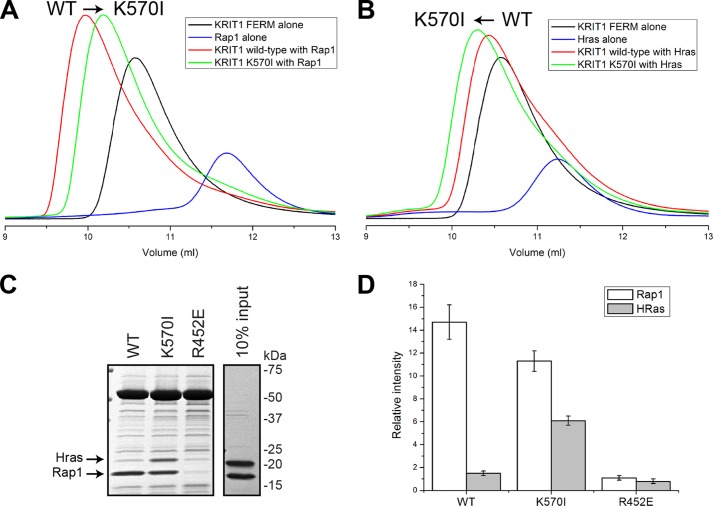
FIGURE 6.
KRIT1 is a Rap1-specific effector, and KRIT1 Lys570 is important for high affinity and specificity binding toward Rap1. A and B, binding of the KRIT1 FERM domain wild-type and FERM(K570I) to the following: A, Rap1, and B, HRas as analyzed on a Superdex-75 (10/300) GL gel filtration column at room temperature. A, incubation of KRIT1(K570I) with Rap1 results in complex formation with a shift toward increased Ve compared with wild-type KRIT1, suggesting a reduced affinity. B, in contrast, incubation of KRIT1(K570I) with HRas results in complex formation with a shift toward lower Ve compared with wild-type KRIT1, suggesting an increase in affinity. C, Coomassie stain of SDS-PAGE after KRIT1 pulldown. GST-KRIT1 wild-type pulls down Rap1 almost exclusively, whereas the KRIT1(K570I) pulls down both Rap1 and HRas. The KRIT1(R452E) does not bind either Rap1 or HRas. KRIT1 Lys570 is important for Rap1 binding specificity. D, quantification of Rap1 and HRas binding from three independent experiments such as shown in C. The KRIT1(K570I) mutation caused a 4-fold increase in HRas binding and a 25% reduction in Rap1 binding.