FIGURE 7.
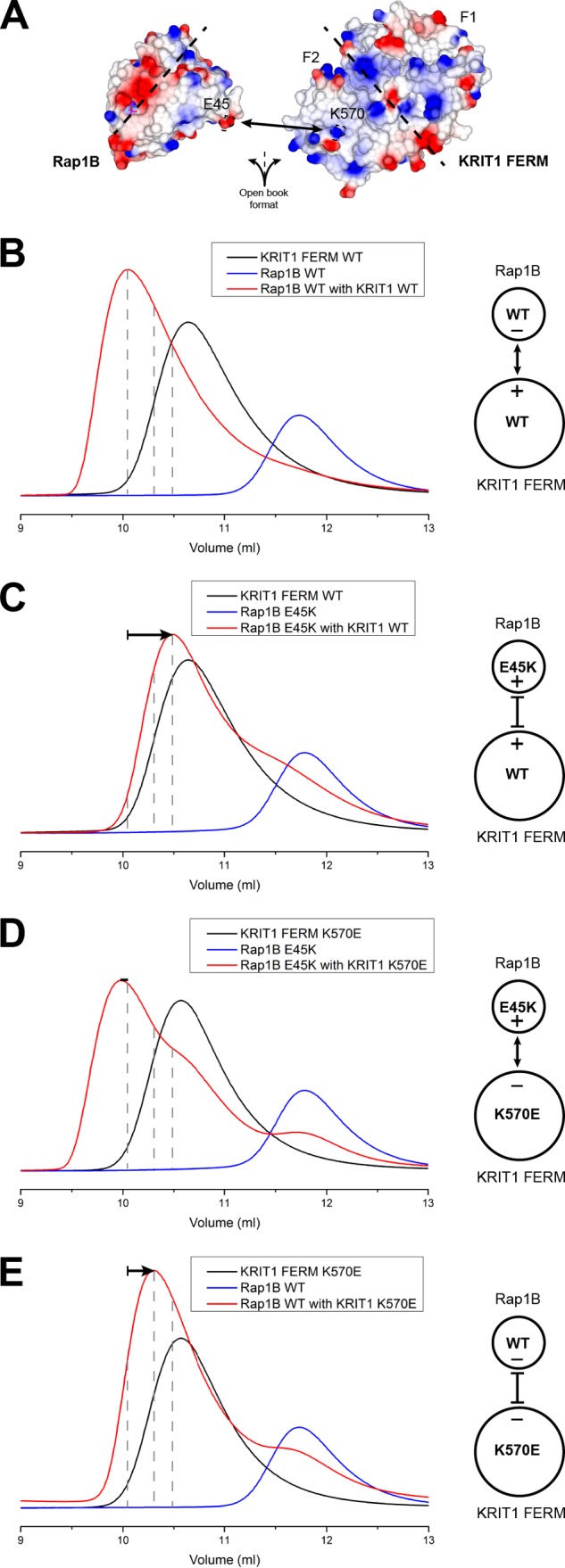
Ionic interaction between Rap1 Glu45 and KRIT1 Lys570 is important for high affinity binding. A, surface electrostatic potential of the Rap1 (left) and KRIT1 FERM (right) binding interface as open book view. The ionic interaction between Rap1 Glu45 and KRIT1 Lys570 is highlighted. The dotted line separates the binding interface for KRIT1 F1 and F2 subdomains. B–E, binding of the KRIT1 FERM domain to Rap1 as analyzed on a Superdex-75 (10/300) GL gel filtration column at room temperature. B, incubation of KRIT1 WT with Rap1 WT results in complex formation. C, incubation of KRIT1 WT with Rap1(E45K) results in complex formation with a shift toward increased Ve compared with wild-type KRIT1, suggesting a reduced affinity. D, incubation of KRIT1(K570E) with Rap(E45K) results in complex formation with a shift comparable with wild-type complex, suggesting binding has been recovered. E, incubation of KRIT1(K570E) with Rap1 WT results in complex formation with a shift toward increased Ve compared with wild-type KRIT1, suggesting a reduced affinity. The shift is less significant than C.
