merion RM: Chronic renal failure after transplantation of a nonrenal organ. N Engl J Med 349: 931–940, 2003 [DOI] [PubMed] [Google Scholar]
- 17.Campbell MS, Kotlyar DS, Brensinger CM, Lewis JD, Shetty K, Bloom RD, Markmann JF, Olthoff KM, Shaked A, Reddy KR: Renal function after orthotopic liver transplantation is predicted by duration of pretransplantation creatinine elevation. Liver Transpl 11: 1048–1055, 2005 [DOI] [PubMed] [Google Scholar]
- 18.Sharma P, Welch K, Eikstadt R, Marrero JA, Fontana RJ, Lok AS: Renal outcomes after liver transplantation in the model for end-stage liver disease era. Liver Transpl 15: 1142–1148, 2009 [DOI] [PubMed] [Google Scholar]
- 19.Nadim MK, Davis CL, Sung R, Kellum JA, Genyk YS: Simultaneous liver-kidney transplantation: A survey of US transplant centers. Am J Transplant 12: 3119–3127, 2012 [DOI] [PubMed] [Google Scholar]
- 20.Nadim MK, Sung RS, Davis CL, Andreoni KA, Biggins SW, Danovitch GM, Feng S, Friedewald JJ, Hong JC, Kellum JA, Kim WR, Lake JR, Melton LB, Pomfret EA, Saab S, Genyk YS: Simultaneous liver-kidney transplantation summit: Current state and future directions. Am J Transplant 12: 2901–2908, 2012 [DOI] [PubMed] [Google Scholar]
- 21.Davis CL, Feng S, Sung R, Wong F, Goodrich NP, Melton LB, Reddy KR, Guidinger MK, Wilkinson A, Lake J: Simultaneous liver-kidney transplantation: Evaluation to decision making. Am J Transplant 7: 1702–1709, 2007 [DOI] [PubMed] [Google Scholar]
- 22.Davis CL: Impact of implementation of the MELD scoring system on the prevalence and incidence of chronic renal disease following liver transplantation. Liver Transpl 12: 707–709, 2006 [DOI] [PubMed] [Google Scholar]
- 23.Ruiz R, Kunitake H, Wilkinson AH, Danovitch GM, Farmer DG, Ghobrial RM, Yersiz H, Hiatt JR, Busuttil RW: Long-term analysis of combined liver and kidney transplantation at a single center. Arch Surg 141: 735–741, discussion 741–742, 2006 [DOI] [PubMed] [Google Scholar]
- 24.United Network for Organ Sharing: 3.6 Organ Distribution: Allocation of Livers, 2002. Available at: http://optn.transplant.hrsa.gov/PoliciesandBylaws2/policies/pdfs/policy_8.pdf Accessed on November 30, 2012
- 25.Levitsky J, Baker T, Ahya SN, Levin ML, Friedewald J, Gallon L, Ho B, Skaro A, Krupp J, Wang E, Spies SM, Salomon DR, Abecassis MM: Outcomes and native renal recovery following simultaneous liver-kidney transplantation. Am J Transplant 12: 2949–2957, 2012 [DOI] [PubMed] [Google Scholar]
- 26.Onaca NN, Levy MF, Netto GJ, Thomas MJ, Sanchez EQ, Chinnakotla S, Fasola CG, Weinstein JS, Murray N, Goldstein RM, Klintmalm GB: Pretransplant MELD score as a predictor of outcome after liver transplantation for chronic hepatitis C. Am J Transplant 3: 626–630, 2003 [DOI] [PubMed] [Google Scholar]
- 27.Saab S, Wang V, Ibrahim AB, Durazo F, Han S, Farmer DG, Yersiz H, Morrisey M, Goldstein LI, Ghobrial RM, Busuttil RW: MELD score predicts 1-year patient survival post-orthotopic liver transplantation. Liver Transpl 9: 473–476, 2003 [DOI] [PubMed] [Google Scholar]
- 28.Narayanan Menon KV, Nyberg SL, Harmsen WS, DeSouza NF, Rosen CB, Krom RA, Wiesner RH: MELD and other factors associated with survival after liver transplantation. Am J Transplant 4: 819–825, 2004 [DOI] [PubMed] [Google Scholar]
- 29.Feng S, Goodrich NP, Bragg-Gresham JL, Dykstra DM, Punch JD, DebRoy MA, Greenstein SM, Merion RM: Characteristics associated with liver graft failure: The concept of a donor risk index. Am J Transplant 6: 783–790, 2006 [DOI] [PubMed] [Google Scholar]