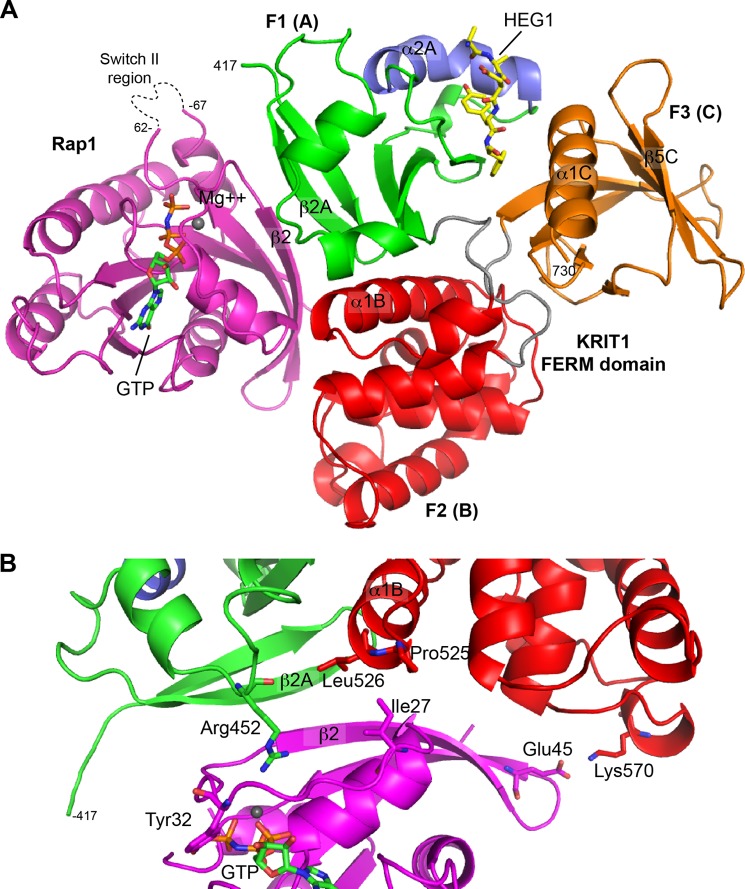
FIGURE 2.
Crystal structure of the ternary complex between the KRIT1 FERM domain, Rap1, and the HEG1 cytoplasmic tail. A, schematic representation of the KRIT1 FERM domain bound to Rap1 and the HEG1 cytoplasmic tail. The KRIT1 FERM domain contains three subdomains; F1 (green), F2 (red), and F3 (red). The F1 helix α2A is not seen in the other ubiquitin fold colored in blue. The HEG1 cytoplasmic tail (yellow) binds at the F1-F3 interface. Rap1 (magenta) bound to GMP-PNP binds at the surface of F1-F2. B, KRIT1 and Rap1 complex shows an extended binding interface involving both F1 and F2 regions of KRIT1. The KRIT1 β2A strand stacks against the β2 strand of Rap1 to form an extended β-sheet. The KRIT1 region surrounding Arg452 makes multiple water-mediated hydrogen bonds with Rap1. The KRIT1 residues Leu526 and Pro525 at the bottom of helix α1B form a hydrophobic surface that stacks with Ile27 of Rap1. A distal salt bridge is found between KRIT1 Lys570 and Glu45 of Rap1.