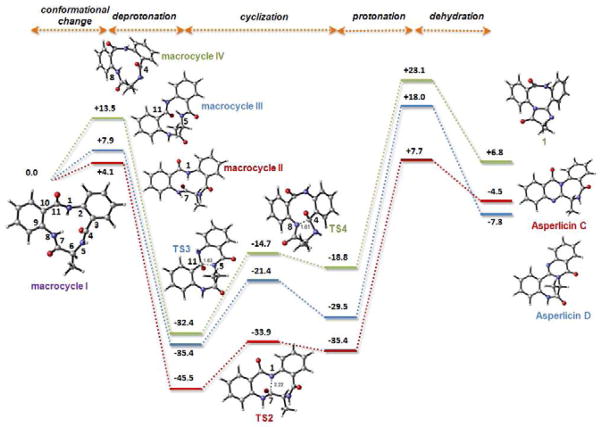
Figure 7.
Structures and relative energies calculated for the different conformations of the initial macrocycle, cyclization transition states and final dehydrated products relevant to the biosynthesis of asperlicin C, D and 1. The calculations were performed at the PCM(water)/B3LYP/6-31G(d) level using reduced models in which the indole ring from Trp side chain has been replaced by a methyl group. Amide bonds subjected to isomerization from trans to cis orientation prior to cyclization are marked in blue. Relative free energies are in kcal mol−1 and distances in Angstroms.