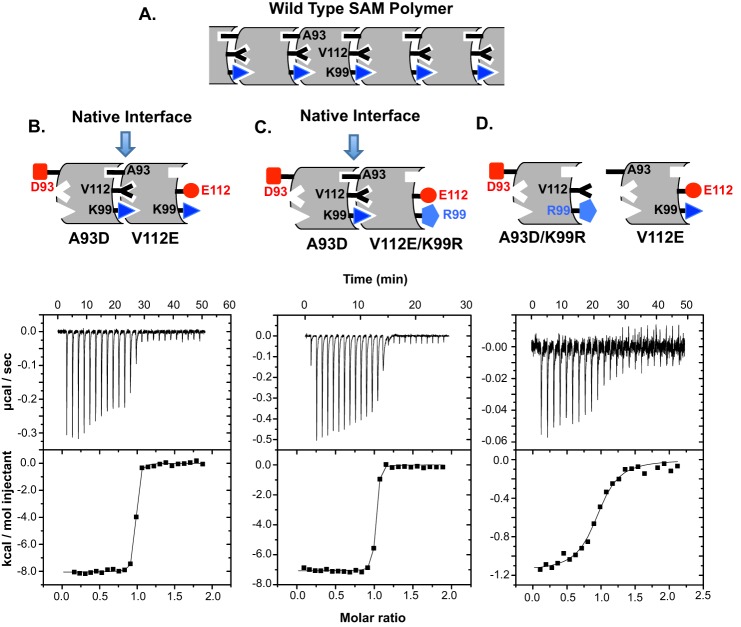
FIGURE 4.
Mutation of Lys-99 reduces the thermodynamic affinity of the ETV6-SAM domain for self-association. A, shown is an illustration of the WT SAM domain polymer structure of the locations of the interfacial residues in question. Shown are examples of ITC data for SAM-A93D titrated into SAM-V112E (B), SAM-A93D titrated into SAM-V112E/K99R (C), and SAM-A93D/K99R titrated into SAM-V112E (D). The raw data were buffer-blank-corrected (upper), integrated, and fit to a simple binding isotherm (lower) to yield the values reported in Table 1. The corresponding dimer structures of the isolated SAM domains with indicated mutations are depicted above each graph.