FIGURE 3.
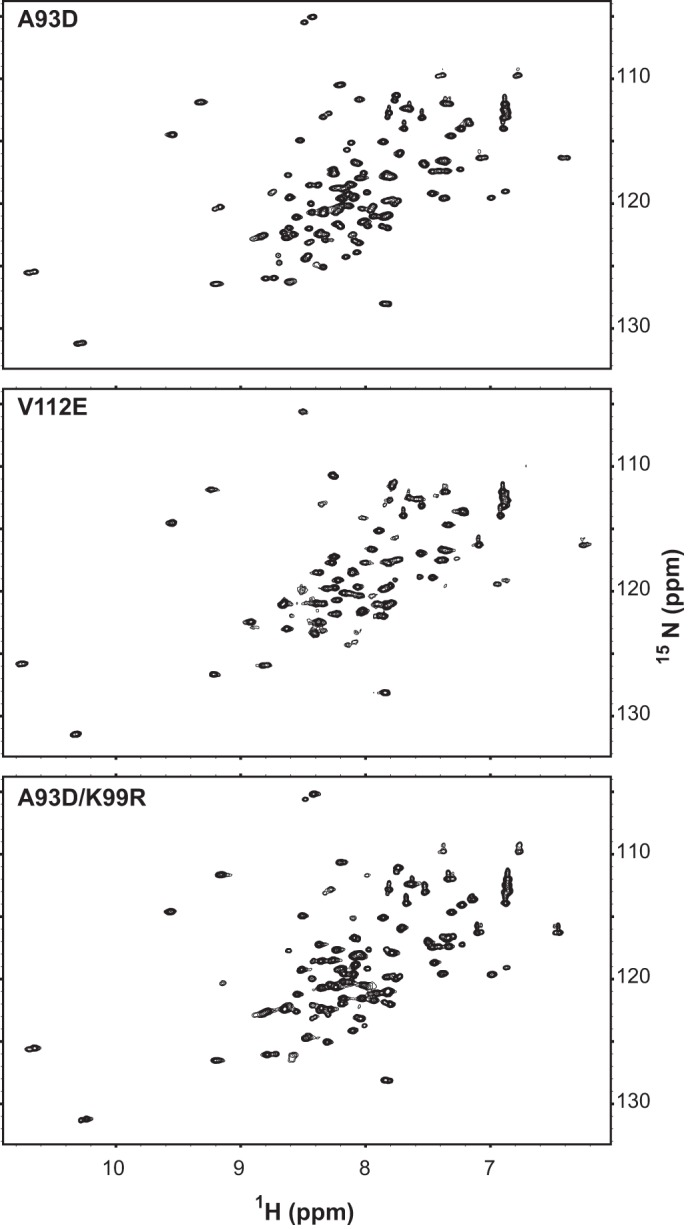
Mutation of Lys-99 does not disrupt the structure of the SAM domain. The well dispersed 15N HSQC (heteronuclear single quantum correlation) spectra of SAM-A93D, SAM-V112E, and SAM-A93D/K99R confirm that each ETV6 fragment (residues 43–125) adopts an independently folded structure. Thus, the reduction in EN self-association due to the K99R mutation does not arise indirectly from disruption of the SAM domain fold. Under these conditions, SAM-V112E showed some propensity to aggregate slightly, leading to a reduction in signal intensities. However, the relatively sharp spectral line widths indicate that each species is predominantly monomer. Based on partial main chain 15N and 13C resonances assignments, the ∼15 N-terminal residues preceding the helical SAM domain are conformationally disordered (not shown).
