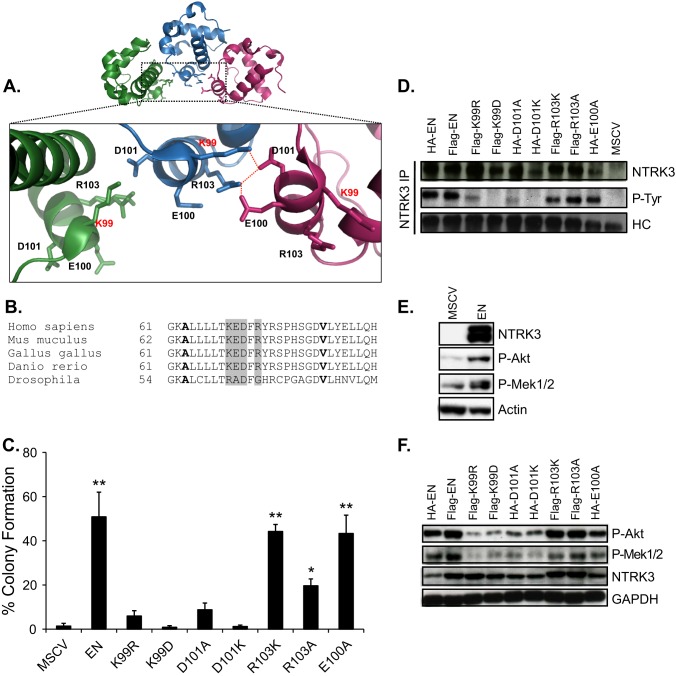
FIGURE 5.
Mutations of the critical residues involved in the salt bridge formation within the interaction face of SAM polymer abrogates EN transformation by inhibiting EN tyrosine phosphorylation and downstream signaling pathways. A, shown is a schematic illustration of a portion of the SAM domain polymer (PDB ID 1JI7) generated using PyMOL. The magnified inset shows a network of electrostatic interactions between Lys-99 and Arg-103 on one domain with Asp-101 and Glu-100 on the neighboring domain. B, shown is sequence alignment of the ETV6-SAM domain from different species. Alanine and the valine residues that form the hydrophobic core of the SAM polymer interface are shown in bold. The residues involved in the salt bridge were highlighted in gray. C, shown is soft agar colony formation assay. NIH3T3 cells stably expressing the indicated constructs were seeded in soft agar medium in triplicates and analyzed as described in Fig. 1A. A two-tailed Student's t test was used for statistical analysis. Each group was compared with MSCV control group. *, p < 0.05; **, p < 0.005. D, analysis of PTK activation status of the non-mutated and mutant EN proteins is shown. EN proteins were immunoprecipitated from the NIH3T3 cells expressing the indicated EN constructs or the MSCV control using the NTRK3 antibody, and EN tyrosine phosphorylation levels were determined by immunoblot analysis using the 4G10 phospho-tyrosine antibody. E, shown is a comparison of phospho-Akt and phospho-Mek1/2 levels in NIH3T3 cells expressing MSCV control vector or EN by immunoblot analysis after 36 h of serum starvation. F, shown is immunoblot analysis of the total cell lysates from the NIH3T3 cells expressing EN and mutant EN proteins. After 36 h of serum starvation, cells were lysed and analyzed by immunoblots. Akt and Mek1/2 activation was determined using the phospho-Akt and phospho-Mek1/2 antibodies. HC, heavy chain; P-Tyr, phosphotyrosine; P-Akt, phospho-Akt; P-Mek1/2, phospho-Mek1/2.