FIGURE 1.
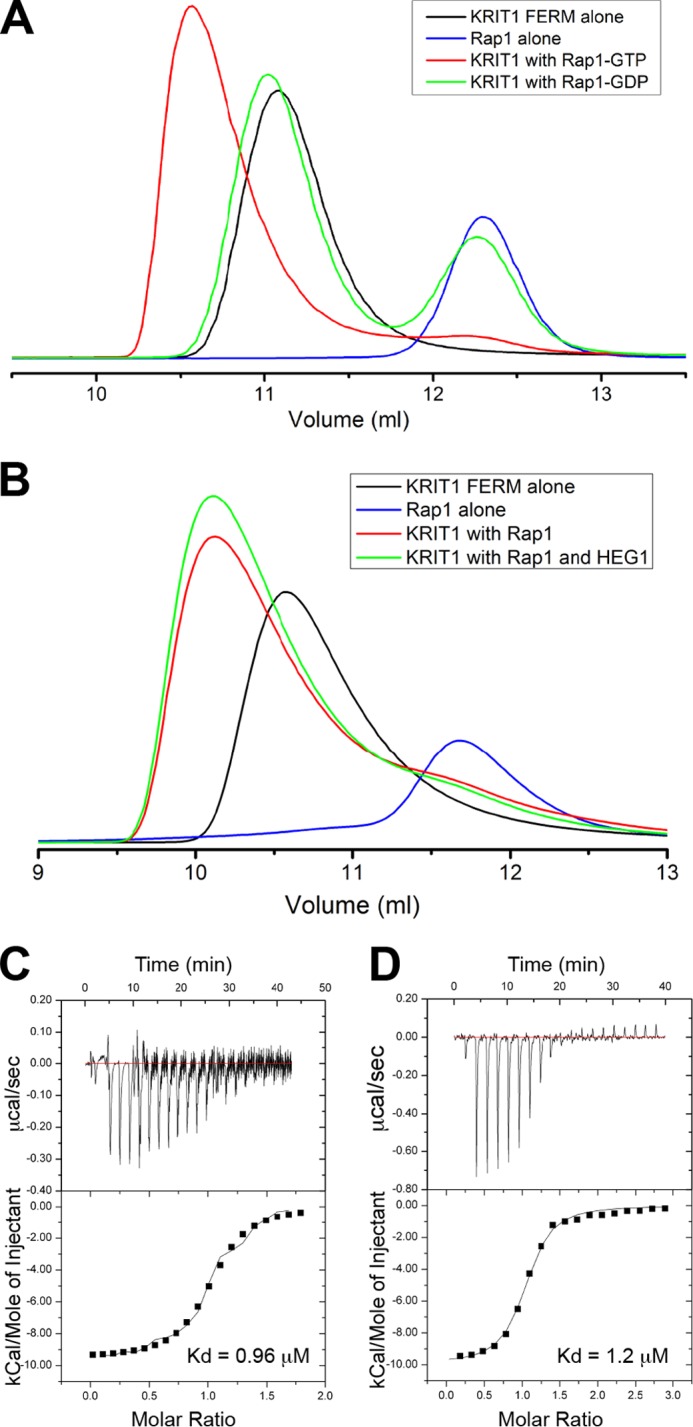
KRIT1-Rap1 binds HEG1, and they form a ternary complex. A and B, binding of the KRIT1 FERM domain to Rap1 was analyzed on a Superdex-75 (10/300) GL gel filtration column at room temperature. A, incubation of KRIT1 with Rap1 bound to GDP resulted in no complex formation, and most of the KRIT1 and Rap1 polypeptides remained in the free form. However, preincubation with Rap1 bound to GMP-PNP resulted in formation of a KRIT1-Rap1 complex. B, incubation of KRIT1 and Rap1-GTP (50 μm each) in the presence or absence 200 μm synthetic HEG1 cytoplasmic tail peptide. There is no competition between Rap1 and HEG1 for KRIT1 binding. C and D, calorimetric titration of HEG1 peptide out of the syringe into the following: C, KRIT1-Rap1 (Kd = 0.96 μm), and D, KRIT1 alone in the sample cell (Kd = 1.2 μm, re-plotted for comparison purposes (14)). For the titration of HEG1 into KRIT1-Rap1 the base line is noisy. We performed this experiment three times and obtained similar results.
