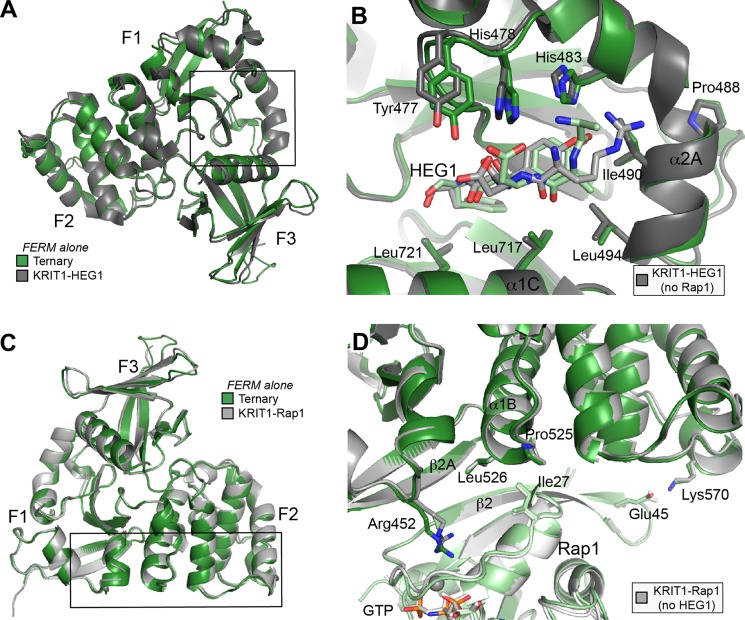
FIGURE 3.
Rap1 or HEG1 binding to the KRIT1 FERM domain does not induce conformational changes affecting binding at the other site. A, comparison of the KRIT1-HEG1 complex with the ternary complex structure. Only the FERM domain is shown to highlight that both structures are very similar. B, same superimposition as A with a focus on the HEG1 binding pocket located at the KRIT1 F1 and F3 interface. Overall the binding pocket is not affected by Rap1 binding. C, comparison of the KRIT1-Rap1 complex with the ternary complex structure. Only the FERM domain is shown to highlight that both structures are almost identical. D, same superimposition as C with a focus on the Rap1 binding interface at the KRIT1 F1/RBD and F2 surface. Overall the binding surface on KRIT1 is not affected by HEG1 binding.