Table 2.
Safety data for cisplatin and doxorubicin
| Parameter | Cisplatin | Doxorubicin |
|---|---|---|
| CAS-/EG no. | 15663-27-1/239-733-8 | 23214-92-8/245-495-6 |
| 25316-40-9/246-818-3 (hydrochloride) | ||
| Formula |
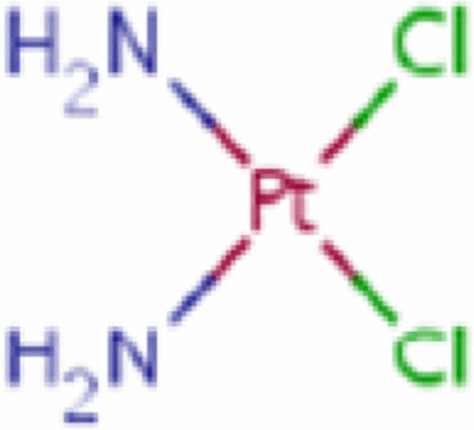
|
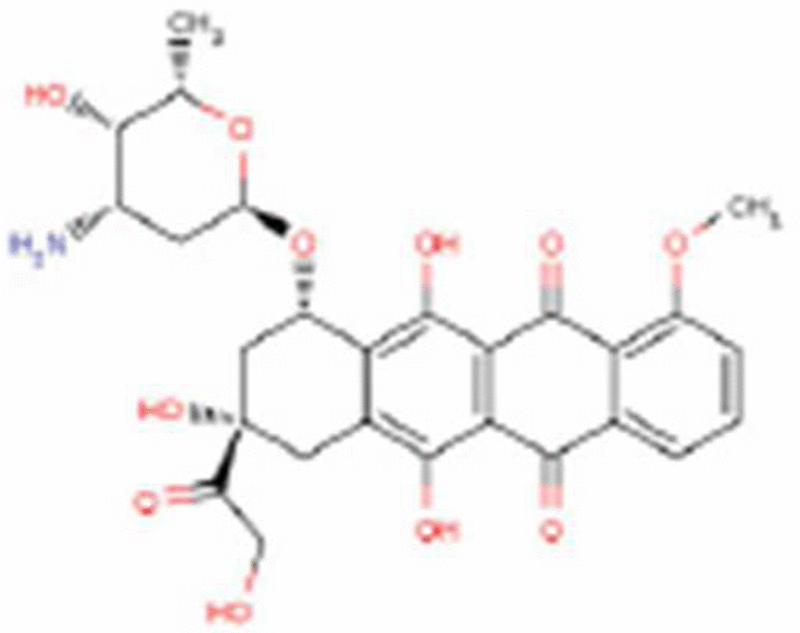
|
| Molecular weight | 300.06 g/mol | 543.52 g/mol |
| Melting point | 270 °C; dark yellow powder at room temperature | 205 °C (degradation); crystalline red powder at room temperature |
| Boiling point | Not applicable | Not applicable |
| Steam pressure | Not applicable | Not applicable |
| Water solubility | 2.530 g/L (25 °C) | 0.0928 g/L (25 °C) |
| LD50 oral | 20 mg/kg (rat) | 570 mg/kg (mouse) |
| 32 mg/kg (mouse) | – | |
| NOAEL | No data | No data |
| Important toxicological details |
Acute toxicity: very toxic Skin and eye irritation No evidence for transdermal absorption Cumulative damage of kidney, bone marrow, and inner ear No evidence of carcinogenicity in human Evidence for carcinogenicity and teratogenicity in mouse and rat Level of carcinogenicity: 2A Anaphylactic reactions reported Sensibilization of skin and airways |
Acute toxicity: harmful Dilatative cardiomyopathy Inflammation of mucosa Leucopenia Evidence for carcinogenicity in animals Evidence for mutagenicity in animals Level of carcinogenicity 2A |
| Total amount applied | 15 mg in 150 ml NaCl 0.9 % | 3 mg in 150 ml NaCl 0.9 % solution |
| Concentration of applied solution | 0.1 mg/ml = 0.1 g/L = 0.01 % | 0.02 mg/ml = 0.02 g/L = 0.002 % |
| Workplace exposure limits | Germany: not available | Germany no upper legal limit |
| Netherlands: 0.00005 mg/m3 | – |
