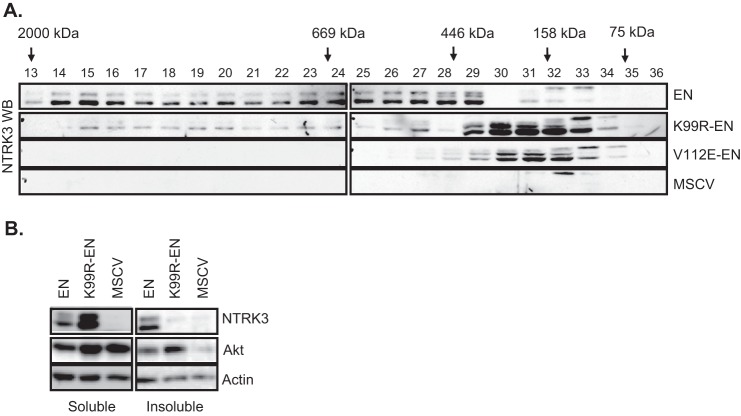
FIGURE 2.
Mutation of Lys-99 inhibits high molecular weight complex formation of EN. A, immunoblot analysis of the NIH3T3 cell lysates fractionated by FPLC is shown. Fraction numbers are indicated above each lane, and retention volumes of molecular mass markers are shown with arrows. EN was eluted in high molecular mass fractions, whereas both K99R-EN and the non-polymerizing mutant V112E-EN were detected in lower molecular mass fractions. Although the expected mass of monomeric EN is ∼73 kDa, its domain structure should result in a larger apparent size. B, immunoblot analysis of TritonX-100 soluble and insoluble fractions of NIH3T3 cell lysates is shown. Total Akt and β-actin antibodies were used as soluble fraction controls. EN proteins were probed with the NTRK3 antibody.