Abstract
In June 2008, the American Association for the Study of Liver Diseases (AASLD) sponsored the Henry M. and Lillian Stratton Basic Research Single-Topic Conference on the Pathobiology of Biliary Epithelia and Cholangiocarcinoma, which was held in Atlanta, GA. Attendees from 12 different countries participated in this conference, making it a truly international scientific event. Both oral and poster presentations were given by multidisciplinary experts, who highlighted important areas of current basic and translational research on biliary epithelial cell biology and pathophysiology, and on the etiology, cellular and molecular pathogenesis, and target-based therapy of cholangiocarcinoma. The specific goals and objectives of the conference were: (1) to advance knowledge of basic and molecular mechanisms underlying developmental and proliferative disorders of the biliary tract; (2) to foster a better and more comprehensive understanding of mechanisms regulating biliary epithelial (cholangiocyte) growth and transport, signaling, cell survival, and abnormalities that result in disease; and (3) to understand basic mechanisms of cholangiocarcinoma development and progression, with the added goal of identifying and exploiting potentially critical molecular pathways that may be targeted therapeutically. A number of interrelated themes emerged from the oral and poster sessions that affected current understandings of the complex organization of transcriptional and signaling mechanisms that regulate bile duct development, hepatic progenitor cell expansion, cholangiocyte secretory functions and proliferation, and mechanisms of cholangiocarcinogenesis and malignant cholangiocyte progression. Most notable were the critical questions raised as to how best to exploit aberrant signaling pathways associated with biliary disease as potential targets for therapy.
The American Association for the Study of Liver Diseases (AASLD) sponsored its first Single-Topic Conference on the Pathobiology of Epithelia in 1994, followed by a second meeting in 2001. Since that time, there has been a rapid expansion of new research aimed at elucidating key signaling pathways that regulate cholangiocyte development, differentiation, proliferation, survival, and function. Moreover, this same period has seen rapid increases in both basic and translational research on cellular and molecular mechanisms of cholangiocarcinoma development and progression, as well as on defining potential molecular targets for cholangiocarcinoma therapy and/or chemoprevention. The topics covered in the third offering of this Single-Topic Conference held in Atlanta, GA, in June 2008 were selected to (1) present new research findings and denote salient advances made particularly in the areas of cholangiocyte signaling in normal and diseased states, and (2) to critically address the increasing need to further our understanding of mechanisms underlying cholangiocarcinoma pathogenesis and progression for the purpose of devising novel therapeutic strategies aimed at improving outcome for patients with this devastating cancer. In addition to providing a timely and comprehensive account of relevant research findings, the objective of this conference was also to frame critical questions that now need to be answered by additional studies in order to gain a better understanding on how signaling mechanisms and pathways are integrated in normal and malignant cholangiocytes, and how aberrant signaling pathways in biliary disease states may serve as targets for therapy.
Biliary Tree Development and Hepatic Progenitor Cells
The opening session of the conference elucidated the role of the Notch signaling pathway on intrahepatic bile duct development and proliferation, profiling various Notch pathway mutant mouse models that recapitulate clinically relevant features of Alagille syndrome.1–3 These models have helped to clarify the roles of different members of the Notch signaling cascade in bile duct morphogenesis and in postnatal bile duct growth and remodeling. As a result of these studies, certain key components of the Notch signaling pathway, including Jagged 1 (Jag1) and Notch2, as well as hairy and enhancer of split 1 (Hes1), a downstream transcription factor in the pathway, are now appreciated as regulators of intrahepatic bile duct development and biliary tract morphogenesis. Hepatocyte nuclear factor-6 (HNF-6), also known as OC1, a member of the onecut family of transcription factors, was also discussed as being crucial to the normal development of the biliary tract,4–6 as well as functioning to differentially regulate the expression of select hepatocyte and cholangiocyte genes governing mature liver differentiation and proliferative responses.5,7 Jagged-mediated Notch signaling and onecut family genes were also described in this session as being regulators of zebrafish biliary development, reflecting an evolutionarily conserved role for these pathways in vertebrate bile duct development.8,9 In addition, the session highlighted that the concept of hepatic progenitor cells and conditions governing their expansion and activation are becoming more well defined. Notably, the extent of progenitor cell activation and proliferation into the liver parenchyma was determined to directly correlate with disease severity in both acute and chronic human liver diseases in which the normal hepatocyte proliferative response becomes impaired.10 Factors that govern hepatic progenitor cell activation and differentiation are beginning to be identified, with the Winglesstype MMTV integration site family (wnt)/ß-catenin and Notch pathways having been implicated in both human and rodent facultative hepatic progenitor cell activation.11–14 In addition, hepatocyte-associated transcription factors, such as HNF-6, have been recently detected in hepatic biliary cells prior to the emergence of progenitor cells for hepatocytes when the normal regenerative capacity of liver becomes compromised.15 However, control mechanisms for hepatic progenitor cell differentiation are only partially understood, and a number of key questions in the areas of biliary development and hepatic progenitor cells remain. Specifically, in the case of intrahepatic bile duct development, what are the precise inductive factors and extracellular matrix components of the periportal environment that induce transcriptional pathways leading to cholangiocyte differentiation and bile duct morphogenesis in the developing liver; what genes and signaling mechanisms control branching morphogenesis in the biliary tract; based on the complexity of interactions between hepatic transcription factors in bile duct development and homeostatic function, what types of models need to be developed to further define downstream effectors by which maturation events take place in the biliary tract, especially for new proteins and markers that are only now being identified; and lastly, in addition to bile duct development, what is the role of Notch and/or onecut family transcriptional signaling in the pathogenesis of various acquired human hepatobiliary diseases. In the case of hepatic progenitor cells, the major question that needs to be addressed is how to fully drive their differentiation into functionally mature hepatocytes and/or cholangiocytes in a way that will permit a therapeutic advantage, both in development and in adulthood. The extent to which hepatic progenitor cells may contribute to primary liver cancer development in humans also needs to be clarified. Presently, there is not enough data to make definitive statements concerning the origin of human hepatocellular carcinoma or of cholangiocarcinoma from hepatic progenitor cells, although the presence of hepatic progenitor cell markers and the presence and extent of the cholangiocellular component are factors that have been reported as being related to the prognosis of hepatocellular carcinomas, as well as of combined hepatocellular-cholangiocarcinomas.16 Another key question raised in this session is to determine the relative role played by hepatocyte-to-bile duct transdifferentiation17–19 and epithelial-mesenchymal transition,20 especially in chronic liver diseases with hepatic injury and cirrhosis. No doubt, screening methods, such as the zebrafish model, select transgenic and gene knockout mouse models, isolation of purified hepatic progenitor cell subpopulations,21,22 transcriptional profiling,23 and antigenic cell marker phenotyping24 will contribute in significant ways toward defining the answers to these questions.
Cholangiocyte Signaling
Two broad themes emerged from the session on cholangiocyte signaling. First, there is now a much better understanding of the complex organization of signaling events that occur within cholangiocytes. Specifically, we now understand not just the importance of signaling proteins such as inositol triphosphate receptors (InsP3Rs), P2Y nucleotide receptors (P2YRs), and adenylate cyclases (ACs), but we also understand that there are multiple isoforms of each and that each isoform has a specific function as well.25–28 Moreover, the subcellular location of each isoform plays an important role in specifying its signaling function. Specific examples include the proximity of specific InsP3R, P2YR, and AC isoforms to the apical membrane and to primary cilia.29,30
Second, there is now a more complex understanding of signaling from the external environment to cholangiocytes. This includes an improved understanding of the relevance of neuropeptides,31,32 autocrine/paracrine stimuli such as ATP, and the immune system.33–35 Some important mechanistic questions remain, such as how ATP is released from cholangiocytes. However, the big questions that address integration of signaling mechanisms and pathways are likely to take center stage next. One example might be to determine how the apparent redundancy of ACs and P2YRs fits together. The answer to this would provide the mechanism for cross-talk between key signaling pathways, but we are likely to find that there will be specific spatial and temporal constraints that regulate this cross-talk. Another example would be to determine how the signals from the many humoral, neural, endocrine, and autocrine stimuli are integrated. The answer to this may well relate to another question, which concerns how gradients in phenotype along the biliary tree are arranged to assure organ-level ductal function. The final big question here is whether there are key common pathways in cholangiocyte signaling that are causes of disease or can be used as targets for therapy.
Hepatic Cystogenesis
The intrahepatic bile ducts are a major target of a diverse group of genetically heterogeneous multiorgan disorders, the polycystic liver diseases (PCLDs). PCLDs may occur alone or in combination with polycystic kidney disease (PKD).36,37 However, autosomal dominant polycystic liver disease (ADPLD) displays no renal involvement, and in both autosomal dominant PKD (ADPKD) and autosomal recessive PKD (ARPKD), development of hepatic cysts is the most frequent extrarenal manifestation. Although the genetic defects involved in initiation of cyst formation in PCLDs have been identified, the mechanisms underlying hepatic cystogenesis are still unclear. In this context, the conference provided new insight into molecular regulators of hepatic cystogenesis. Importantly, all PCLDs are characterized by an accelerated cholangiocyte proliferation that is accompanied by alterations in the cell cycle.37 Two important and separate areas of research, notably involving cell division cycle 25 homolog A (Cdc25A) and angiogenic factors, respectively, were described as playing important roles in promoting the growth of hepatic cysts in PCLD (Fig. 1). Cdc25A, a dual-specificity phosphatase cell cycle regulator,38 is now known to be overexpressed in cystic cholangiocytes of human patients with ADPKD and ARPKD, as well as in intrahepatic cholangiocytes of the PCK (polycystic kidney) rat, an animal model of ARPKD. Notably, Cdc25A up-regulation is associated with down-regulation of microRNA-15a (miR-15a), a small noncoding RNA that post-transcriptionally inhibits Cdc25A messenger RNA via sequence-specific base-pairing. Of particular interest is the novel finding that experimental overexpression of miR-15a in cultured PCK-derived cholangiocytes decreased their Cdc25A levels, inhibited cell proliferation and reduced cyst growth, thereby functionally implicating Cdc25A up-regulation with intrahepatic cholangiocyte proliferation, and suggesting that this regulatory pathway is involved in hepatic cystogenesis. These results also suggest that Cdc25A overexpression is due to posttranscriptional inhibition of miR-15a, raising the possibility of modulating microRNA expression as a potential novel therapeutic approach to benign hyperproliferative hepatobiliary diseases.
Fig. 1.
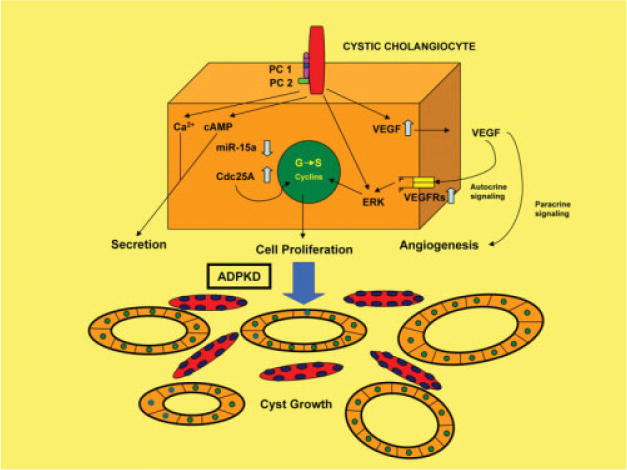
Scheme depicting signaling pathways linked to primary cilia in cholangiocytes, and alterations that occur in polycystic liver disease. It is proposed that abnormalities in the miR-15a/Cdc25A complex, VEGF signaling, cytosolic Ca2+, and cyclic adenosine monophosphate (cAMP) signaling play essential roles in the regulation of cholangiocyte secretion, proliferation, and hepatic cystogenesis. In ADPKD, miR-15a is down-regulated and its target Cdc25A is up-regulated in cystic cholangiocytes, contributing to cystic growth by accelerating entry of the biliary cyst cells into S phase, whereas VEGF appears to contribute to cystic growth via a mechanism involving autocrine stimulation of cystic cholangiocyte proliferation and paracrine stimulation of vascular supply to the cysts. Stimulation of cilia via luminal flow or changes in luminal osmolarity modulates ductular secretion via the second messengers Ca2+ and cAMP. See Masyuk et al.30, Masyuk and LaRusso37, and Fabris et al.39 for additional details.
Marked overexpression of vascular endothelial growth factor (VEGF) and VEGF receptors tyrosine kinase with immunoglobulin-like and EGF-like domains 2 (Tie-2) and angiopoietin-1 (Ang-1) has also now been implicated in cholangiocyte proliferation and progressive hepatic cyst enlargement in ADPKD, and is correlated with the aberrant vascularization present in PCLDs.39 These interesting new findings further raise the possibility that clinically available pharmacological inhibitors used to block VEGF signaling may have significant therapeutic implications with respect to halting the progression of AD-PKD.
Cholangiocarcinoma
The conference underscored the rising clinical importance of cholangiocarcinoma, with a focus on epidemiology and risk factors, cellular and molecular mechanisms, novel preclinical models, and opportunities for targeted therapies. The rising worldwide incidence and mortality rates for cholangiocarcinoma over the last quarter century now seem to be real.40,41 However, misclassification or reclassification of intrahepatic and extrahepatic cholangiocarcinomas, especially of Klatskin tumors,42 has complicated efforts to more accurately gauge the trends in intrahepatic and extrahepatic cholangiocarcinoma incidence. Moreover, despite the fact that a number of wellestablished risk factors are now known for intrahepatic cholangiocarcinoma, it is also clear that most cases of intracellular carcinoma have no specific risk factors, and the causes of the increased incidences of cholangiocarcinoma in the United States and in other countries remains poorly understood. Future epidemiological studies that incorporate microdissection and gene expression profiling into their experimental design could help to clarify the confusion brought about by misclassification, and potentially establish molecular trends underlying pattern changes in cholangiocarcinoma incidence rates.
Significant progress has been made over the past several years in defining critical epigenetic pathways regulating cholangiocarcinoma cell growth and progression, as well as resistance to apoptosis. Aberrant expression and activation of growth factor receptor tyrosine kinases, such as epidermal growth factor receptor B (ErbB) family receptor tyrosine kinases, hepatocyte growth factor (HGF)-Met, and interleukin-6 (IL-6) signaling,43 have each been linked to autonomous proliferation, apoptosis resistance, and progression of cholangiocarcinoma cells (Fig. 2). The link between cyclooxygenase-2 (COX-2) and cholangiocarcinogenesis is also now well established, with the interplay between COX-2-derived prostaglandin signaling and other growth factor regulatory pathways, including those of ErbB1, ErbB2, and Met, have potentially important implications for cholangiocarcinoma therapy and/or chemoprevention.44 Transmembrane mucins (MUCs), including MUC1 and MUC4, have further been reported to be prognostic factors for intrahepatic cholangiocarcinoma,45,46 and to interact with select ErbB family receptors,47 leading to altered tumor cell growth, cell signaling, and progression, as well as having potential diagnostic and therapeutic value. Enzymatically active aspartyl (asparaginyl) ß-hydroxylase, whose expression is regulated through insulin-like growth factor-1 receptor signaling, and which also interacts with Notch, may also prove to be useful as an early diagnostic marker of cholangiocarcinoma and as a monitor of disease progression,48 as well as a therapeutic target. Disruption of epigenetic pathways regulating serine/threonine Akt/PK B (Akt) and FADD-like interleukin-1 β converting enzyme (Flice)–like inhibitory protein activation, and targeted inhibition of myeloid cell leukemia 1 (Mcl1), a potent member of the B-cell lymphoma-2 (Bcl-2) antiapoptotic family, which is overexpressed in cholangiocarcinoma cells, also merit further study as potential molecular therapeutic strategies for cholangiocarcinoma.49,50 Of further interest, aberrantly expressed microRNAs are providing mechanistic insight into hepatic cystogenesis and tumorigenesis, cholangiocarcinoma cell proliferation and chemoresistance,51,52 and may serve as potential targets for cholangiocarcinoma therapy. Similarly, understanding the role played by cholangiocyte signaling pathways activated by angiogenic factors is likely to provide new insight into the treatment of proliferative cholangiopathies, including cholangiocarcinoma and polycystic liver diseases.39,53
Fig. 2.

Molecular events in cholangiocarcinogenesis. Chronic biliary inflammation and cholestasis lead to cellular changes leading to cholangiocarcinoma development and progression. Inflammatory cytokines induce iNOS (inducible nitric oxide synthase) in cholangiocytes, favoring mutagenesis, impaired DNA repair, and COX-2 up-regulation. Aberrant bile acid signaling contributes to the promotion of cholangiocyte growth via transactivation of the epidermal growth factor receptor (EGFR), and is also linked to activation of the COX-2 pathway. Alterations in various genetic and epigenetic molecular pathways selectively depicted in the figure then combine to promote the features of immortalization, evasion of apoptosis, autonomous proliferation, and invasion and metastasis characteristic of malignant cholangiocytes. MMP, matrix metalloproteinase; PDGF, platelet-derived growth factor; TGF, transforming growth factor; TNF, tumor necrosis factor.
With our increasing understanding of the molecular pathways involved in cholangiocarcinogenesis and cholangiocarcinoma progression, we are seeing increased opportunities for targeted therapies that can have a future impact on improving the outcome of patients with cancers of the biliary tract.54 Currently, there is an increasing number of targeted therapies (i.e., receptor tyrosine kinase inhibitors, multiple kinase inhibitors, antiangiogenic agents, and Akt and mammalian target of rapamycin [mTOR] inhibitors) either approved or in different phases of clinical trial for several types of solid malignancies. The fact that molecular targets such as epidermal growth factor, ErbB2, Met, Raf1, Mcl1, COX-2, Akt, and VEGF have been identified in cholaniocarcinomas, and that global gene expression profiling of biliary tract cancers is helping to identify other novel molecular targets clearly justifies efforts to continue to develop new targetbased strategies for cholangiocarcinoma therapy and/or chemoprevention. However, as was emphasized at the conference, single-agent treatments with targeted agents have shown only modest activity against various solid tumors, including cholangiocarcinoma. Even when an improved patient outcome was achieved, there has been minimal effort to correlate use or efficacy of a targetdirected agent with molecular biomarkers that could be used to predict or monitor a targeted therapeutic response. The conference also served to underscore a variety of challenges and limitations that need to be overcome to insure more effective clinical trials of target-based therapies for cholangiocarcinoma. Obviously, a major challenge with securing support for target-based clinical trials against a relatively rare cancer like cholangiocarcinoma is to be able to achieve adequate patient selection and numbers that will require cooperation from multiple treatment centers located within the United States and in other countries, particularly those where this malignant disease is more prevalent. It is also becoming evident that targeting the tumor microenvironment may be just as critical as targeting the cancer cells themselves as a means for controlling tumorigenic growth and progression. In addition, factors such as intratumoral heterogeneity, varied mechanisms of acquired resistance, constitutive overexpression of autocrine and/or paracrine growth factor ligands, coactivation of multiple receptor tyrosine kinases resulting in signaling redundancy, cross-talk and interplay, tumor hypovascularization limiting bioavailability, and impaired liver function can all act to circumvent the efficacy of target-based therapies. In this context, it is obvious that our approaches to developing novel target-based strategies for cholangiocarcinoma therapy need to focus more on combinational rather than single agent-based treatments. We also need to develop biomarkers that could more reliably and objectively predict a therapeutic response from such treatments. Finally, there is an important need to further develop and validate novel preclinical animal models, such as the rat BDEneu orthotopic model of cholangiocarcinoma progression55 and the Smad4Co/CoPtenCo/CoAlb-Cre mouse model of intrahepatic cholangiocarcinoma56 for their ability to rapidly screen for and more accurately predict therapeutic responses in human-based clinical trials of target-based therapies.
Acknowledgments
We wish to express our deep appreciation and gratitude to Ms. Pamela S. Bogart of the Mayo College of Medicine, Ms. Jennifer L. DeWitt of Virginia Commonwealth University School of Medicine, and Ms. Dianne M. Freeman and Ms. Melissa A. Parrish of the AASLD for their valuable assistance in helping to coordinate the organization and logistics of this Single-Topic Conference.
Abbreviations
- ADPLD
autosomal dominant polycystic liver disease
- ACs
adenylate cyclases
- COX-2
cyclooxygenase-2
- HGF
hepatocyte growth factor
- IL-6
interleukin-6
- InsP3Rs
inositol triphosphate receptors
- MUCs
mucins
- P2YRs
P2Y nucleotide receptors
- PCLDs
polycystic liver diseases
- PKD
polycystic kidney disease
- VEGF
vascular endothelial growth factor
Footnotes
Potential conflict of interest: Nothing to report.
References
- 1.Kodama Y, HijkataM, Kageyama R, Shimotohno K, Chiba T. The role of Notch signaling in the development of intrahepatic bile ducts. Gastroenterology. 2004;127:1775–1786. doi: 10.1053/j.gastro.2004.09.004. [DOI] [PubMed] [Google Scholar]
- 2.Loomes KM, Russo P, Ryan M, Nelson A, Underkoffler L, Glover C, et al. Bile duct proliferation in liver-specific Jag1 conditional knockout mice: effects of gene dosage. Hepatology. 2007;45:323–330. doi: 10.1002/hep.21460. [DOI] [PubMed] [Google Scholar]
- 3.Lozier J, McCright B, Gridley T. Notch signaling regulates bile duct morphogenesis in mice. PLoS ONE. 2008;3:e1851. doi: 10.1371/journal.pone.0001851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Clotman F, Lannoy VJ, ReberM, Cereghini S, Cassiman D, Jacquemin P, et al. The onecut transcription factor HNF6 is required for normal development of the biliary tract. Development. 2002;129:1819–1828. doi: 10.1242/dev.129.8.1819. [DOI] [PubMed] [Google Scholar]
- 5.Costa RH, Kalinichenko VV, Holterman A-XL, Wang X. Transcription factors in liver development, differentiation, and regeneration. Hepatology. 2003;38:1331–1347. doi: 10.1016/j.hep.2003.09.034. [DOI] [PubMed] [Google Scholar]
- 6.Yamasaki H, Sada A, Iwata T, Niwa T, Tomizawa M, Xanthopoulos KG, et al. Suppression of C/EBPα expression in periportal hepatoblasts may stimulate biliary cell differentiation through increased Hnf6 and Hnf1b expression. Development. 2006;133:4233–4243. doi: 10.1242/dev.02591. [DOI] [PubMed] [Google Scholar]
- 7.Wang M, Tan Y, Costa RH, Holterman A-XL. In vivo regulation of murine CYP7A1 by HNF-6: a novel mechanism for diminished CYP7A1 expression in biliary obstruction. HEPATOLOGY. 2004;40:600–608. doi: 10.1002/hep.20349. [DOI] [PubMed] [Google Scholar]
- 8.Lorent K, Yeo SY, Oda T, Chandrasekharappa S, Chitnis A, Matthews RP, et al. Inhibition of Jagged-mediated Notch signaling disrupts zebrafish biliary development and generates multi-organ defects compatible with an Alagille syndrome phenocopy. Development. 2004;131:5753–5766. doi: 10.1242/dev.01411. [DOI] [PubMed] [Google Scholar]
- 9.Matthews RP, Lorent K, Russo P, Pack M. The zebrafish onecut gene hnf-6 functions in an evolutionary conserved genetic pathway that regulates vertebrate biliary development. Dev Biol. 2004;274:245–259. doi: 10.1016/j.ydbio.2004.06.016. [DOI] [PubMed] [Google Scholar]
- 10.Katoonizadeh A, Nevens F, Verslype C, Pirenne J, Roskams T. Liver regeneration in acute severe liver impairment: aclinicopathological correlation study. Liver Int. 2006;26:1225–1233. doi: 10.1111/j.1478-3231.2006.01377.x. [DOI] [PubMed] [Google Scholar]
- 11.Hu M, Kurobe M, Jeong YJ, Fuerer C, Ghole S, Nusse R, et al. Wnt/ß-catenin signaling in murine hepatic transit amplifying progenitor cells. Gastroenterology. 2007;133:1579–1591. doi: 10.1053/j.gastro.2007.08.036. [DOI] [PubMed] [Google Scholar]
- 12.Yang W, Yan HX, Chen L, Liu Q, He YQ, Yu LX, et al. Wnt/β-catenin signaling contributes to activation of normal and tumorigenic liver progenitor cells. Cancer Res. 2008;68:4287–4295. doi: 10.1158/0008-5472.CAN-07-6691. [DOI] [PubMed] [Google Scholar]
- 13.Crosby HA, NijjarSS, deGoyetJV, Kelly DA, StrainAJ Progenitor cells of the biliary epithelial cell lineage. Semin Cell Dev Biol. 2002;13:397–403. doi: 10.1016/s108495210200126x. [DOI] [PubMed] [Google Scholar]
- 14.Jensen CH, Jauho EI, Santoni-Rugiu E, Holmskov U, Teisner B, Tygstrup N, et al. Transit-amplifying ductular (oval) cells and their hepatocytic progeny are characterized by a novel and distinctive expression of delta-like protein/preadipocyte factor1/fetal antigen 1. Am J Pathol. 2004;164:1347–1359. doi: 10.1016/S0002-9440(10)63221-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Limaye PB, Alarcón G, Walls AL, Nalesnik MA, Michalopoulos GK, Demetris AJ, et al. Expression of specific hepatocyte and cholangiocyte transcription factors in human liver disease and embryonic development. Lab Invest. 2008;88:865–872. doi: 10.1038/labinvest.2008.56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Libbrecht L. Hepatic progenitor cells in human liver tumor development. World J Gastroenterol. 2006;12:6261–6265. doi: 10.3748/wjg.v12.i39.6261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Nishikawa Y, Doi Y, Watanabe H, Tokairin T, Omori Y, Su M, et al. Transdifferentiation of mature rat hepatocytes into bile duct-like cells in vitro. Am J Pathol. 2005;166:1077–1088. doi: 10.1016/S0002-9440(10)62328-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Limaye PB, Bowen WC, Orr AV, Luo J, Tseng GC, Michalopoulos GK. Mechanisms of hepatocyte growth factor-mediated and epidermal growth factor-mediated signaling in transdifferentiation of rat hepatocytes to biliary epithelium. Hepatology. 2008;47:1702–1713. doi: 10.1002/hep.22221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Watanabe H, Hata M, Terada N, Ueda H, Yamada N, Yamanegi K, et al. Transdifferentiation into biliary ductular cells of hepatocytes transplanted into the spleen. Pathology. 2008;40:272–276. doi: 10.1080/00313020801911546. [DOI] [PubMed] [Google Scholar]
- 20.Rygiel KA, Robertson H, Marshall HL, Pekalski M, Zhao L, Booth TA, et al. Epithelial-mesenchymal transition contributes to portal tract fibrogenesis during human chronic liver disease. Lab Invest. 2008;88:112–123. doi: 10.1038/labinvest.3700704. [DOI] [PubMed] [Google Scholar]
- 21.Simper-Ronan R, Brilliant K, Flanagan D, Carreiro M, Callanan H, Sabo E, et al. Cholangiocyte marker-positive and -negative fetal liver cells differ significantly in their ability to regenerate the livers of adult rats exposed to retrorsine. Development. 2006;133:4269–4279. doi: 10.1242/dev.02589. [DOI] [PubMed] [Google Scholar]
- 22.Castillo GD, Álvarez-Barrientos A, Carmona-Cuenca I, Fernández M, Sánchez A, Fabregat I. Isolation and characterization of a putative liver progenitor population after treatment of fetal rat hepatocytes with TGF-beta. J Cell Physiol. 2008;215:846–855. doi: 10.1002/jcp.21370. [DOI] [PubMed] [Google Scholar]
- 23.Ochsner SA, Strick-Marchand H, Qui Q, Venable S, Dean A, Wilde M, et al. Transcriptional profiling of bipotential embryonic liver cells to identify liver progenitor cell surface markers. Stem Cells. 2007;25:2476–2487. doi: 10.1634/stemcells.2007-0101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hixson DC, Brown J, McBride AC, Affigne S. Differentiation status of rat ductal cells and ethionine-induced hepatic carcinomas defined with surface-reactive monoclonal antibodies. Exp Mol Pathol. 2000;68:152–169. doi: 10.1006/exmp.2000.2302. [DOI] [PubMed] [Google Scholar]
- 25.Salter KD, Fitz JG, Roman RM. Domain-specific purinergic signaling in polarized rat cholangiocytes. Am J Physiol Gastrointest Liver Physiol. 2000;278:G492–G500. doi: 10.1152/ajpgi.2000.278.3.G492. [DOI] [PubMed] [Google Scholar]
- 26.Shibao K, Hirata K, Robert ME, Nathanson MH. Loss of inositol 1, 4, 5-triphosphate receptors from bile duct epithelia is a common event in cholestasis. Gastroenterology. 2003;125:1175–1187. doi: 10.1016/s0016-5085(03)01201-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Minagawa N, Nagata J, Shibao K, Masyuk AI, Gomes DA, Rodrigues M, et al. Cyclic AMP regulates bicarbonate secretion in cholangiocytes through release of ATP into bile. Gastroenterology. 2007;133:1592–1602. doi: 10.1053/j.gastro.2007.08.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Fiorotto K, Spirlì C, Fabris L, Cadamuro M, Okolicsanyi L, Strazzabosco M. Ursodeoxycholic acid stimulates cholangiocytes fluid secretion in mice via CFTR-dependent ATP secretion. Gastroenterology. 2007;133:1603–1613. doi: 10.1053/j.gastro.2007.08.071. [DOI] [PubMed] [Google Scholar]
- 29.Gradilone SA, Masyuk AI, Splinter PL, Banales JM, Huang BQ, Tietz PS, et al. Cholangiocyte cilia express TRPV4 and detect changes in luminal tonicity including bicarbonate secretion. Proc Natl Acad Sci U S A. 2007;104:19138–19143. doi: 10.1073/pnas.0705964104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Masyuk AI, Masyuk TV, LaRusso NF. Cholangiocyte primary cilia in liver health and disease. Dev Dyn. 2008;237:2007–2012. doi: 10.1002/dvdy.21530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.AlvaroD, Mancino MG, Glaser S, Gaudio E, Marzioni M, Francis H, et al. Proliferating cholangiocytes: a neuroendocrine compartment in the diseased liver. Gastroenterology. 2007;132:415–431. doi: 10.1053/j.gastro.2006.07.023. [DOI] [PubMed] [Google Scholar]
- 32.Marzioni M, Fava G, Alvaro D, Alpini G, Benedetti A. Control of cholangiocyte adaptive responses by visceral hormones and neuropeptides. Clin Rev Allergy Immunol. 2008 doi: 10.1007/S12016-008-8090-1. PMID. 18548352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mack CL, Tucker RM, Lu BR, Sokol RJ, Fontenot AP, Ueno Y, et al. Cellular and humoral autoimmunity directed at bile duct epithelia in murine biliary atresia. Hepatology. 2006;44:1231–1239. doi: 10.1002/hep.21366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Harada K, Nakanuma Y. Biliary innate immunity and cholangiopathy. Hepatol Res. 2007;37:S430–S437. doi: 10.1111/j.1872-034X.2007.00247.x. [DOI] [PubMed] [Google Scholar]
- 35.Chen XM, O’Hara SP, LaRusso NF. The immunobiology of cholangiocytes. Immunol Cell Biol. 2008;86:497–505. doi: 10.1038/icb.2008.37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Everson GT, Taylor MR, Doctor RB. Polycystic disease of the liver. Hepatology. 2004;40:774–782. doi: 10.1002/hep.20431. [DOI] [PubMed] [Google Scholar]
- 37.Masyuk T, LaRusso N. Polycystic liver disease: new insights into disease pathogenesis. Hepatology. 2006;43:906–908. doi: 10.1002/hep.21199. [DOI] [PubMed] [Google Scholar]
- 38.Karlsson-Rosenthal C, Millar JD. Cdc25: mechanisms of checkpoint inhibition and recovery. Trends Cell Biol. 2006;16:285–292. doi: 10.1016/j.tcb.2006.04.002. [DOI] [PubMed] [Google Scholar]
- 39.Fabris L, Cadamuro M, Fiorolto R, Roskams T, Spirlì C, Melero S, et al. Effects of angiogenic factor overexpression by human and rodent cholangiocytes in polycystic liver diseases. Hepatology. 2006;43:1001–1012. doi: 10.1002/hep.21143. [DOI] [PubMed] [Google Scholar]
- 40.Shaib Y, El-Serag HB. The epidemiology of cholangiocarcinoma. Semin Liver Dis. 2004;24:115–125. doi: 10.1055/s-2004-828889. [DOI] [PubMed] [Google Scholar]
- 41.McGlynn KA, Tarone RE, El-Serag HB. A comparison of trends in the incidence of hepatocellular carcinoma and intrahepatic cholangiocarcinoma in the United States. Cancer Epidemiol Biomarkers Prev. 2006;15:1198–1203. doi: 10.1158/1055-9965.EPI-05-0811. [DOI] [PubMed] [Google Scholar]
- 42.Ben-Menachem T. Risk factors for cholangiocarcinoma. Eur J Gastroenterol Hepatol. 2007;19:615–617. doi: 10.1097/MEG.0b013e328224b935. [DOI] [PubMed] [Google Scholar]
- 43.Berthiaume EP, Wands J. The molecular pathogenesis of cholangiocarcinoma. Semin Liver Dis. 2004;24:127–137. doi: 10.1055/s-2004-828890. [DOI] [PubMed] [Google Scholar]
- 44.Wu T. Cyclooxygenase-2 and prostaglandin signaling in cholangiocarcinoma. Biochim Biophys Acta. 2005;1755:135–150. doi: 10.1016/j.bbcan.2005.04.002. [DOI] [PubMed] [Google Scholar]
- 45.Matsumura N, Yamamoto M, Aruga A, Takasaki K, Nakano M. Correlation between expression of MUC1 core protein and outcome after surgery in mass-forming intrahepatic cholangiocarcinoma. Cancer. 2002;94:1770–1776. doi: 10.1002/cncr.10398. [DOI] [PubMed] [Google Scholar]
- 46.Shibahara H, Tamada S, Higashi M, Goto M, Batra SK, Hollingsworth MA, et al. MUC4 is a novel prognostic factor of intrahepatic cholangiocarcinoma-mass forming type. Hepatology. 2004;39:220–229. doi: 10.1002/hep.20031. [DOI] [PubMed] [Google Scholar]
- 47.Singh PK, Hollingsworth MA. Cell surface-associated mucins in signal transduction. Trends Cell Biol. 2006;16:467–476. doi: 10.1016/j.tcb.2006.07.006. [DOI] [PubMed] [Google Scholar]
- 48.Maeda T, Taguchi K, Aishima S, Shimada M, Hintz D, LaRusso N, et al. Clinicopathological correlates of aspartyl (asparaginyl) β-hydroxylase overexpression in cholangiocarcinoma. Cancer Detect Prev. 2004;28:313–318. doi: 10.1016/j.cdp.2004.06.001. [DOI] [PubMed] [Google Scholar]
- 49.Chen Y, Xu J, Jhala N, Pawar P, Zhu ZB, Ma L, et al. Fas-mediated apoptosis in cholangiocarcinoma cells is enhanced by 3, 3′ diindolylmethane through inhibition of AKT signaling and FLICE-like inhibitory protein. Am J Pathol. 2006;169:1833–1842. doi: 10.2353/ajpath.2006.060234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Isomoto H, Mott JL, Kobayashi S, Werneburg NW, Bronk SF, Haan S, et al. Sustained IL-6/STAT-3 signaling in cholangiocarcinoma cells due to SOCS-3 epigenetic silencing. Gastroenterology. 2007;132:384–396. doi: 10.1053/j.gastro.2006.10.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Meng F, Henson R, Lang M, Wehbe H, Maheshwari S, Mendell J, et al. Involvement of human micro-RNA in growth and response to chemotherapy in human cholangiocarcinoma cell lines. Gastroenterology. 2006;130:2113–2129. doi: 10.1053/j.gastro.2006.02.057. [DOI] [PubMed] [Google Scholar]
- 52.Meng F, Wehbe-Janek H, Henson R, Smith H, Patel T. Epigenetic regulation of microRNA-370 by interleukin-6 in malignant human cholangiocytes. Oncogene. 2008;27:378–386. doi: 10.1038/sj.onc.1210648. [DOI] [PubMed] [Google Scholar]
- 53.Tang D, Nagano H, Yamamoto H, Wada H, Nakamura M, Kondo M, et al. Angiogenesis in cholangiocellular carcinoma: expression of vascular endothelial growth factor, angiopoeitin-1/2, thrombospondin-1 and clinicopathological significance. Oncol Rep. 2006;15:525–532. [PubMed] [Google Scholar]
- 54.Thomas MB. Biological characteristics of cancers in the gallbladder and biliary tract and targeted therapy. Crit Rev Oncol Hematol. 2007;61:44–51. doi: 10.1016/j.critrevonc.2006.07.006. [DOI] [PubMed] [Google Scholar]
- 55.Sirica AE, Zhang Z, Lai G-H, Asano T, ShenX-N, Ward DJ, et al. A novel “patient-like” model of cholangiocarcinoma progression based on bile duct inoculation of tumorigenic rat cholangiocyte cell lines. Hepatology. 2008;47:1178–1190. doi: 10.1002/hep.22088. [DOI] [PubMed] [Google Scholar]
- 56.Xu X, Kobayashi S, Qiao W, Li C, Xiao C, Radaeva S, et al. Induction of intrahepatic cholangiocellular carcinoma by liver-specific disruption of Smad4 and Pten in mice. J Clin Invest. 2006;116:1843–1852. doi: 10.1172/JCI27282. [DOI] [PMC free article] [PubMed] [Google Scholar]


