Abstract
Background and Aims
Ontogenetic patterns of odour emissions and heating associated with plant reproductive structures may have profound effects on insect behaviour, and consequently on pollination. In some cycads, notably Macrozamia, temporal changes in emission of specific odour compounds and temperature have been interpreted as a ‘push–pull’ interaction in which pollinators are either attracted or repelled according to the concentration of the emitted volatiles. To establish which mechanisms occur in the large Encephalartos cycad clade, the temporal patterns of volatile emissions, heating and pollinator activity of cones of Encephalartos villosus in the Eastern Cape (EC) and KwaZulu Natal (KZN) of South Africa were investigated.
Methods and Key Results
Gas chromatography–mass spectrometry (GC-MS) analyses of Encephalartos villosus cone volatiles showed that emissions, dominated by eucalyptol and 2-isopropyl-3-methoxypyrazine in EC populations and (3E)-1,3-octadiene and (3E,5Z)-1,3,5-octatriene in the KZN populations, varied across developmental stages but did not vary significantly on a daily cycle. Heating in male cones was higher at dehiscence than during pre- and post-dehiscence, and reached a maximum at about 1830 h when temperatures were between 7·0 and 12·0 °C above ambient. Daily heating of female cones was less pronounced and reached a maximum at about 1345 h when it was on average between 0·9 and 3·0 °C above ambient. Insect abundance on male cones was higher at dehiscence than at the other stages and significantly higher in the afternoon than in the morning and evening.
Conclusions
There are pronounced developmental changes in volatile emissions and heating in E. villosus cones, as well as strong daily changes in thermogenesis. Daily patterns of volatile emissions and pollinator abundance in E. villosus are different from those observed in some Macrozamia cycads and not consistent with the push–pull pattern as periods of peak odour emission do not coincide with mass exodus of insects from male cones.
Keywords: Encephalartos villosus, Zamiaceae, cycads, monoterpenes, developmental stages, nitrogen-containing compounds, unsaturated hydrocarbons, gas chromatography–mass spectrometry, push–pull, odour emission, floral volatiles, thermogensis, pollination
INTRODUCTION
Floral volatiles have been identified as important signals for chemical communication between plants and animal pollinators across a range of plants including archaic angiosperms (Pellmyr and Thien, 1986) and cycads (Pellmyr et al., 1991; Tang, 1993). These olfactory signals are usually characterized primarily by fatty acid derivatives, benzenoids and terpenoids (Thien et al., 2000; Raguso, 2004, 2008; Knudsen et al., 2006). Pellmyr and Thien (1986) noted that some chemical attractants were the same as or derived from known herbivore deterrents, which led them to suggest that olfactory attractants could have evolved from herbivore deterrents in circumstances where the interaction confers a net benefit to the plant because the herbivore also functions as a pollinator.
All plant reproductive structures undergo metabolic biochemical activity, with heat as a product of these reactions (Seymour and Schultz-Motel, 1997), but thermogenic plants produce large amounts of heat beyond that which is simply a by-product of regular metabolic activity (Seymour and Shultz-Motel, 1997). Periods of heat production normally correspond to the release of volatile compounds and insect activity in receptive female flowers of angiosperms (Seymour and Shultz-Motel, 1997, 1999; Seymour, 1999; Gibernau and Barabé, 2000; Seymour and Baylock, 2000; Seymour and Matthews, 2006) and have been shown to occur in dehiscing male cones of cycads (e.g. Tang, 1987a, 1993; Seymour et al., 2004; Suinyuy et al., 2010). Heat production is considered to enhance production and volatilization of volatile compounds, which attracts insects (Tang, 1993; Seymour and Schultz-Motel, 1997, 1999; Ervik and Barfod, 1999; Seymour and Matthews, 2006).
The simultaneous production of floral odour and heat when pollinating insects are active on flowers suggests that they individually or collectively influence pollinator behaviour. Floral odour and heat production are dynamic processes, and variations in these floral signals are probably adaptive mechanisms that regulate insect pollinator behaviour. Ontogenetic (developmental) changes in floral odour and heat production have also been shown to influence pollinator behaviour (e.g. Schiestl et al., 1997; Seymour and Matthews, 2006). In such scenarios, changes in floral odour composition and heat production may correspond to specific pollinator behaviour at different stages of flower development (Kumano and Yamaoka, 2006; Maia and Schlindwein, 2006; Theis et al., 2007).
Floral odour and heat are characteristic of the reproductive structures of some cycads and archaic angiosperms (e.g. Pellmyr et al., 1991; Tang, 1993, 1987a). All cycads are dioecious, and insect pollinators have to visit both male and female plants for pollination to occur. Effective pollination requires pollinators to move from male to female cones or to move back and forth between the male and female cones, as observed by Terry et al. (2007a, b). The known pollinators of almost all cycads are insect herbivores whose larvae feed on male cone tissues (including pollen) of the host cycad, so that the male cone serves as the larval brood site (Norstog et al., 1986; Tang, 1987b; Donaldson, 1997; Mound and Terry, 2001; Hall et al., 2004; Terry et al., 2005; Suinyuy et al., 2009). In such a system, the cues affecting specific insect pollinator behaviour, such as aggregation, attraction, repellence, mating and oviposition, may differ between male and female plants of the host species as well as at different times of the day or stages of cone development.
Until recently it was assumed that volatiles and heat production in cycads function solely to attract pollinators to inflorescences, but Terry et al. (2004) observed that insect pollinators actually left male cones of several Australian cycads (Macrozamia spp.) during periods of peak volatile emission, which also coincided with peaks in cone temperature as a result of thermogenesis. Their olfactometer experiments showed that the pollinator Cycadothrips chadwickii was attracted to Macrozamia lucida sporophylls early in the day, repelled at mid-day, and attracted to them again in the late afternoon to early evening (Terry et al., 2007a, b). This behaviour corresponded to daily field observations which showed that thrips were present in male cones in the morning when volatile emissions and cone temperature were low, left male cones at mid-day when volatile emissions and cone temperature were high, and were attracted later in the day when cone emissions and temperature were low again (e.g. Terry et al., 2004). Considering β-myrcene, it was shown that attraction and repellence were concentration dependent, and this system was referred to as a ‘push–pull’ pollination strategy (Terry et al., 2007a, b).
Although a push–pull pollination strategy has been observed in the Australian Macrozamia cycads, it is not known whether it is a general phenomenon in cycads or whether there are different pollination mechanisms in this group of plants. Cone volatiles in the African cycad genus Encephalartos have been linked to the attraction of pollinators since Pearson's (1906) and Rattray's (1913) interpretations that weevils, such as Porthetes spp. (Coleoptera: Curculionidae: Molytinae, initially identified as Phlaeophagus), were possible pollinators of Encephalartos villosus Lehm. and E. altensteinii. The pollination period in male and female cones of Encephalartos is characterized by fluctuating daily emissions of odour and/or heat production (Tang, 1987a, 1993; Donaldson, 1997; Suinyuy et al., 2010). During early stages of development, male cones of Encephalartos spp. have tightly packed sporophylls that separate a few days before pollen release. At the time of pollination, male and female cones may emit similar volatile compounds and male cones typically undergo periods of heating. These changes in the cone coincide with more frequent insect visitation on male cones (e.g. Suinyuy et al., 2010). These observations suggest that volatile emissions are linked to insect behaviour and function in attracting insect pollinators (Tang, 1993). It has also been suggested that cone heating may enhance release of attractive odours and increase insect movement between cones (Tang, 1993). In a detailed investigation of the pollination biology of E. villosus, Donaldson (1997) showed that peak temperatures in the evenings in dehiscing male cones coincided with the presence of insects. The simultaneous production of cone volatiles and heat and the occurrence of insects on cones strongly suggests that these physiological phenomena are important signals in regulating pollinator behaviour, but do not provide specific evidence for attraction and repellence.
In dioecious plants, it is necessary to determine the mechanisms that regulate pollinator behaviour and consequently influence pollination. In some Macrozamia cycads, such mechanisms involve push–pull interactions which comprise regular daily changes in levels of volatile emissions and heat production (Terry et al., 2004, 2007a, b) such that periods of attraction alternate with periods of repellence. Based on observations of some Encephalartos species, Donaldson (2007) proposed several possible mechanisms in which the factors influencing movement of pollinators away from male cones could vary with changes in the composition of volatile emissions and heat production over the developmental cycle of cones. This is especially true for the brood site in male cones which transform from a pre-dehiscent state to dehiscence and subsequent drying or decomposition over a period of 2–3 weeks. The simplest alternative to the push–pull interaction proposed for Macrozamia is that insects will respond to specific cues associated with cycad host plants at the time of pollen dehiscence and disperse from the male cone later due to declining quality of the cone. More detailed studies of patterns of cone volatile emission and heat production over the period of pollination in cycads such as Encephalartos spp. can be used to test whether the original push–pull concept or any of the hypothetical alternatives applies to other cycad genera.
The main aim of this study was to examine patterns of cone odour emissions and heating in both male and female E. villosus to determine whether these patterns are consistent with daily push–pull interactions such as those observed in Macrozamia cycads (Terry et al., 2004, 2007a, b) or with the mixed models proposed by Donaldson (2007). These alternative hypotheses were tested by measuring odour emissions, heating and insect abundance daily over the life span of both male and female cones.
MATERIALS AND METHODS
Study species and area
Populations of Encephalartos villosus occur mainly in patches of Scarp Forest and Northern Coastal Forest (Mucina and Geldenhuys, 2006). Monoterpenes and nitrogen-containing compounds characterize the odour profiles of E. villosus plants from Eastern Cape Province (EC), while unsaturated hydrocarbons are dominant in plants from KwaZulu Natal (KZN) populations (Suinyuy et al., 2012). The study focused on populations from both regions, comprising Mount Sullivan and Ocean View Guest Farm, occurring in forests in the Transkei Coastal Belt vegetation type of the EC, and additional populations in the Kranzkloof Nature Reserve in the KZN Coastal Belt. The study also included cultivated plants at the Kirstenbosch Botanic Garden in Cape Town which are derived from areas in the EC region, and at the Pietermaritzburg campus of the University of KwaZulu Natal which are derived from localities in the KZN region. Male and female cones of E. villosus emerge during January and February. The male cone extends and the sporophylls become separated around March and April when pollen is shed. After pollination, the male cone disintegrates but the female cone remains on the plants until they mature and eventually disintegrate between September and November.
Developmental stages of cones
The developmental stages were determined through observation of the cones, starting several weeks before pollen dehiscence and receptivity in male and female cones, respectively. Male cones were classified as pre-dehiscent until the period when the cone elongated and the sporophylls separated (Fig. 1A), as dehiscent when the cone elongated and there was clear separation between the sporophylls and the microsporangia released pollen (Fig. 1B), and as post-dehiscent when the microsporangia ceased shedding pollen, typically when the cone also started to disintegrate (Fig. 1C). Male cones before pollen dehiscence had a height of about 300 mm and diameter of 6·2 mm, and at pollen dehiscence the cones extend up to 620 mm in height and about 130 mm in diameter until they start disintegrating. Female cones were classified as pre-receptive until the cone had reached its full size and the sporophylls on the top half of the cone had separated to reveal small gaps; receptive when the sporophylls were maximally separated; and post-receptive when the sporophylls were again tightly closed.
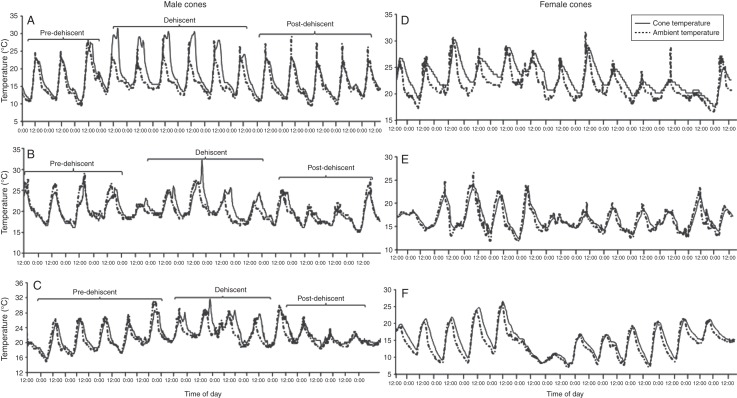
Fig. 1.
Developmental stages in male cones of E. villosus: (A) pre-dehiscent; (B) dehiscent; (C) post-dehiscent. Scale bars: (A) = 300 mm, (B, C) = 600 mm.
Temperature measurements
Temperatures of male and female cones of E. villosus were monitored in situ for each key developmental stage during two reproductive events (2008 and 2009). Temperatures were measured using thermochron ibutton data loggers and Climastats software (FairBridge Technologies, South Africa) accurate to ±0·5 °C and set to record the temperature at 10 min intervals over at least a 24 h period. At the Kirstenbosch Botanic Garden, temperatures were measured for two male and female cones, respectively, throughout their life cycle during the first reproductive event in the 2008 field season. During the second reproductive event in the 2009 field season, cone temperatures were measured for plants growing at the Ocean View Guest Farm (four male cones and two female cones), Mount Sullivan (seven male cones and one female cone), Kranzkloof Nature Reserve (six male cones and four female cones) and the Botanic Garden of the University of KwaZulu Natal, Pietermaritzburg campus (three male cones and two female cones). Two ibuttons were used for each cone, one inserted between the cone sporophylls to measure cone temperature and the other placed between the leaves of the same cycad to measure ambient temperature. Both ibuttons were positioned in such a way as to avoid direct exposure to sunlight. Because E. villosus occur in forest habitat such that the canopy prevents sunlight from reaching the forest floor, the cones were not shaded.
Cone odour sampling
Headspace sampling was used to collect volatiles from male and female cones of E. villosus in 2007–2009. Polyacetate bags (Nalo Bratfolie Kalle GmbH, Germany) were placed over the entire cone just prior to sampling in order to concentrate the volatile compounds. Air from inside the bags was suctioned for 30 min into an adsorbent trap using a portable battery-operated pump (Spectrex Personal Air Sampler PAS 500, USA) calibrated at 200 mL min−1. Air samples were simultaneously collected from empty polyacetate bags placed way from the plant as controls to identify background contamination. The trap samples were stored at –20 °C in a sealed vial until analysis. The traps contained 2 mg of a 50:50 mixture of Tenax TA® (Alltech Associates, USA) and graphitized carbon (Carbotrap™, Supelco, USA) in a glass tube closed at both ends with glass wool. At each key developmental stage, cones were sampled in the morning (between 0900 h and 1100 h), afternoon (1330–1530 h) and evening (1700–1900 h), and the number of replicates depended on the availability of cones. In the EC, odour samples were collected from five male and four female plants at each developmental stage for each time period (morning, afternoon and evening). In KZN, odour samples were obtained from nine pre-dehiscent male cones, 11 dehiscent and post-dehiscent male cones, four pre-receptive female cones, five receptive female cones and three post-receptive female cones.
Chemical analysis and compound identification
Volatile samples were analysed using a coupled Varian 3800 gas chromatograph (Varian Palo Alto, CA, USA) and Varian 1200 mass spectrometer. The gas chromatograph was equipped with an Alltech EC–WAX column of 30 m × 0·32 mm internal diameter × 0·25 µm film thickness (Alltech Associates Inc., Deerfield, IL, USA). Helium was used as the carrier gas at a flow rate of 1 mL min−1. After sampling, traps were placed in a Varian 1079 injector by means of a Chromatoprobe fitting and thermally desorbed. The injector was heated to 40 °C for 2 min with a 20:1 split ratio and then increased to 200 °C, and then held at 200 °C min−1 in splitless mode for thermal desorption. After a 3 min hold at 40 °C, the gas chromatograph oven was ramped up to 240 °C at 10 °C min−1 and held there for 12 min. Compound identification was carried out using the Varian Workstation software with NIST05 mass spectral library and comparisons with retention times of chemical standards, where available, as well as comparisons between calculated Kovats retention indices and those published in the literature. A homologous series of alkanes (C8–C20) was used to determine Kovats retention indices. Compounds were verified using retention times of authentic standards (97–99·5 %, Sigma Aldrich Inc. GmbH, Germany), and (3E)-1,3-octadiene (98 %, ChemSampco, USA), and published Kovats indices. Compounds present at higher or similar percentages in controls were considered as contaminants and excluded from the analysis. For quantification of emission rates, known amounts of standards of dominant compounds were injected into cartridges and thermally desorbed under identical conditions to the samples. The peak areas of compounds in the samples were compared with those of the standards and used to calculate the total emission rates of compounds per cone per hour and emission rate per compound per cone per hour. Individual compounds comprising ≥10 % of the average relative amount that were emitted in almost all stages and during some times of day in males and females were considered dominant compounds.
Insect visitors to male and female cones
Insects present on male and female cones of E. villosus in the field were sampled at all cone stages throughout the duration of field work (2007–2009). They were sampled at approximately the same times as odour sampling in the morning (0900–1100 h), afternoon (1330–1530 h) and evening (1700–1900 h). The insects were collected from male cones by shaking the cones over a beating sheet. Five male cones were surveyed for each key developmental stage in both EC and KZN, and for each stage the same cones were sampled in the morning, afternoon and evening. Sampling of insects on five female cones was restricted to insects crawling on the surface of the cones as these could be collected without damaging the cone. The insects were counted and recorded before storing in alcohol, and were later identified based on keys by Endrödy-Younga (1991), Donaldson (1991) and Oberprieler (1996). Voucher specimens of insects have been deposited in the entomological collection of the school of Life Sciences of the University of KwaZulu Natal, Pietermaritzburg campus, South Africa.
Statistical analysis
The mean temperatures above ambient and mean maximum cone temperatures were determined, as well as the standard errors of the mean of the cone temperature. To test for significant differences in the rates of emission of compounds for different cone stages and times of day, data were analysed using analysis of variance (ANOVA) in STATISTICA version 7. The first analysis tested the overall differences in the rate of emission of all compounds for different cone stages and times of day and the second analysis tested differences in rates of emission of individual dominant compounds across all the stages. This was followed by Tukey's honest significant difference (HSD) method of pairwise multicomparison (Zar, 1984). In graph construction, the mean emission rates as well as the standard error of the mean of each of the major compounds were determined.
To test whether there were significant differences among the total number of insects associated with cones at different developmental stages and times of day, data were analysed using the Kruskal–Wallis test (because data were not normally distributed), followed by the Tukey's HSD method for pairwise multicomparisons (Sokal and Rohlf, 1995). Because few insects were collected from female cones, analyses were carried out only for insects collected from male cones.
RESULTS
Temperature changes in male and female cones
In male plants, the general pattern of cone heating at the pollen dehiscent stage comprised a consistent rise in cone temperature above concurrent ambient temperature in the late afternoon when air temperatures were cooling (Fig. 2A–C). In some cones, heating occurred in the late morning to early afternoon when both cone and ambient temperatures increased simultaneously. Such cones sometimes show a bimodal pattern of heating, with a first peak in the late morning to early afternoon and a second peak in the evening. In comparison, near ambient cone temperatures were recorded at pre-dehiscent and post-dehiscent stages (Fig. 2A–C).
Fig. 2.
Pattern of cone heating in relation to ambient temperature in: (A–C) three male cones of E. villosus over the developmental cycle; and (D–F) three female cones over the developmental cycle. Ambient and cone temperatures as indicated in the key in (D).
Cone heating due to thermogenesis appeared to be limited to between 3 and 7 days during the dehiscent phase and peaks in cone temperature occurred mostly between 1800 and 1830 h during sunset. At this time, cone temperatures rose above concurrent ambient temperatures which were cooling, making thermogenesis easy to identify (Fig. 2A–C). Cones in the EC and KZN reached their mean maximum temperatures of between 29·7 ± 0·8 °C (n = 5) and 31·7 ± 0·4 °C (n = 5), respectively, between 1600 and 1900 h, and reached the peak at an average time of 1822 h. Cone temperatures were between 7·1 ± 1·3 °C and 9·3 ± 1·0 °C warmer than ambient. Despite the absence of thermogenesis in the majority of pre-dehiscent male cones, five male cones (three in EC and two in KZN) did have temperatures above ambient in the pre-dehiscent stage. In these cases, heating began from late morning (between 1000 h and 1040 h) and reached a daily mean maximum between 1150 h and 1507 h. The cone temperature exceeded mean ambient temperature by between 2·0 and 4·5 °C. Because the cones were not shaded from sunlight, peaks in temperature on pre-dehiscent cones could be a response to solar heating as early sun rays in the morning that strike the cones could quickly cause a spike in heat production (Seymour and Shultz-Motel, 1999).
In general, there was little or no increase in temperature in female cones throughout the life cycle of the cone (Fig. 2D–F). In some cases, cone temperatures increased simultaneously with ambient temperature and only rose above concurrent ambient temperature when ambient temperature started decreasing in the evening (Fig. 2D, E). In contrast to this pattern, a relatively small thermogenic effect appeared to occur in three female cones: cone temperature continued to increase up to approx. 2·0 °C above the maximum ambient temperature over a number of days during the life cycle of the cone (Fig. 2F). However, the small increases in cone temperature that follow the peaks in ambient temperature could simply represent a lag in the cooling of the cone relative to a more rapid cooling in ambient temperature.
Volatile emissions
As previously described, E. villosus cones emit a large number of different volatile compounds (Suinyuy et al., 2012; see Supplementary Data Tables S1 and S2). The cone odour of E. villosus from EC is dominated by the nitrogen-containing compound 2-isopropyl-3-methoxypyrazine and the monoterpene eucalyptol, with α-terpinene, α-pinene and β-pinene contributing substantial amounts; whereas unsaturated hydrocarbons (3E)-1,3-octadiene and (3E,5Z)-1,3,5-octatriene characterize the odour of KZN plants (Suinyuy et al., 2012; see Supplementary Data Tables S1 and S2). Monoterpenes are the most numerous compounds emitted by E. villosus cones, followed by aliphatic compounds which consist of benzenoids, aldehydes, ketones, alcohols and aliphatic acids. In the EC plants, male and female cones emitted 63 identified and one unidentified compound, while in the KZN plants they emitted 69 identified and one unidentified compound, respectively (Supplementary Data Tables S1 and S2).
Variation between cone stages and times of day
Changes in emission rates
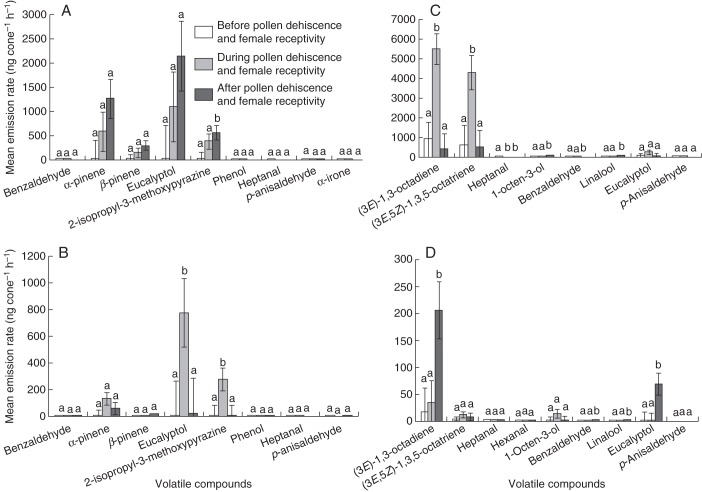
There is a clear quantitative variation in odour production patterns between the different life stages (Supplementary Data Tables S1 and S2). In male EC plants, the mean emission rates per cone per hour were highest in cones at the post-dehiscent stage (6140·91 ± 2663·46 ng cone−1 h−1, n = 15) and were significantly greater than in cones at the dehiscence (2779·55 ± 812·06, n = 15) and pre-dehiscence stages (29·43 ± 10·12, n = 15) (ANOVA, F2,36 = 3·29; P < 0·05). Variation in volatile emission rates between the cone stages was accounted for by a few dominant compounds emitted in almost all cone stages at different emission rates (Fig. 3A; Supplementary Data Table S1). The emission rates of 2-isopropyl-3-methoxypyrazine at the post-dehiscent stage were significantly greater (P ≤ 0·05) than at the dehiscent and pre-dehiscent stage, and those of α-pinene and eucalyptol were greater at post-dehiscence but not significantly different from those at dehiscence and pre-dehiscence (P > 0·05) (Fig. 3A), and largely accounted for the overall high emission rates at post-dehiscence.
Fig. 3.
Variation of scent emission rates (ng cone−1 h−1) of dominant compounds emitted before, during and after pollen dehiscence and female receptivity in: (A) male cones (number of samples n = 15 at all stages) and (B) female cones (number of samples n = 12, 13 and 12, respectively) in the Eastern Cape; and (C) male cones (number of samples n = 27, 33 and 33, respectively) and (D) female cones (number of samples n = 12, 15 and 9, respectively) in KwaZulu Natal. Bars represents the mean (± s.e.). Letters above bars denote homogeneous groups for each compound using Tukey's honest significant difference (HSD) test.
In female EC plants, the mean emission rate was highest in cones at the pollen receptive stage (1375·20 ± 702·05 ng cone−1 h−1, n = 12) and significantly greater than emissions in cones at the pre-receptive (20·45 ± 7·06, n = 13) and post-receptive stages (121·19 ± 52·51, n = 12) (ANOVA, F2,28 = 2·89; P ≤ 0·05). As in the male cones, a few compounds dominated the volatile profile of the female cones, with eucalyptol, 2-isopropyl-3-methoxypyrazine and α-pinene having the highest emission rates, with maximum emission recorded at the receptive stage (Fig. 3B). Emissions rates at the receptive stage were significantly greater for eucalyptol and 2-isopropyl-3-methoxypyrazine (P ≤ 0·05) than at the post-receptive and pre-receptive stages (Fig. 3B).
Although scent emission rates differed between the different cone stages in male and female cones of E. villosus from EC plants (Supplementary Data Table S1), overall emissions did not differ between different times of the day for male cones (ANOVA, F2,36 = 5·31, P > 0·05) and female cones (ANOVA, F2,28 = 0·23, P > 0·05), nor did they differ for individual dominant compounds.
In KZN plants, variation in volatile emission rates between the different cone stages were also accounted for by a few dominant compounds emitted by male and female cones (Fig. 3C, D). In male cones, the mean emission rates in cones at the dehiscent stage (10 638·69 ± 2057·14 ng cone−1 h−1, n = 33) were significantly greater than in cones at the pre-dehiscent stage (1761·58 ± 743·24, n = 27) and post-dehiscent stage (1603·31 ± 466·34, n = 33) (ANOVA, F2,84 = 17·20; P < 0·001). These differences were accounted for by emissions of (3E)-1,3-octadiene and (3E,5Z)-1,3,5-octatriene which were significantly higher (P ≤ 0·05) than those at pre-dehiscent and post-dehiscent stages (Fig. 3C). Benzaldehyde, 1-octen-3-ol and linalool were significantly higher (P ≤ 0·05) at post-dehiscence, and heptanal was significantly higher (P ≤ 0·05) at pre-dehiscence (Fig. 3C).
In female KZN plants, the mean emission rates at the post-receptive stage (360·88 ± 123·90 ng cone−1 h−1, n = 9) were significantly greater than in cones at the receptive stage (72·92 ± 22·30, n = 15) and pre-receptive stage (23·58 ± 17·32, n = 12) (ANOVA, F2,27 = 7·64; P < 0·01). The differences were accounted for by (3E)-1,3-octadiene, eucalyptol, benzaldehyde and linalool that were emitted in significantly higher amounts at the post-receptive stage (P ≤ 0·05) than at the pre-receptive and receptive stages (Fig. 3D).
In male cones, overall rates of odour emission at different times of the day were highest in the evening (7606·42 ± 2020·19 ng cone−1 h−1) and were significantly greater than emissions in the afternoon (4180·30 ± 1465·16) and in the morning (2777·37 ± 818·58) (ANOVA, F2,84 = 3·40, P < 0·05, n = 31). The overall significant difference between times of day observed in male cones at dehiscence was accounted for by the emission of two compounds, 1-octen-3-ol and p-anisaldehyde. The emission rate of 1-octen-3-ol in the evening (83·69 ± 35·72 ng cone−1 h−1) was significantly greater than emissions in the morning (13·51 ± 4·21) and afternoon (11·51 ± 4·09) (ANOVA, F2,30 = 3·51, P < 0·05, n = 31). The emission rate of p-anisaldehyde was significantly greater in the afternoon (4·71 ± 1·83) (ANOVA, F2, 30 = 4·02, P <0·05, n = 31) than in the evening (1·16 ± 0·41) and morning (0·74 ± 0·18). There were no significant differences in the emission rates of compounds in female cones at different times of the day (P > 0·05).
Compositional change
Change in volatile composition was prominent across life stages of EC and KZN plants (Supplementary Data Tables S1 and S2). The male cones of EC plants emitted detectable levels of 30 compounds at pre-dehiscence, 37 at dehiscence and 32 at post-dehiscence stages. Compositional change occurred mostly in minor compounds and was dominated by the terpenoids, with particular compounds detected only in two stages. For example, β-thujene, camphene, α-terpinene, carveol, 4-terpineol, citral, piperitone oxide, cis-geraniol and p-cymen-3-ol were detected at dehiscence and post-dehiscence (Supplementary Data Table S1). In contrast, two terpenoids, limonene and cis-p-menth-2-en-ol, and the benzenoids methyl benzoate and benzyl alcohol were present in scent of E. villosus before and during pollen dehiscence (Supplementary Data Table S1).
The female cones emitted 35 compounds at pre-receptive, 41 at receptive and 25 at post-receptive stages. Compositional change in female cones was similar to that of male cones in that it occurred mostly in minor compounds. Aliphatic acids were detected mostly before and during pollen receptivity or before and after receptivity, with a few compounds detected at one stage (Supplementary Data Table S1). Terpernoids were detected mostly in two stages either during and after pollen receptivity or before and during pollen receptivity (Supplementary Data Table S1).
In the KZN plants, the male cones emitted detectable levels of 31 compounds at pre-dehiscence, 42 at dehiscence and 52 at post-dehiscence (Supplementary Data Table S2). Compositional change in male cones of KZN plants was dominated by terpenoids, benzenoids and the aldehydes of aliphatic compounds (Supplementary Data Table S2). A few minor compounds were only detected during dehiscence (e.g. trans-β-ocimene, piperitone oxide and carvone), while some were detected at dehiscence and post-dehiscence stages [e.g. α-terpineol, borneol, (E, E)-α-farnesene and α-curcumene]. The majority of the compounds were detected only at post-dehiscence (Supplementary Data Table S2).
The female cones emitted 11 compounds at pre-receptive, 25 at receptive and 24 at post-receptive stages (Supplementary Data Table S2). In the female cones, compositional change occurred in a few compounds, but most of the compounds were detected in one stage. For example, acetic and heptanoic acids, 1-octanol, α-irone, camphor, myrtenal and myrtenol were detected at the post-receptive stage, while (2E,4E)-hepta-2,4-dienal, 2,4-octadienal, anisole, α-pinene and cis-β-ocimene occurred only at the receptive stage (Supplementary Data Table S2).
Insect visitors to male and female cones
Four species of beetles (Coleoptera) were collected from E. villosus male cones in both EC and KZN: Antliarhinus zamiae Thunberg (Curculionoidea: Brentidae), Erotylidae sp. nov. (Cucujoidea), Metacucujus goodei Endrödy-Younga (Cucujoidea; Boganiidae) and Porthetes sp. (Curculionidae) (Suinyuy et al., 2012). The Porthetes species was provisionally named as P. pearsonii by Oberprieler (1996). Antliarhinus zamiae is sexually dimorphic, and the female insect, which is about 25 mm long, has an elongated rostrum about the same length as the body while the male insect with a short rostrum is about 7 mm long (Donaldson, 1997). Metacucujus goodei, Erotylidae sp. nov. and Porthetes sp. all range in length from 3 to 8 mm (Donaldson, 1997).
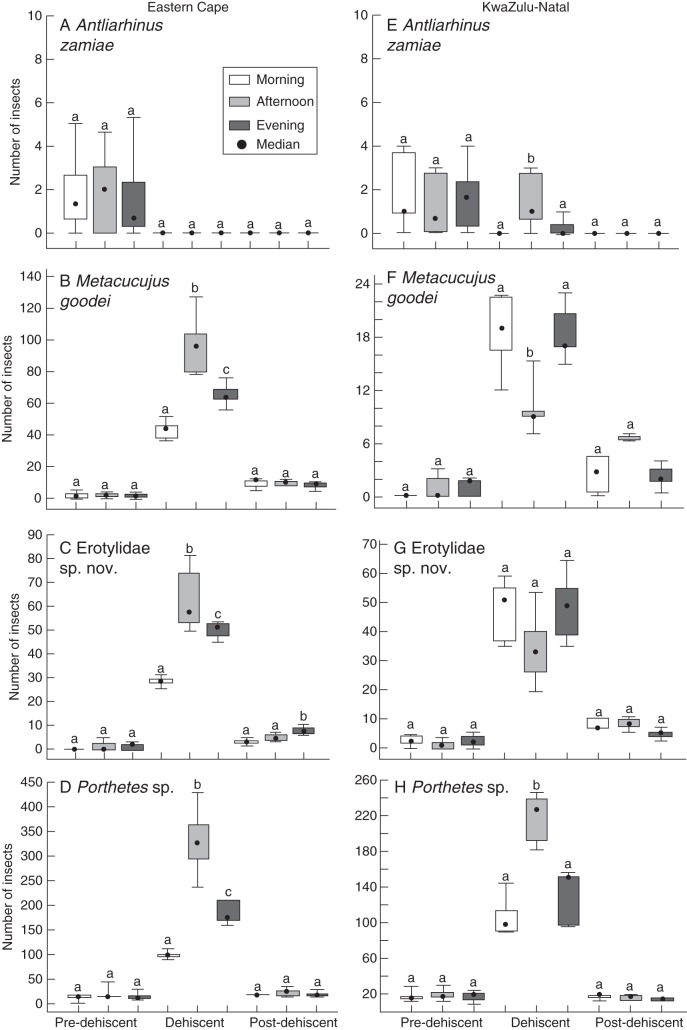
With the exception of small numbers of A. zamiae individuals on pre-dehiscent cones, all the beetles were most abundant on dehiscent male cones (Fig. 4A–H). At pre-dehiscence, A. zamiae were observed crawling over the cone surface while Erotylidae sp. nov. and M. goodei were mostly located at the base of the cones and Porthetes sp. were seen crawling over the cone surface, forcing their way between the tightly packed sporophylls. At dehiscence, the insects present on cones (Erotylidae sp. nov., M. goodei and Porthetes sp.) were all covered with pollen while moving in between the sporophylls as well as over the cone surface. At post-dehiscence, the abundance of all insect species declined and A. zamiae was absent.
Fig. 4.
Insect abundance in male cones of E. villosus at pre-dehiscence, dehiscence and post-dehiscence in: (A–D) Eastern Cape; (D–H) KwaZulu-Natal. Boxes within each insect group that share letters are not significantly different [Tukey's honest significant difference (HSD) test at P ≤ 0·05, n = 5]. Box = 25–75 %, whisker = Min–Max.
In EC populations, the three beetle species observed on dehiscent male cones (n = 5) showed similar patterns of abundance (Fig. 4B–D). The relative numbers of beetles were significantly higher in the afternoon than in the evening or morning for Erotylidae sp. nov. (Kruskal–Wallis χ2 = 13·52, P < 0·01), Porthetes sp. (Kruskal–Wallis, χ2 = 30·06, P < 0·001 and M. goodei (Kruskal–Wallis χ2 = 16·16, P < 0·001) (Fig. 4B–D). In post-dehiscent cones, there was a tendency for Erotylidae sp. nov. to be more abundant in the evening than in the morning or afternoon (Kruskal–Wallis, χ2 = 13·68; P < 0·001) (Fig. 4C).
The pattern of insect abundance was more variable on dehiscent male cones (n = 5) in KZN populations (Fig. 4E–H). At dehiscence, there were significantly more individuals of the uncommon A. zamiae (Kruskal–Wallis, χ2 = 6·67, P < 0·05) and the more abundant Porthetes sp. (Kruskal–Wallis, χ2 = 23·66, P < 0·001) during the afternoon than in the evening or morning. In contrast, M. goodei was significantly more abundant in the morning and evening than in the afternoon (Kruskal–Wallis, χ2 = 6·96, P < 0·05). The abundance of Erotylidae sp. nov. followed a similar pattern to that of M. goodei although there were no significant differences among times of day.
DISCUSSION
Results from this study show that the temperature of male cones follows strong daily cycles during the dehiscent stage when thermogenesis significantly increases cone temperature above ambient temperature. In contrast, there were only relatively small differences in the composition and emission rates of cone volatiles at different times of day. Instead, the composition and emission rates varied most markedly between different stages of cone development. Field observations of insect pollinators showed similarly large differences in abundance between developmental stages, with relatively smaller (but significant) differences in abundance on a daily cycle. These results are not fully consistent with the push–pull mechanism described previously for Macrozamia cycads (sensu Terry et al., 2007a, b), and deserve further discussion.
In considering the mechanisms that regulate insect pollinator behaviour, it is important to determine whether populations from EC and KZN behave differently. Both populations exhibit similar patterns of cone thermogenesis and they share the same pollinator species. The key differences occur in chemical composition between cone odours of E. villosus plants from EC and KZN populations, and the results of this study are in agreement with other studies on this species (e.g. Suinyuy et al., 2012). The odours from EC plants were dominated by eucalyptol and 2-isopropyl-3-methoxypyrazine (Supplementary Data Table S1), whereas odours from KZN populations were dominated by (3E)-1,3-octadiene and (3E,5Z)-1,3,5-octatriene (Supplementary Data Table S2). The occurrence of the same insects on cones in EC and KZN (see also Suinyuy et al., 2012) suggests that the insects respond to different chemicals in each region or they are responding to the same chemicals that occur in both regions.
The majority of the compounds were emitted at the dehiscent and receptive stages in male and female cones, respectively, when insects were active especially on male cones. Moreover, few of the compounds that occurred in the pollen dehiscent and receptive phases also occurred in the pre-dehiscent and post-dehiscent stages in male cones and the pre-receptive and post-receptive stages in female cones. The results suggest that these compounds play some role in mediating the behaviour of pollinators during the key period of pollen dehiscence and receptivity.
In general, emission of volatiles varies between the cone developmental stages, but not between different times of the day. In pollen-shedding cones of E. villosus, the daily pattern of cone volatile emissions indicated that there were no significant differences in the emission rates of the dominant compounds eucalyptol and 2-isopropyl-3-methoxypyrazine emitted by EC plants and (3E)-1,3-octadiene and (3E,5Z)-1,3,5-octatriene emitted by KZN plants. The only distinct daily pattern was evident in cone temperature, with low cone temperatures in the morning, high cone temperatures in the afternoon and peak cone temperatures in the evening. Despite the apparent uniformity in odour emission rates, there were differences in insect pollinator abundance on dehiscent cones at different times of day. The implication is that insects may be responding to daily patterns of cone heating, albeit on a less dramatic scale than that observed in Macrozamia spp. Close observations of dehiscing male cones of E. villosus at peak temperatures in the evening indicated that, although insects were generally less abundant than in the afternoon, few insects were leaving the male cones at this time. Relatively few beetles crawled out from between the sporophylls onto the surface of the cone before flying away. This differs from M. lucida and M. macleayi (pollinated by C. chadwickii thrips) and M. machinii (pollinated by Tranes weevils) in which there was a notable mass movement of pollinators away from male cones at the time of peak odour emission and thermogenesis (Terry et al., 2004). The daily pattern of insect movement on E. villosus therefore appears to be different from the very clear push–pull interactions observed in M. lucida and M. macleayi (Terry et al., 2004).
Several alternative patterns of attraction and repellence occur in cycads, including ones in which there is active attraction of pollinators at some stages of male cone development followed by passive dispersal associated with declining quality of the cone as a brood site (e.g. Donaldson, 2007). In this case, specific male cone attributes (volatile compounds and/or heat) that are considered as adaptations to facilitate pollination would be expected to influence only the attraction of pollinators but not their dispersal. A further option of no active attraction accompanied by active dispersal (Donaldson, 2007) was based on the assumption that pollinators would visit male cones as brood and feeding sites and that heat and volatiles would not be necessary to attract pollinators to male cones. Therefore, heat and volatiles that influence pollinator behaviour would be expected to have only repellent effects that would enhance dispersal of insect pollinators from male cones.
The developmental changes over the life cycle of male cones observed in this study indicate that there is a significant change in the emission rates of cone volatiles and cone thermogenesis at the onset of pollen dehiscence. Some of the developmental changes occurred in cone volatiles that comprised relatively low emission rates of the overall volatile profile. The changes in E. villosus cones were associated with a consistent increase in the abundance and activity of all three of the confirmed pollinator taxa on male cones (Fig. 4B–D, F–H). In contrast, activity by A. zamiae, which is predominantly a herbivore with a negligible role in pollination (Donaldson, 1997), was restricted to the period preceding pollen dehiscence (Fig. 4A, E). These results provide circumstantial evidence that male cone traits that are specific to the period of dehiscence do actively attract pollinators, in contradiction to the model where there is no active attraction (Donaldson, 2007).
Developmental variations in different floral volatiles, which result in the attraction of pollinator insects in one developmental stage and repellence in another stage, have been found in the orchid Ophrys sphegodes (Schiestl et al., 1997; Schiestl and Ayasse, 2001). The insect activity on male cones of E. villosus also showed a dramatic decline following dehiscence, which could indicate that the cone is producing repellent compounds at the stage following dehiscence or that high concentration of volatiles at post-dehiscence as in the EC plants repel pollinators. At this stage, most of the insects appeared to be second-generation adults that were emerging after undergoing larval development in the male cone sporophylls. The beetles were still covered in pollen and would therefore still be effective pollinators that will visit female cones by mistake because they produce attractive cues that are similar to those of male cones. However, by the time of emergence, the male cone had started to disintegrate and the sporophylls were decomposing so that the cone would have been unsuitable as a brood site. This is consistent with the possibility of dispersal associated with decline in the suitability of the cone as a brood site, without any specific adaptation to repel pollinators. A similar phenomenon seems to occur in basal angiosperms, where insects that visit the female flower parts during pollination stay there until the scent emission decreases and temperatures cool down, and they will then leave in search of another scented male or female flower part (e.g. Miyake and Yafuso, 2003; Seymour and Matthews, 2006).
The data for volatiles from E. villosus seem to best fit the model of active attraction at dehiscence followed by dispersal associated with deterioration of male cones after dehiscence, but this does not explain the daily changes in patterns of insect abundance observed on male cones. Because there was relatively little daily variation in the emission rates of cone volatiles, one implication is that thermogenesis influences insect behaviour on a daily basis, especially to disperse pollinators. The low abundance of pollinators observed on cones of E. villosus in the evenings (Fig. 4B–D, F–H) coincides with periods of peak temperatures in male cones of E. villosus. With the exception of Stangeria that does not produce heat (Tang, 1987a; Proches and Johnson, 2009), all cycad genera studied so far produce heat at some point in their developmental cycle. Cone volatile emissions and thermogenesis typically occur together in cycads (e.g. Tang, 1987a, 1993; Pellmyr et al., 1991; Stevenson et al., 1998; Seymour et al., 2004; Suinyuy et al., 2010), a phenomenon that also occurs in basal angiosperms (e.g. Seymour and Schultz-Motel, 1997, 1999; Jürgens et al., 2000; Ivancic et al., 2004; Seymour and Matthews, 2006; Thien et al., 2009). The biological role of thermogenesis in pollination of Encephalartos is still unclear. In M. machinii, the insects were highly active during thermogenesis compared with non-thermogenic periods, and this suggests that the heating is associated with the greater activity due to the higher body temperatures (Seymour et al., 2004). In other systems, thermogenesis augments scent production in plants to attract insects, functions as a reward for visiting insects and enhances larval development (Seymour and Schultz-Motel, 1997; Ervik and Barfod, 1999; Seymour and Shultz-Motel, 1999; Seymour et al., 2003, 2009). Some of the above roles may apply for the Encephalartos pollination system, but this study did not show any particular increase in volatiles associated with thermogenesis so Encephalartos may differ from other systems and represent a system where daily patterns of thermogenesis may result in small movements of pollinators on a daily basis compared with larger movements associated with developmental changes in cones.
In conclusion, our preliminary results, based on analysis of cone thermogenesis and cone volatiles together with observational data on pollinators, suggest that E. villosus have different patterns that regulate insect behaviour and is therefore not similar to the push–pull interaction that occurs in Macrozamia. Further experimental studies are needed to elucidate the response of insect pollinators to individual compounds in the laboratory followed by field tests to determine whether the response indicates attraction or repellence. This can be done by using varying amounts of whole cone scents or varying concentrations of individual compounds. Also studies are needed to understand the role of heat production alone in the behaviour of the insects that pollinate various Encephalartos species. Overall there is a need to carry out tests to determine whether cone odour and thermogenesis act in synergy or independently on pollinators of Encephalartos and other cycads.
SUPPLEMENTARY DATA
ACKNOWLEDGEMENTS
We thank Jacques De Wet Bösenberg and Natalie Uys for data collection, Andreas Jürgens for compound identification, Thimotheüs van der Niet for data analysis, and Sandy-Lynn Steenhuisen, Gail Van Aswegen, Rowena Stevens and Johanne Van Ryneveld for their support. We thank the horticulturist of the University of KwaZulu Natal (UKZN) Botanic Garden, Pietermaritzburg campus, and the caretakers of the cycad collection at the Kirstenbosch National Botanic Garden for their assistance. The Eastern Cape Parks Board and Ezemvelo KwaZulu Natal Wildlife issued permits to work on cycads. This study was funded by the Fairchild Tropical Botanical Garden, The Andrew W. Mellon Foundation, The South African National Biodiversity Institute (SANBI), The University of Cape Town and The University of KwaZulu Natal.
LITERATURE CITED
- Donaldson JS. Adaptation to the host and evolution of host specialisation in cycad weevils (Coleoptera: Brentidae) Cape Town, South Africa: University of Cape Town; 1991. PhD thesis. [Google Scholar]
- Donaldson JS. Is there floral parasite mutualism pollination in cycads? Pollination biology of Encephalartos villosus (Zamiaceae) American Journal of Botany. 1997;84:1398–1406. [Google Scholar]
- Donaldson JS. Hot and smelly sex! Theoretical considerations of the adaptive significance of cone thermogenesis and volatile odours in cycad reproduction. In: Vovides AP, Stevenson DW, Osborne R, editors. Proceedings of Cycad 2005. The 7th International Conference on Cycad Biology. New York: The New York Botanical Garden Press; 2007. pp. 308–325. [Google Scholar]
- Endrödy-Younga S. Boganiidae (Coleoptera: Cucujoidea) associated with cycads in South Africa: two new species and a new synonym. Annals of the Transvaal Museum. 1991;35:285–293. [Google Scholar]
- Ervik F, Barfod A. Thermogenesis in palm inflorescences and its ecological significance. Acta Botanica Venezuela. 1999;22:195–212. [Google Scholar]
- Gibernau M, Barabé D. Thermogenesis in three Philodendron species (Araceae) of French Guiana. Canadian Journal of Botany. 2000;78:685–689. [Google Scholar]
- Hall JA, Walter GH, Bergstrom DM, Machin P. Pollination ecology of the Australian cycad Lepidozamia peroffskyana (Zamiaceae) Australian Journal of Botany. 2004;52:333–343. [Google Scholar]
- Ivancic A, Lebot V, Roupsard O, Garcia JQ, Okpul T. Thermogenic flowering of taro (Colocasia esculenta, Araceae) Canadian Journal of Botany. 2004;82:1557–1565. [Google Scholar]
- Jürgens A, Webber A.C, Gottsberger G. Floral scent compounds of the Amazonian Annonaceae species pollinated by small beetles and thrips. Phytochemistry. 2000;55:551–558. doi: 10.1016/s0031-9422(00)00241-7. [DOI] [PubMed] [Google Scholar]
- Knudsen JT, Eriksson R, Gershenzon J, Ståhl B. Diversity and distribution of floral scent. Botanical Review. 2006;72:1–120. [Google Scholar]
- Kumano Y, Yamaoka R. Synchronization between temporal variation in heat generation, floral scent and pollinator arrival in beetle-pollinated tropical Araceae Homalomena propinqua. Plant Species Biology. 2006;3:173–183. [Google Scholar]
- Maia ACD, Schlindwein C. Caladium bicolor (Araceae) and Cyclocephala celata (Coleoptera, Dynastinae): a well-established pollination system in the Northern Atlantic Rainforest of Pernambuco, Brazil. Plant Biology. 2006;8:529–534. doi: 10.1055/s-2006-924045. [DOI] [PubMed] [Google Scholar]
- Miyake T, Yafuso M. Floral scents affect reproductive success in fly-pollinated Alocasia odora (Araceae) American Journal of Botany. 2003;90:370–376. doi: 10.3732/ajb.90.3.370. [DOI] [PubMed] [Google Scholar]
- Mound LA, Terry I. Thrips pollination of the Central Australian cycad, Macrozamia macdonnellii (Cycadales) International Journal of Plant Sciences. 2001;162:147–152. [Google Scholar]
- Mucina L, Geldenhuys CJ. Afrotemperate, subtropical and azonal forests. In: Mucina L, Rutherford MC, editors. The vegetation of South Africa, Swaziland and Lesotho. Pretoria: South African National Biodiversity Institute; 2006. pp. 584–615. Stretlizia 19. [Google Scholar]
- Norstog KJ, Stevenson DW, Niklas KJ. The role of beetles in the pollination of Zamia furfuracea L. fil. (Zamiaceae) Biotropica. 1986;18:300–306. [Google Scholar]
- Oberprieler RG. Bloemfontein: South Africa; 1996. Systematics and evolution of the tribe Amorphocerini (Coleoptera: Curculionidae), with a review of the cycad weevils of the world. PhD dissertation. University of The Orange Free State. [Google Scholar]
- Pearson HHW. Notes on South African cycads. Transactions of the South African Philosophical Society. 1906;16:341–354. [Google Scholar]
- Pellmyr O, Thien LB. Insect reproduction and floral fragrances: keys to the evolution of angiosperms. Taxon. 1986;35:76–85. [Google Scholar]
- Pellmyr O, Tang W, Groth I, Bergström G, Thien LB. Cycad cone and angiosperm floral volatiles: inferences for the evolution of insect pollination. Biochemical Systematics and Ecology. 1991;19:623–627. [Google Scholar]
- Proches S, Johnson SD. Beetle pollination of the fruit-scented cones of the South African cycad Stangeria eriopus. American Journal of Botany. 2009;9:1722–1730. doi: 10.3732/ajb.0800377. [DOI] [PubMed] [Google Scholar]
- Raguso RA. Why do flowers smell? The chemical ecology of fragrance-driven pollinator. In: Cardé RT, Millar JG, editors. Advances in insect chemical ecology. Cambridge: Cambridge University Press; 2004. pp. 141–178. [Google Scholar]
- Raguso RA. Wake up and smell the roses: the ecology and evolution of floral scent. Annual Review of Ecology, Evolution and Systematics. 2008;39:549–569. [Google Scholar]
- Rattray G. Notes on the pollination of some South African cycads. Transactions of the Royal Society of South Africa. 1913;3:259–270. [Google Scholar]
- Schiestl FP, Ayasse M. Post-pollination emission of a repellent compound in a sexually deceptive orchid: a new mechanism for maximizing reproductive success? Oecologia. 2001;126:531–534. doi: 10.1007/s004420000552. [DOI] [PubMed] [Google Scholar]
- Schiestl FP, Ayasse M, Paulus HF, Erdmann D, Francke W. Variation of floral scent emission and post pollination changes in individual flowers of Ophrys sphegodes subsp. sphegodes. Journal of Chemical Ecology. 1997;23:2881–2895. [Google Scholar]
- Seymour RS. Pattern of respiration by intact inflorescences of the thermogenic arum lily Philodendron selloum. Journal of Experimental Botany. 1999;50:845–852. [Google Scholar]
- Seymour RS, Baylock AJ. Stigma peroxidase activity in association with thermogenesis in Nelumbo nucifera. Aquatic Botany. 2000;67:155–159. [Google Scholar]
- Seymour RS, Matthews PDG. The role of themogenesis in the pollination biology of the Amazon waterlilly Victoria amazonica. Annals of Botany. 2006;98:1129–1135. doi: 10.1093/aob/mcl201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seymour RS, Schultze-Motel P. Heat producing flowers. Endeavour. 1997;21:125–129. [Google Scholar]
- Seymour RS, Schultz-Motel P. Respiration, temperature regulation and energetic of thermogenic inflorescences of the dragon lily Dracunculus vulgaris (Araceae) Proceedings of the Royal Society B: Biological Sciences. 1999;266:1975–1983. [Google Scholar]
- Seymour RS, White CR, Gibernau M. Heat reward for insect pollinators. Nature. 2003;426:243–244. doi: 10.1038/426243a. [DOI] [PubMed] [Google Scholar]
- Seymour RS, Terry I, Roemer RB. Respiration and thermogenesis by cones of the Australian cycad Macrozamia machinii. Functional Ecology. 2004;18:925–930. [Google Scholar]
- Seymour RS, Ito Y, Onda Y, Ito K. Effects of floral thermogenesis on pollen function in Asian skunk cabbage Symplocarpus renifolius. Biology Letters. 2009;5:568–570. doi: 10.1098/rsbl.2009.0064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SokaL RR, Rohlf FJ. Biometry: the principles and practice of statistics in biological research. 3rd edn. New York: WH Freeman; 1995. [Google Scholar]
- Stevenson DW, Norstog KJ, Fawcett PKS. Pollination biology of cycads. In: Owens SJ, Rudall PJ, editors. Reproductive biology. Kew: Royal Botanic Gardens; 1998. pp. 277–294. [Google Scholar]
- Suinyuy TN, Donaldson JS, Johnson SD. Insect pollination in the African cycad Encephalrtos friderici-guilielmi Lehm. South African Journal of Botany. 2009;75:682–688. [Google Scholar]
- Suinyuy TN, Donaldson JS, Johnson SD. Scent chemistry and patterns of thermogenesis in male and female cones of the African cycad Encephalartos natalensis (Zamiaceae) South African Journal of Botany. 2010;76:717–725. [Google Scholar]
- Suinyuy TN, Donaldson JS, Johnson SD. Geographical variation in cone volatile composition among populations of African cycad Encephalartos villosus. Biological Journal of the Linnean Society. 2012;106:514–527. [Google Scholar]
- Tang W. Heat production in cycad cones. Botanical Gazette. 1987a;148:165–174. [Google Scholar]
- Tang W. Insect pollination in the cycad Zamia pumila (Zamiaceae) American Journal of Botany. 1987b;74:90–99. [Google Scholar]
- Tang W. Heat and odour production in cycad cones and their role in insect pollination. In: Stevenson DW, Norstog KJ, editors. Proceedings of Cycad 90. The Second International Conference on Cycad Biology. Queensland: Palm and Cycad Societies of Australia; 1993. pp. 140–147. [Google Scholar]
- Terry I, Moore CJ, Walter GH, et al. Association of cone thermogenesis and volatiles with pollinator specificity in Macrozamia cycads. Plant Systematics and Evolution. 2004;243:233–247. [Google Scholar]
- Terry I, Walter GH, Donaldson JS, Snow E, Forster PI, Machin PJ. Pollination of Australian Macrozamia cycads: effectiveness and behaviour of specialist vectors in a dependent mutualism. American Journal of Botany. 2005;92:116–125. doi: 10.3732/ajb.92.6.931. [DOI] [PubMed] [Google Scholar]
- Terry I, Walter GH, Moore C, Roemer R, Hull C. Odour-mediated push–pull pollination in cycads. Science. 2007a;318:70. doi: 10.1126/science.1145147. [DOI] [PubMed] [Google Scholar]
- Terry I, Walter GH, Hull C, Moore C. Responses of pollinating thrips and weevils to specific Macrozamia cycad cone volatiles. In: Vovides AP, Stevenson DW, Osborne R, editors. Proceedings of Cycad 2005. The 7th International Conference on Cycad Biology. New York: The New York Botanical Garden Press; 2007b. pp. 346–371. [Google Scholar]
- Theis N, Lerdau M, Raguso RA. The challenge of attracting pollinators while evading floral herbivores: patterns of fragrance emission in Cirsium arvense and Cirsium repandaum (Asteraceae) International Journal of Plant Science. 2007;168:587–601. [Google Scholar]
- Thien LB, Azuma H, Kawano S. New perspectives on the pollination biology of basal angiosperms. International Journal of Plant Science. 2000;161:225–235. [Google Scholar]
- Thien LB, Bernhardt P, Devall MS, et al. Pollination biology of basal angiosperms (ANITA grade) American Journal of Botany. 2009;96:166–182. doi: 10.3732/ajb.0800016. [DOI] [PubMed] [Google Scholar]
- Zar JH. Biostatical analysis. 2nd edn. New Jersey: Prentice-Hall Inc; 1984. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.