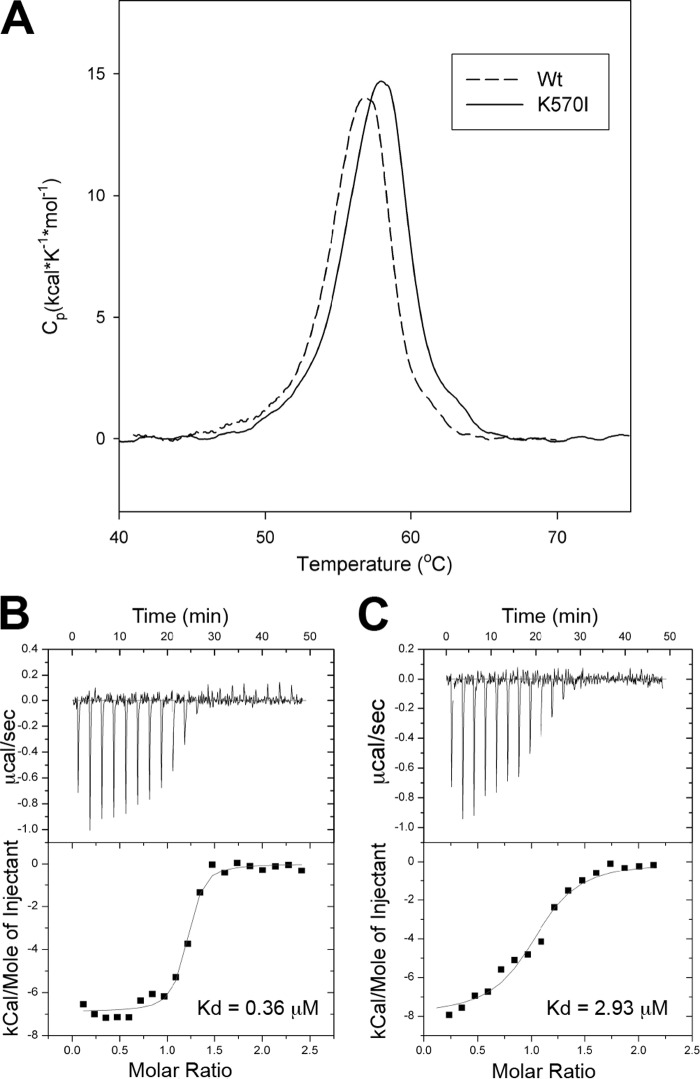
FIGURE 5.
KRIT1(K570I) has reduced binding affinity for Rap1. A, KRIT1(K570I) FERM mutant does not disrupt protein folding. Differential scanning calorimetry results of FERM WT and K570I proteins exhibit narrowly defined melting points, indicating that they are well folded. B and C, calorimetric titration of Rap1, out of the syringe into the following: A, KRIT1 wild-type, and B, KRIT1(K570I) mutant in the sample cell (Kd = 0.36 and 2.93 μm, respectively). The binding affinity is reduced by 8-fold.