FIGURE 8.
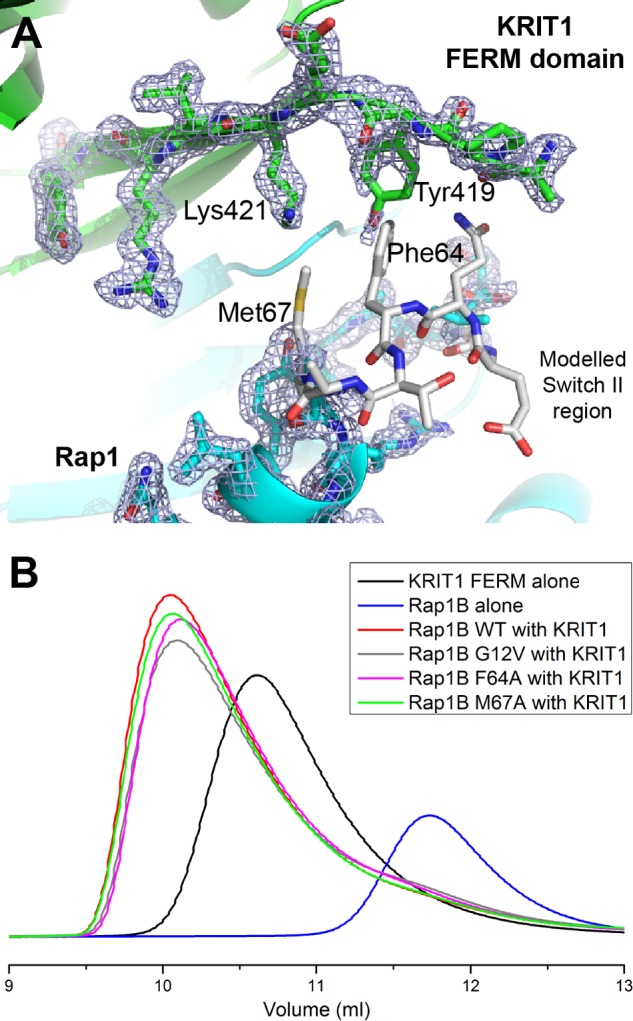
Rap1 Switch II is disordered in the crystal structure and does not play an important role in KRIT1 binding. A, close-up view of the KRIT1-Rap1 interaction in the vicinity of Rap1 Switch II region. The electron density is shown to highlight the absence of density for Rap1 residues 63–66 (2Fo − Fc map contoured at 1.0σ). KRIT1 is shown in green, Rap1 in blue, and the modeled region of Rap1 Switch II in white. B, binding of the KRIT1 FERM domain to Rap1 as analyzed on a Superdex-75 (10/300) GL gel filtration column at room temperature. Incubation of KRIT1 WT with various Rap1 mutants results in complex formation with a Ve comparable with wild-type complex, suggesting that binding is not affected.
