Abstract
The tumor suppressor genes MGMT and DAPK1 become methylated in several cancers including diffuse large B-cell lymphoma (DLBCL). However, allelic methylation patterns have not been investigated in DLBCL. We developed a fast and cost-efficient method for the analysis of allelic methylation based on pyrosequencing of methylation specific PCR (MSP) products including a SNP. Allelic methylation patterns were reliably analyzed in standards of known allelic methylation status even when diluted in unmethylated DNA to below 1% methylation. When studying 148 DLBCL patients MGMT and DAPK1 methylation was observed in 19% and 89%, respectively, and among methylated and heterozygous patients 29% and 55%, respectively, were biallelically methylated. An association between the T-allele of the rs16906252 SNP and MGMT methylation was observed (p-value = 0.04), and DAPK1 methylation of the A-allele was associated with shorter overall survival (p-value = 0.006). In future cancer research allelic MSP-pyrosequencing may be used to study a wide range of other loci.
Diffuse Large B-Cell lymphoma (DLBCL) is the most common type of non-Hodgkin's lymphoma in adults. However, DLBCL represent a heterogeneous group of lymphomas, with different sensitivity to the standard rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) treatment.
The MGMT gene is implicated in several human malignancies including DLBCL due to its function as a DNA repair gene. Specifically, the MGMT protein removes alkyl adducts from the O6 position of guanine thereby preventing G > A mutations in the genome. When MGMT is inactivated or lost this may contribute to tumor development and progression, however, it may also result in increased sensitivity to alkylating agents. It has been shown that MGMT promoter methylation, as assessed by methylation specific PCR (MSP), predicts response to the alkylating agent temozolomide in glioblastoma patients1,2. In DLBCL, early studies suggest MGMT promoter methylation as a useful marker for predicting survival in patients treated with multidrug regimens including the alkylating agent cyclophosphamide3,4. However, these results were obtained before the introduction of rituximab, a monoclonal antibody against CD20, which is primarily found on the surface of B cells. A more recent study of 53 DLBCL patients who received R-CHOP showed that inactivation of MGMT does not appear to play a role as a marker of response to this treatment5.
A functional role of MGMT methylation in DLBCL has been confirmed several times at the RNA and protein levels3,5,6, and in other cancers a strong association between the T-allele of the rs16906252 MGMT promoter SNP and MGMT promoter methylation has been observed7,8,9,10. The T-allele has been shown to confer an approximately 30% reduction in MGMT promoter activity compared to the wild-type haplotype using a luciferase reporter assay11. This reduced activity may, in turn, predispose the allele to become methylated. Interestingly, it has also been shown that the presence of a T-allele may result in longer overall survival in glioblastoma patients treated with temozolomide and that patients carrying both the T-allele and MGMT methylation survived longer compared to patients having either the T-allele or MGMT methylation11.
DAPK1 is a pro-apoptotic gene, which undergo methylation mediated silencing in several cancers12,13, including DLBCL, where hypermethylation of its promoter has been shown to be an independent prognostic factor in predicting shortened overall survival14.
Most studies of MGMT and DAPK1 methylation have been performed using traditional MSP. However, traditional MSP assays do not provide information on the methylation status of individual alleles. Furthermore, MSP may be prone to false-positive results caused by amplification of unmethylated or incompletely bisulfite converted molecules15,16,17. Allelic methylation information is traditionally obtained by bisulfite sequencing of single clones, however, this method is labor-intensive, expensive, and may not be suitable for use in clinical settings18.
We have developed a novel method for allelic methylation analysis based on pyrosequencing of MSP products including a SNP. Non-CpG cytosines and CpG sites are also interrogated by the assays as a control for the amplification of fully converted and methylated templates, respectively. Initially, the analytical sensitivity of the assays and their ability to distinguish different allelic methylation states were investigated using standards of known allelic methylation status. Then 148 samples from DLBCL patients were genotyped for the SNPs used to provide allelic methylation information and tested using the allelic MSP-pyrosequencing assays. It was investigated if MGMT methylation was associated with the T-allele of the rs16906252 SNP, and if any of the DAPK1 alleles were associated with methylation. Finally, potential correlations of MGMT or DAPK1 methylation and MGMT or DAPK1 SNP genotypes with clinical parameters were investigated.
Results
The allelic MSP-pyrosequencing assays can resolve the methylation status of individual alleles
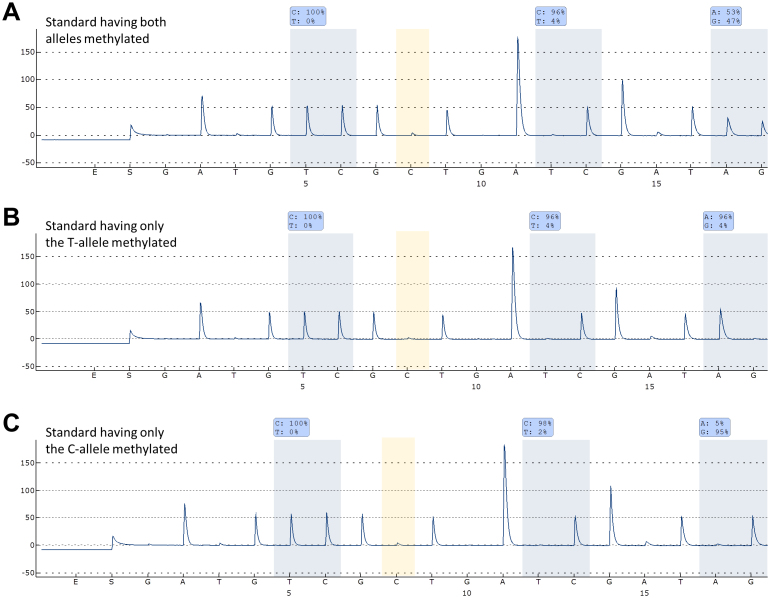
Standards of known allelic methylation status were tested using the allelic MGMT and DAPK1 MSP-pyrosequencing assays. When using the MGMT assay a strong signal from both alleles was observed for the standard having both alleles methylated (53% T, 47% C). For the standard having only the T-allele methylated a strong signal was only observed from the T-allele (96% T, 4% C), and for the standard having only the C-allele methylated a strong signal was only observed from the C-allele (5% T, 95% C) (Fig. 1). When using the DAPK1 assay a strong signal from both alleles was observed for the standard having both alleles methylated (45% A, 55% G). For the standard having only the A-allele methylated a signal was only observed from the A-allele (100% A, 0% G), and for the standard having only the G-allele methylated a signal was only observed from the G-allele (0% A, 100% G) (data not shown).
Figure 1. The MGMT allelic MSP-pyrosequencing assay.
The non-CpG C position analyzed is highlighted in yellow. The antisense strand was analyzed. (A) Allelic MSP-pyrosequencing on a standard having both alleles methylated. (B) Allelic MSP-pyrosequencing on a standard having only the T-allele methylated. (C) Allelic MSP-pyrosequencing on a standard having only the C-allele methylated.
Sensitivity of the allelic MSP-pyrosequencing assays
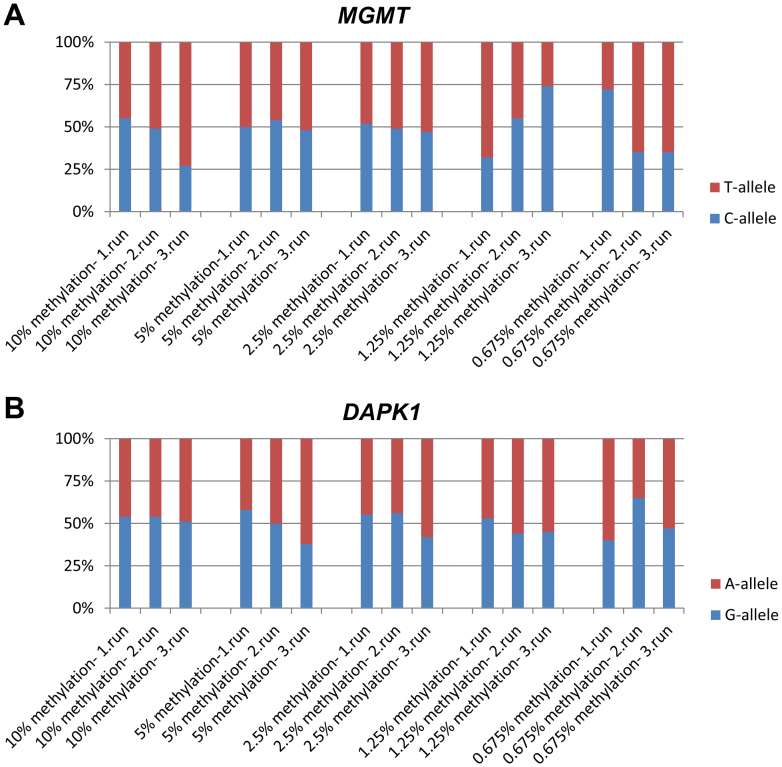
The biallelically methylated standards were diluted in unmethylated DNA to 10%, 2.5%, 1.25%, and 0.625% methylation and analyzed using the allelic MGMT and DAPK1 MSP-pyrosequencing assays. Each experiment was repeated three times. For all of the standards a band of the expected size could be observed on an agarose gel and all were successfully pyrosequenced. For both assays a signal of at least 27% from one of the alleles could be observed for all dilutions (Fig. 2). Thus, the assays can be expected to be able to resolve the methylation status of individual alleles even in samples containing low-level methylation.
Figure 2. The sensitivity of the allelic MSP-pyrosequencing assays.
The standards having both alleles methylated were serially diluted into unmethylated DNA and tested using the allelic MSP-pyrosequencing assays. The experiments were repeated three times for each of the assays. It can be observed that a signal from both alleles above 25% can be detected down to 0.675% methylation. For MGMT the antisense strand was analyzed. (A) The MGMT assay (B) The DAPK assay.
Genotyping the rs16906252 MGMT and the rs13300553 DAPK1 SNPs in DLBCL samples
For the MGMT SNP 129 of the patients were homozygous for the C-allele, one patient was homozygous for the T-allele, and 18 patients were heterozygous (data not shown). The frequency of the T-allele was 7% and 12% carried the T-allele, conforming to Hardy–Weinberg equilibrium (p-value = 0.67). This is close to what has previously been reported in a study using healthy individuals19. For the DAPK1 SNP 48 of the patients were homozygous for the A-allele, 28 patients were homozygous for the G-allele, and 67 patients were heterozygous (data not shown). For five patients no data was obtained. The frequency of the A-allele was 57% and 81% carried the A-allele, conforming to Hardy–Weinberg equilibrium (p-value = 0.60). The results have been summarized in Supplementary Table S1.
MGMT methylation analysis of DLBCL samples using allelic MSP-pyrosequencing
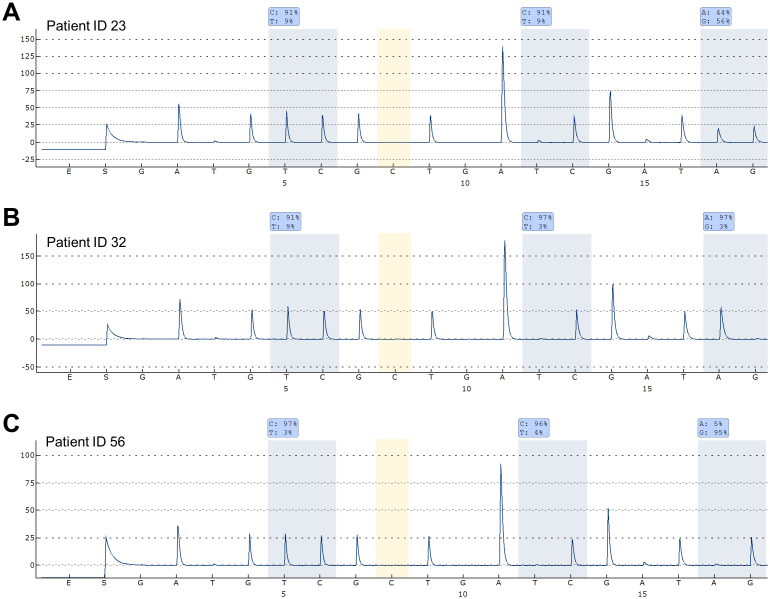
Thirty-two of the 148 samples analyzed were methylation positive as assessed by gel electrophoresis of the MSP products. Twenty-eight of these samples were confirmed to be methylated by pyrosequencing. The two CpG sites (serving as a control for the amplification of methylated molecules) analyzed by pyrosequencing were both methylated above 90% in all samples, except for the sample from patient ID 78. This sample was heterogeneously methylated (one site methylated at 96% and the other at 38%). Seven of the 28 methylated samples were heterozygous. Two of these were methylated at both alleles (patient ID 23 and 39), four were methylated only at the T-allele (patient ID 32, 88, 96, and 135) (defined as C-allele below 20%), and one was methylated only at the C-allele (patient ID 56) (defined as T-allele below 20%). The results for three samples having different allelic MGMT methylation patterns are shown in Fig. 3. The frequency of methylated samples was 22% by gel electrophoresis and 19% by pyrosequencing. All 148 samples amplified using primers specific for unmethylated DNA and a band of the expected size could be observed on the gel (data not shown). A significant association between MGMT methylation and the T-allele of the rs16906252 SNP was observed (p-value = 0.04). Methylation frequencies were similar between FFPE samples and fresh frozen samples (p-value = 0.80). The results have been summarized in Supplementary Table S1. Bisulfite conversion and allelic MSP-pyrosequencing were repeated for three samples having different allelic MGMT methylation patterns (patient ID 23, 32, and 56). The same results were observed as for the first analysis (data not shown).
Figure 3. Allelic MGMT methylation analysis in DLBCL samples.
The non-CpG C position analyzed is highlighted in yellow. The antisense strand was analyzed. (A) Patient ID 23 was methylated at both alleles. (B) Patient ID 32 was methylated at the T-allele. (C) Patient ID 56 was methylated at the C-allele.
Confirmation of the allelic MGMT methylation results
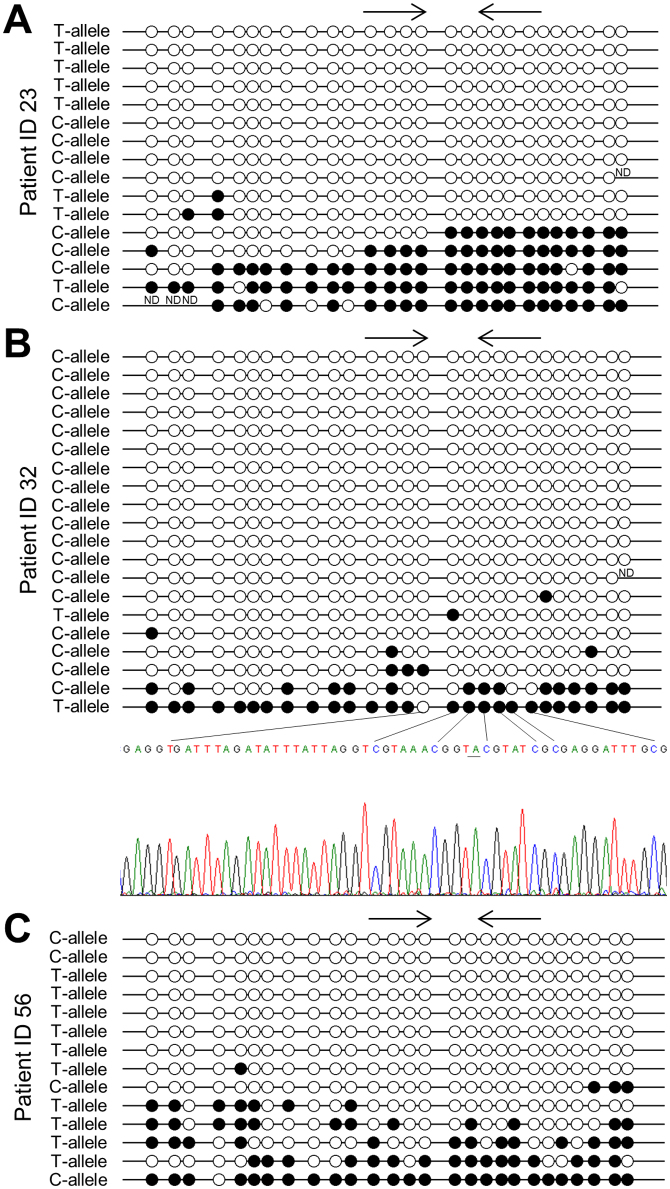
Methylation independent PCR (MIP) pyrosequencing was used to investigate the methylation levels of six CpG sites surrounding the rs16906252 SNP in the five heterozygous fresh frozen samples showing methylation by allelic MSP-pyrosequencing. The average methylation levels for the two samples having both alleles methylated (patient ID 23 and 39) and the three samples having only one allele methylated (patient ID 32, 56, and 88) were 26.5% and 23.7%, and 15.2%, 18%, and 3.7%, respectively. However, a PCR bias towards amplification of the unmethylated allele was observed by sequencing a standard of 50% methylated DNA as the average methylation level of this sample was 29%. Examples are shown in Supplementary Fig. S1. Bisulfite sequencing of single clones was performed for three samples having different allelic MGMT methylation patterns (patient ID 23, 32, and 56). Heavily methylated C- and T-alleles were observed for patient ID 23. For patient ID 32 most of the sequenced alleles were unmethylated C-alleles. Given the PCR bias towards amplification of unmethylated DNA this was expected. One heavily methylated T-allele and a heterogeneously methylated C-allele were also observed for this patient. For patient ID 56 most of the sequenced alleles were unmethylated T-alleles, again as expected. One heavily methylated C-allele and a few heterogeneously methylated T-alleles were also observed for this patient (Fig. 4). Thus, the data from allelic MSP-pyrosequencing and bisulfite sequencing of single clones correlated well, and it was confirmed that the allelic MSP-pyrosequencing assay is stringent, as heterogeneously methylated molecules are not amplified.
Figure 4. Bisulfite sequencing of single clones for patient ID 23, 32, and 56.
ND is no data (A) For patient ID 23, heavily methylated C- and T-alleles could be observed. (B) For patient ID 32, most of the clones successfully sequenced were unmethylated C-alleles, while one heavily methylated T-allele was observed. Part of the sequencing trace for this allele is shown, and the SNP position is underlined. (C) For patient ID 56, most of the sequenced clones were unmethylated T-alleles, while one heavily methylated C-allele was observed.
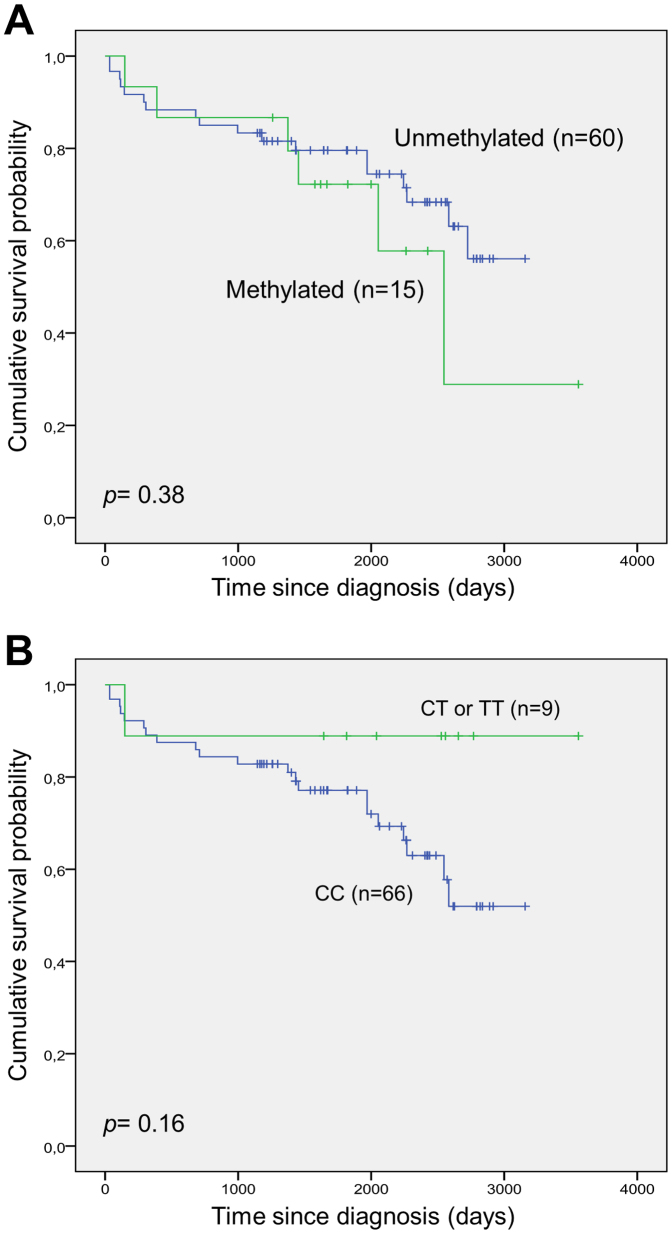
MGMT methylation according to patient characteristics among R-CHOP treated patients
The clinical characteristics of R-CHOP treated patients as a function of MGMT methylation status are shown in Table 1. We found no significant differences in patient or disease characteristics according to MGMT methylation status. Also, according to MGMT methylation status no significant difference in the response rate was observed. Survival analysis according to MGMT methylation status did not reveal any significant differences when evaluating overall survival (Fig. 5A). The same was true for survival analysis according to presence of the T-allele of the rs16906252 SNP (Fig. 5B).
Table 1. Clinical characteristics of R-CHOP treated patients as a function of MGMT methylation status.
| Total (n = 75) | Unmethylated (n = 60) | Methylated (n = 15) | p-value | |
|---|---|---|---|---|
| Sex | 0.91 | |||
| Men | 39 | 31 | 8 | |
| Women | 36 | 29 | 7 | |
| Extranodal involvement | 0.80 | |||
| Yes | 32 | 25 | 7 | |
| No | 41 | 33 | 8 | |
| Missing | 2 | 2 | 0 | |
| Stage | 0.49 | |||
| I-II | 36 | 30 | 6 | |
| III-IV | 39 | 30 | 9 | |
| Elevated LDH | 0.16 | |||
| Yes | 33 | 24 | 9 | |
| No | 42 | 36 | 6 | |
| B symptoms | 0.12 | |||
| Yes | 31 | 22 | 9 | |
| No | 40 | 36 | 6 | |
| Missing | 2 | 2 | 0 | |
| IPI score | 0.41 | |||
| 0–2 | 54 | 44 | 10 | |
| 3–5 | 21 | 16 | 5 | |
| Performance score* | 0.35 | |||
| Good | 69 | 56 | 13 | |
| Bad | 6 | 4 | 2 | |
| Response | 0.097 | |||
| CR/Cru | 62 | 51 | 11 | |
| PD/PR | 6 | 3 | 3 | |
| Missing/Mors | 7 | 6 | 1 | |
| Age, mean (range) | 60.8 (22–86) | 60.6 years (22–83) | 61.5 years (27–86) | 0.84 |
*Eastern Cooperative Oncology Group.
LDH, lactate dehydrogenase; IPI, international prognostic index; CR, complete response, unconfirmed complete response; PD, progressive disease; PR, partial response.
Figure 5. Overall survival after frontline R-CHOP treatment according to MGMT methylation status and rs16906252 genotypes.
(A) Overall survival according to MGMT methylation status. (B) Overall survival according to rs16906252 genotype.
DAPK1 methylation analysis of DLBCL samples using allelic MSP-pyrosequencing
Of the 146 samples analyzed 130 were methylation positive as assessed by gel electrophoresis of the MSP products. Of these 127 were confirmed to be methylated by pyrosequencing. Three samples showed failed bisulfite conversion and were scored as uncertain methylation status (data not shown). The three CpG sites analyzed by pyrosequencing were on average methylated above 85% in the majority of the samples. Heterogeneous methylation patterns (defined as less than 85% average methylation) were observed for the samples from patient ID 20 (80%, 74%, 96%), patient ID 92 (64%, 63%, 97%), patient ID 95 (49%, 97%, 98%), patient ID 109 (51%, 97%, 98%), patient ID 123 (99%, 13%, 99%), patient ID 128 (98%, 98%, 14%), patient ID 138 (96%, 41%, 97%), and patient ID 146 (96%, 41%, 97%). Fifty-eight of the 130 methylated samples were heterozygous. Thirty-two of these were methylated at both alleles, 15 were methylated only at the A-allele (defined as G-allele below 20%), and 11 were methylated only at the G-allele (defined as A-allele below 30%). The results for three representative samples having different allelic DAPK1 methylation patterns are shown in Supplementary Fig. S2. The frequency of methylated samples was 89.0% by gel electrophoresis and 88.8% by pyrosequencing. All unmethylated samples amplified using primers specific for unmethylated DNA and a band of the expected size could be observed on an agarose gel (data not shown). No significant associations between DAPK1 methylation and genotypes of the rs13300553 SNP were observed. Methylation frequencies were similar between FFPE samples and fresh frozen samples (p-value = 0.88). The results have been summarized in Supplementary Table S1.
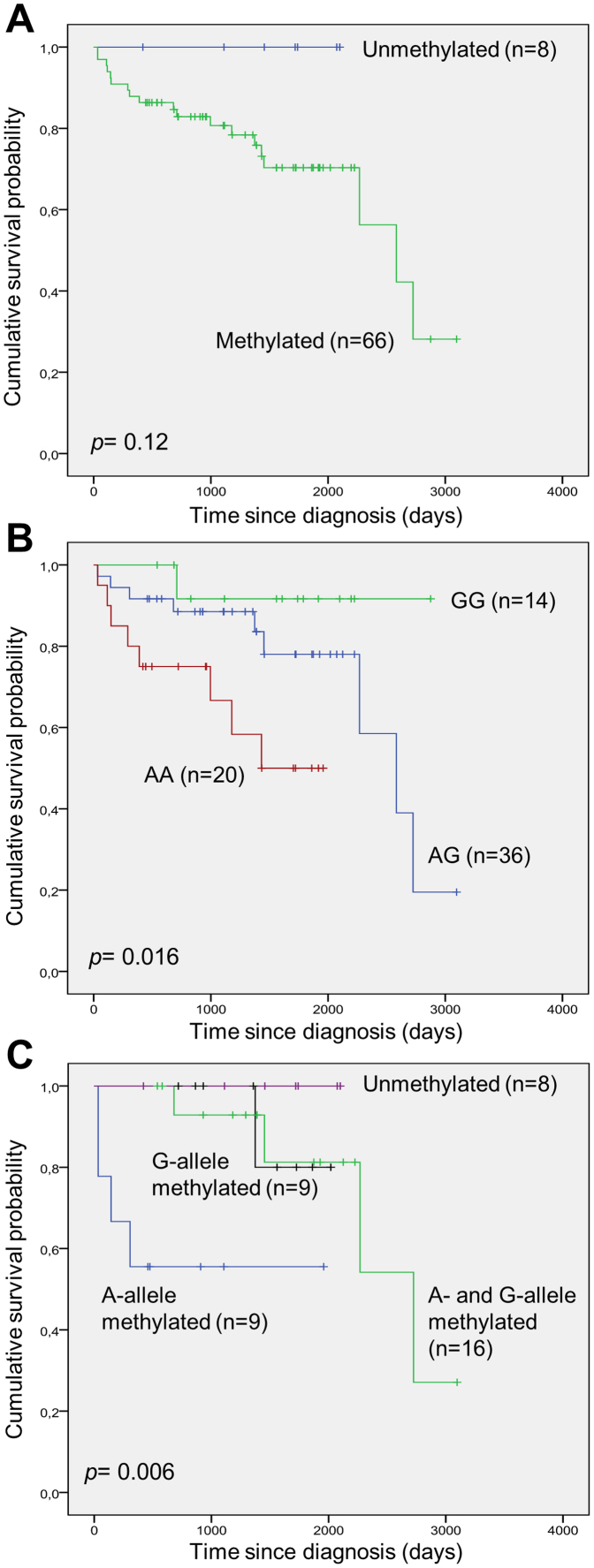
DAPK1 methylation according to patient characteristics among R-CHOP treated patients
Clinical characteristics of R-CHOP treated patients as a function of DAPK1 methylation status were investigated similar to what was done as a function of MGMT methylation shown in Table 1. No significant difference in patient or disease characteristics according to DAPK1 methylation status was observed (data not shown). Also, according to DAPK1 methylation status no significant difference in the response rate was observed. Survival analysis according to DAPK1 methylation status did not reveal any significant differences when evaluating overall survival, however, a tendency towards longer survival of patients without DAPK1 methylation could be observed (Fig. 6A). For survival analysis according to the genotypes of the rs13300553 SNP a significantly shorter survival was associated with the A-allele (Fig. 6B). Among the heterozygous individuals of the rs13300553 SNP survival analysis according to allelic methylation revealed a significantly shorter survival associated with methylation of the A-allele (Fig. 6C).
Figure 6. Overall survival after frontline R-CHOP treatment according to DAPK1 methylation status, rs13300553 genotypes, and allelic DAPK methylation patterns.
(A) Overall survival according to DAPK1 methylation. (B) Overall survival according to rs13300553 genotypes. (C) Overall survival according to allelic DAPK1 methylation patterns in individuals heterozygous for rs13300553.
Methylation analysis of DLBCL cell lines
MGMT and DAPK1 methylation analyses were performed for seven DLBCL cell lines: DB, DOHH2, Farage, HT, Pfeiffer, RL, and Toledo. For MGMT only the DOHH2 and Toledo cell lines were methylated. Both of these cell lines were homozygous for the C-allele of the rs16906252 SNP. For DAPK1 all seven cell lines were methylated. Farage, DB, DOHH2, and Toledo were heterozygous for the rs13300553 SNP, and these were all methylated at both alleles (data not shown).
Discussion
We developed a fast and reliable method for allelic methylation analysis at single loci based on MSP followed by pyrosequencing. Compared to bisulfite sequencing of single clones this method does not suffer from PCR bias and cloning bias, and is fast and cost-effective. While bisulfite sequencing of single clones takes several days to perform, allelic MSP-pyrosequencing can be performed in less than four hours. The costs associated with cloning and sequencing of several clones per sample are at least 10 fold higher compared to allelic MSP-pyrosequencing, which only requires a single sequencing reaction. Allelic MSP-pyrosequencing also has the advantage of a higher analytical sensitivity. We have shown that allelic methylation patterns can be reliably analyzed in samples methylated at levels below 1% when using 25 ng input DNA. To obtain a similar analytical sensitivity using bisulfite sequencing would require sequencing hundreds of clones.
Compared to conventional MSP20 (which does not provide allelic methylation information) and allelic SMART-MSP8,19, allelic MSP-pyrosequencing has the advantage of analyzing additional CpG sites and non-CpG cytosines as a control for the amplification of fully methylated and bisulfite-converted molecules, respectively.
MIP followed by pyrosequencing using allele-specific sequencing primers incorporating a SNP in the 3′ end to discriminate the alleles has also been used to analyze allelic methylation patterns21. This method requires four independent pyrosequencing reactions, one for each genotype and two negative controls using primers that do not recognize either allele and, in general, methods using MIP primers have a low analytical sensitivity compared to MSP based methods and may potentially be compromised by PCR bias22,23.
Traditional MIP-based assays, such as combined bisulfite restriction analysis (COBRA)24 and Melting curve analysis (MCA)25, measure the percentage of methylation in a sample, which is often a mixture of malignant and non-malignant cells likely to show different methylation patterns. Therefore, a measurement of about 50% methylation using a MIP based method could mean that all cells in the sample were methylated at only one allele, or that half of the cells were methylated at both alleles. As the fraction of tumor cells in a sample is normally difficult to determine with high accuracy26, and intra-tumor heterogeneity with respect to methylation patterns cannot be ruled out, traditional MIP-based assays cannot provide allelic methylation information.
Alternatively, deep sequencing27 or methylation analysis on SNP arrays28 may be used to obtain allelic methylation information, however, these approaches require specialized equipment and complicated bioinformatic analyses and may not be recommendable for analyzing single loci.
Using allelic MSP-pyrosequencing we analyzed the methylation status of the MGMT gene in 148 DLBCL patients. We detected methylation in 19% of the patient samples, and the amplified molecules appeared to be homogeneously methylated in all samples, as both of the CpG sites analyzed by pyrosequencing was above 90%, with the exception of one sample for which one site showed a significantly lower methylation level. In addition, the bisulfite conversion status as assessed by pyrosequencing was excellent for all methylation positive samples, of which seven were heterozygous for the rs16906252 SNP. Two of these were shown to be methylated at both alleles, four were methylated only at the T-allele, and one was methylated only at the C-allele. When analyzing these samples by MIP pyrosequencing the methylation levels were found to be higher for the two samples showing bi-allelic methylation as could be expected and allelic methylation patterns were confirmed by bisulfite sequencing of single clones, which also revealed some heterogeneously methylated molecules not amplifiable by the MSP primers.
Most previous studies of MGMT methylation in DLBCL have been performed using conventional MSP3,5,29,30. These studies found slightly higher frequencies of methylation positive patients compared to our study. This may, in part, be explained by slight differences in primer design, and the analytical sensitivity of each particular assay. It is also possible that our assay is more stringent as we used an annealing temperature of 70°C. The use of less stringent PCR conditions may result in amplification of heterogeneously methylated molecules15. The frequency of methylated DLBCL samples may also be influenced by the percentage of tumor cells in the samples and contamination with peripheral blood cells, which have been shown to be methylated at low levels in healthy individuals19. However, the samples analyzed in the present study contained at least 50% tumor cells.
An association between MGMT methylation and the T-allele of the rs16906252 SNP was observed in this patient cohort. In comparison, several previous studies have found more statistically significant associations between MGMT methylation and the T-allele of the rs16906252 SNP7,8,9,10,19. However, these studies were not of DLBCL, and it cannot be excluded that the effect of the T-allele on MGMT methylation propensity is cell-type specific.
We did not observe any significant differences in overall survival according to MGMT methylation status or genotypes of the rs16906252 SNP among the R-CHOP treated patients. This was expected as a recent study of a R-CHOP treated cohort of DLBCL patients found no difference in response or survival according to MGMT methylation status5. We speculate that the negative effects of having a silenced MGMT tumor suppressor gene in these patients are larger than the positive effects of not being able to repair the DNA damage introduced by the treatment, which consists of several other classes of chemotherapeutica than the alkylating agent, cyclophosphamide. To this end, it may be interesting to compare whole-genome or exome sequencing data to MGMT methylation data in a large patient cohort, though an association between MGMT methylation and G > A mutations may not be observed for single genes in DLBCL, such as TP5330.
In future projects it may also be interesting to investigate if monoallelic methylation of MGMT results in complete silencing or a reduced expression of the protein, as we found more cases of monoallelic than biallelic methylation in the present study. If patients with detectable MGMT methylation only have one allele methylated and still express the protein this may have implications for its use as a predictive biomarker in other diseases such as glioblastoma multiforme, as such patients may not respond or require different drug doses.
Methylation analysis of the DAPK1 promoter was successfully performed for 146 of the DLBCL samples using allelic MSP-pyrosequencing. Methylation was detected in 89% of the patient samples, and the amplified molecules appeared to be homogeneously methylated in all samples, except eight, where the average methylation level of the three CpG sites was below 85%. These were interpreted as being heterogeneously methylated. In addition, three samples showed failed bisulfite conversion status as assessed by pyrosequencing. These could potentially be false positives and were scored as uncertain methylation status. Among the methylated samples 58 were heterozygous, and biallelic methylation was observed for 55% of these.
Other previous studies of DAPK1 methylation in DLBCL also found high methylation frequencies of DAPK1 promoter methylation31,32,33, and DAPK1 methylation at low levels has been found in B cells from normal individuals34. Though not statistically significant (p-value = 0.12), a tendency towards a shortened overall survival among patients with DAPK1 methylation was observed, similar to what has previously been published14. Interestingly, we found a relatively high fraction of the heterozygous samples to be methylated at only one allele, and we observed a statistically significant correlation between methylation of the A-allele and overall survival (p-value = 0.006). Likewise, carrying the A-allele was associated with shortened overall survival (p-value = 0.016). While these data are preliminary, and should be interpreted with caution, it could be interesting to further investigate a possible role of allelic DAPK1 methylation patterns in the pathogenesis of DLBCL.
A current limitation of allelic MSP-pyrosequencing is that is does not measure the actual amount of methylation in the promoter. However, the use of a real-time PCR instrument instead of a traditional thermocycler would allow quantitative methylation information to be obtained35,36.
In conclusion, we have developed a novel MSP-based pyrosequencing assay, which is capable of resolving the methylation status of individual alleles. The assays presented here were designed to analyze the methylation status of the MGMT and DAPK1 genes using the rs16906252 and rs13300553 SNPs, respectively, for the allelic analysis. Many other frequently methylated tumor suppressor genes, such as APC, BRCA1, CDH1, CDH13, HIC1, hMLH1, RUNX3, and SOCS1 harbor polymorphic SNPs in their promoter CpG islands, which will allow allelic methylation analyses to be performed. Thus, allelic MSP-pyrosequencing may be valuable for the analysis of DNA methylation of many different tumor suppressor genes in different malignancies.
Methods
Patient samples and DNA extraction
Fresh frozen lymphoma biopsies were obtained from 93 cases and Formalin-Fixed Paraffin Embedded (FFPE) lymphoma biopsies were obtained from 55 cases diagnosed with DLBCL. The diagnoses were based on standard histology and immunphenotyping according to the 2008 WHO lymphoma classification using anti-CD20, -CD79alpha, CD43, and -BCL2. The fraction of tumor cells was more than 50% of the total tissue for all samples. All patients were treated with antracycline containing regimens, however, only 75 patients received immunotherapy with rituximab.
For the fresh frozen samples genomic DNA was isolated after proteinase K digestion using the Purescript DNA Isolation Kit (Gentra Systems, Minneapolis, MN, USA). For the FFPE samples five to seven sections of 10 μm were cut from the FFPE blocks and deparaffinization was performed using xylene followed by two rounds of ethanol treatment. DNA was extracted using the QIAamp DNA Mini Kit (Qiagen, Hilden, Germany).
DNA from peripheral blood (PB), obtained from healthy donors, was extracted following a modified salt precipitation protocol as previously described37. Written informed consent was obtained from all subjects.
The project has been approved by the regional ethics committees (De Videnskabsetiske Komitéer, Region Midtjylland and Region Hovedstaden).
Genotyping the rs16906252 MGMT SNP
Genotyping was performed using pyrosequencing. The primer sequences are listed in Supplementary Table S2. PCR cycling was performed on the Gene PCR System 9700 (Applied Biosystems, Foster City, CA, USA). The cycling protocol started with one cycle of 95°C for 15 min for enzyme activation, followed by 38 cycles of 94°C for 20 s, 60°C for 20 s, 72°C for 20 s, and one cycle of 72°C for 10 min. For the reaction mixtures the PyroMark PCR Master Mix (Qiagen) was used at a final concentration at 1 ×, resulting in a final MgCl2 concentration of 1.5 mM. Final primer concentrations were 200 nM of each primer, and 25 ng of DNA was used. The final reaction volume was 25 μL. Samples were sequenced on the PyroMark Q24 (Qiagen) using the PyroMark Gold Q24 reagents (Qiagen), according to the manufactures' instructions.
Genotyping the rs13300553 DAPK1 SNP
Genotyping was performed using Sanger sequencing. The PCR product was created with primers containing an M13-tag (Supplementary Table S2). The cycling protocol started with one cycle of 95°C for 15 min, followed by 38 cycles of 95°C for 10 s, 63°C for 20 s, 72°C for 20 s, and one cycle of 72°C for 10 min. For the reaction mixtures the PyroMark PCR Master Mix (Qiagen) was used at a final concentration at 1 ×, resulting in a final MgCl2 concentration of 1.5 mM. Final primer concentrations were 200 nM of each primer, and 25 ng of DNA was used. The final reaction volume was 25 μL. The sequencing reaction was done using an M13 primer corresponding to the M13-tag of the forward PCR primer.
Preparation of controls for DNA methylation analysis
Standards of known MGMT and DAPK1 allelic methylation status were prepared by M.SssI (New England Biolabs, Ipswich, MA, USA) treatment, according to the manufactures' instructions, of PB DNA samples of known genotypes for the rs16906252 and rs13300553 SNPs. Thus, standards having both alleles methylated were created by M.SssI treatment of a heterozygous sample. Standards having only the C-allele or G-allele methylated was created by mixing an M.SssI treated sample homozygous for the C-allele or G-allele with an untreated and unmethylated sample homozygous for the T-allele or A-allele, and vice versa for the standard having only the T-allele or A-allele methylated. Standards of 10%, 2.5%, 1.25%, and 0.625% methylation were prepared by serially diluting the standards having both alleles methylated into unmethylated DNA. Unmethylated DNA was prepared by whole genome amplification of genomic DNA (Roche, Mannheim, Germany) as previously described36. Universal methylated DNA (Chemicon International, Temecula, CA, USA) was also used as a positive methylation control in all of the DNA methylation assays.
Cell lines
Diffuse large B-cell derived lymphoma cell lines: Farage, DB, DOHH2, HT, Pfeiffer, RL, and Toledo were purchased from the American Type Culture Collection (ATCC). The cells were cultured in RPMI 1640 medium with Glutamax supplemented with 10% fetal calf serum.
Bisulfite conversion
250 ng DNA from fresh frozen patient samples and controls were converted with the EZ DNA Methylation kit (Zymo Research) according to the manufactures' recommendations, with slight modifications; samples were incubated at 42°C for 30 minutes instead of 37°C for 15 minutes. For the bisulfite reaction the alternative incubation conditions described in the appendix were used. For the FFPE samples one μg of DNA was converted using the same protocol.
Allelic MSP-pyrosequencing
The MGMT MSP primers were designed to target the antisense strand and amplify the region surrounding the rs16906252 SNP. The DAPK1 MSP primers were designed to target the sense strand and amplify the region surrounding the rs13300553 SNP. Several non-CpG cytosines in each of the primers select against the amplification of incompletely converted molecules. Unmethylation specific PCR (uMSP) assays were designed to target the same regions (Supplementary Fig. S3A and S3B). The primer sequences are listed in Supplementary Table S2. PCR cycling was performed on the Gene PCR System 9700 (Applied Biosystems). The cycling protocols started with one cycle of 95°C for 15 min, followed by 45 cycles of 94°C for 20 s, 70°C (MGMT MSP) or 64°C (MGMT uMSP) or 66°C (DAPK1 MSP) or 64°C (DAPK1 uMSP) for 20 s, 72°C for 20 s, and one cycle of 72°C for 10 min. For the reaction mixtures the PyroMark PCR Master Mix (Qiagen) was used at a final concentration at 1 ×, resulting in a final MgCl2 concentration of 1.5 mM. Final primer concentrations were 200 nM of each primer, and 25 ng (fresh frozen samples and controls) or 100–400 ng (FFPE samples) of bisulfite converted DNA was used. The final reaction volumes were 25 μL. The PCR amplified DNA was run on a 2% agarose gel stained with ethidium bromide and visualized under UV-light. Samples, for which a band of the expected size was observed, were sequenced on the PyroMark Q24 (Qiagen) using the PyroMark Gold Q24 reagents (Qiagen), according to the manufactures' instructions.
Methylation independent PCR pyrosequencing assay
A traditional methylation independent PCR (MIP) pyrosequencing assay38 was designed to target the antisense strand of MGMT to allow simultaneous confirmation of the samples genotype for the rs16906252 SNP (Supplementary Fig. S3C). The sequencing primer was placed to allow six CpG sites around the rs16906252 SNP to be interrogated. The primers were designed to be methylation independent by avoiding CpG sites. When CpG sites could not be avoided a base that is equally mismatched to methylated and unmethylated states were placed at the potentially variable position. The primer sequences are listed in Supplementary Table S2. PCR cycling was performed on the Gene PCR System 9700 (Applied Biosystems). The cycling protocol started with one cycle of 95°C for 15 min, followed by 45 cycles of 94°C for 20 s, 58°C for 20 s, 72°C for 20 s, and one cycle of 72°C for 10 min. For the reaction mixtures the PyroMark PCR Master Mix (Qiagen) was used at a final concentration at 1 ×, resulting in a final MgCl2 concentration of 1.5 mM. Final primer concentrations were 200 nM of each primer, and 25 ng (fresh frozen samples and controls) or 100 ng (FFPE samples) of bisulfite converted DNA was used. The final reaction volume was 25 μL. Samples were sequenced on the PyroMark Q24 (Qiagen) using the PyroMark Gold Q24 reagents (Qiagen), according to the manufactures' instructions.
Bisulfite sequencing of single clones
To analyze the methylation status of individual alleles, bisulfite-converted DNA was amplified using MIP primers targeting the antisense strand (Supplementary Fig. S3C). The primer sequences are listed in Supplementary Table S2. PCR cycling was performed on the Gene PCR System 9700 (Applied Biosystems). The cycling protocols started with one cycle of 95°C for 15 min, followed by 45 cycles of 94°C for 20 s, 58°C for 20 s, 72°C for 20 s, and one cycle of 72°C for 10 min. For the reaction mixtures the 1 × PCR buffer, resulting in a final MgCl2 concentration of 1.5 mM, 666 nmol/L of each primer, 133 μmol/L of each dNTP, 0.5 U of HotStarTaq (Qiagen), and 25 ng of bisulfite converted DNA in a final volume of 15 μL. The PCR amplified DNA was run on a 2% agarose gel stained with ethidium bromide and visualized under UV-light, and cloned into the pCR2.1 vector using the TOPO-TA cloning kit (Invitrogen, Carlsbad, CA, USA) as previously described39. Plasmid DNA from individual clones was sequenced using an M13 primer (5′-CAGGAAACAGCTATGACC-3′) on the ABI Genetic Analyzer 3130 XL (Applied Biosystems) using the BigDye terminator kit v1.1 (Applied Biosystems) according to the manufactures' instructions.
Statistical analysis
Statistical analyses were performed in SPSS 19.0 for Windows (SPSS Inc.). Associations between the genotypes and methylation as well as Hardy–Weinberg equilibrium were assessed using Pearson chi-square tests. Correlations between overall survival and genotype or methylation status were estimated using the Kaplan-Meier method with the use of a log-rank test. The clinical characteristics and treatment outcomes were compared according to methylation status using one-way ANOVA, Person chi-squared tests, or Fisher exact tests when expected values were below five. Any differences were considered to be statistically significant when the p-value was < 0.05.
Author Contributions
M.S.G., H.M.N. and T.K. carried out the molecular genetic studies and analysed the data. F.A. collected the clinical data and performed the statistical analysis. E.R. classified the DLBCL samples. M.B.T. participated in the design of the study, carried out the molecular genetic studies, and analysed the data. L.H.H. participated in the design of the study and contributed reagents and materials. L.S.K. participated in the design of the study, carried out the molecular genetic studies, analysed the data, and wrote the manuscript. K.G. conceived the study, contributed reagents and materials, and helped to draft the manuscript. All authors read and approved the final manuscript.
Supplementary Material
Supplementary Information
Acknowledgments
This study was supported by grants from The Novo Nordisk Foundation, Rigshospitalets Research Foundation, The Danish Cancer Society, and The Danish Strategic Research Foundation. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- Esteller M. et al. Inactivation of the DNA-repair gene MGMT and the clinical response of gliomas to alkylating agents. N Engl J Med 343, 1350–4 (2000). [DOI] [PubMed] [Google Scholar]
- Hegi M. E. et al. MGMT gene silencing and benefit from temozolomide in glioblastoma. N Engl J Med 352, 997–1003 (2005). [DOI] [PubMed] [Google Scholar]
- Esteller M. et al. Hypermethylation of the DNA repair gene O(6)-methylguanine DNA methyltransferase and survival of patients with diffuse large B-cell lymphoma. J Natl Cancer Inst 94, 26–32 (2002). [DOI] [PubMed] [Google Scholar]
- Ohno T. et al. Loss of O6-methylguanine-DNA methyltransferase protein expression is a favorable prognostic marker in diffuse large B-cell lymphoma. Int J Hematol 83, 341–7 (2006). [DOI] [PubMed] [Google Scholar]
- Lee G. W. et al. Is inactivation of O6-methylguanine DNA methyltransferase still a favorable prognostic factor of patients with diffuse large B-cell lymphoma in the era of R-CHOP chemotherapy? Leuk Lymphoma 50, 1992–8 (2009). [DOI] [PubMed] [Google Scholar]
- Pike B. L. et al. DNA methylation profiles in diffuse large B-cell lymphoma and their relationship to gene expression status. Leukemia 22, 1035–43 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hawkins N. J. et al. MGMT methylation is associated primarily with the germline C > T SNP (rs16906252) in colorectal cancer and normal colonic mucosa. Mod Pathol (2009). [DOI] [PubMed] [Google Scholar]
- Kristensen L. S., Nielsen H. M., Hager H. & Hansen L. L. Methylation of MGMT in malignant pleural mesothelioma occurs in a subset of patients and is associated with the T allele of the rs16906252 MGMT promoter SNP. Lung Cancer 71, 130–6 (2011). [DOI] [PubMed] [Google Scholar]
- Ogino S. et al. MGMT germline polymorphism is associated with somatic MGMT promoter methylation and gene silencing in colorectal cancer. Carcinogenesis 28, 1985–90 (2007). [DOI] [PubMed] [Google Scholar]
- Leng S. et al. The A/G allele of rs16906252 predicts for MGMT methylation and is selectively silenced in premalignant lesions from smokers and in lung adenocarcinomas. Clin Cancer Res 17, 2014–23 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- McDonald K. L. et al. The T genotype of the MGMT C > T (rs16906252) enhancer single-nucleotide polymorphism (SNP) is associated with promoter methylation and longer survival in glioblastoma patients. Eur J Cancer 49, 360–8 (2013). [DOI] [PubMed] [Google Scholar]
- Zochbauer-Muller S. et al. Aberrant promoter methylation of multiple genes in non-small cell lung cancers. Cancer Res 61, 249–55 (2001). [PubMed] [Google Scholar]
- Mittag F. et al. DAPK promotor methylation is an early event in colorectal carcinogenesis. Cancer Lett 240, 69–75 (2006). [DOI] [PubMed] [Google Scholar]
- Amara K. et al. Prognostic significance of aberrant promoter hypermethylation of CpG islands in patients with diffuse large B-cell lymphomas. Ann Oncol 19, 1774–86 (2008). [DOI] [PubMed] [Google Scholar]
- Kristensen L. S., Raynor M. P., Candiloro I. & Dobrovic A. Methylation profiling of normal individuals reveals mosaic promoter methylation of cancer-associated genes. Oncotarget 3, 450–61 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rand K., Qu W., Ho T., Clark S. J. & Molloy P. Conversion-specific detection of DNA methylation using real-time polymerase chain reaction (ConLight-MSP) to avoid false positives. Methods 27, 114–20 (2002). [DOI] [PubMed] [Google Scholar]
- Shaw R. J., Akufo-Tetteh E. K., Risk J. M., Field J. K. & Liloglou T. Methylation enrichment pyrosequencing: combining the specificity of MSP with validation by pyrosequencing. Nucleic Acids Res 34, e78 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mikeska T. et al. Optimization of quantitative MGMT promoter methylation analysis using pyrosequencing and combined bisulfite restriction analysis. J Mol Diagn 9, 368–81 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Candiloro I. L. & Dobrovic A. Detection of MGMT promoter methylation in normal individuals is strongly associated with the T allele of the rs16906252 MGMT promoter single nucleotide polymorphism. Cancer Prev Res (Phila Pa) 2, 862–7 (2009). [DOI] [PubMed] [Google Scholar]
- Herman J. G., Graff J. R., Myohanen S., Nelkin B. D. & Baylin S. B. Methylation-specific PCR: a novel PCR assay for methylation status of CpG islands. Proc Natl Acad Sci U S A 93, 9821–6 (1996). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong H. L. et al. Rapid and quantitative method of allele-specific DNA methylation analysis. Biotechniques 41, 734–9 (2006). [DOI] [PubMed] [Google Scholar]
- Kristensen L. S. & Hansen L. L. PCR-based methods for detecting single-locus DNA methylation biomarkers in cancer diagnostics, prognostics, and response to treatment. Clin Chem 55, 1471–83 (2009). [DOI] [PubMed] [Google Scholar]
- Kristensen L. S., Treppendahl M. B. & Gronbaek K. Analysis of epigenetic modifications of DNA in human cells. Curr Protoc Hum Genet 77, 20.2.1–20.2.22 (2013). [DOI] [PubMed] [Google Scholar]
- Xiong Z. & Laird P. W. COBRA: a sensitive and quantitative DNA methylation assay. Nucleic Acids Res 25, 2532–4 (1997). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guldberg P., Worm J. & Gronbaek K. Profiling DNA methylation by melting analysis. Methods 27, 121–7 (2002). [DOI] [PubMed] [Google Scholar]
- Lade-Keller J. et al. Evaluation of BRAF mutation testing methodologies in formalin-fixed, paraffin-embedded cutaneous melanomas. J Mol Diagn 15, 70–80 (2013). [DOI] [PubMed] [Google Scholar]
- Shoemaker R., Deng J., Wang W. & Zhang K. Allele-specific methylation is prevalent and is contributed by CpG-SNPs in the human genome. Genome Res 20, 883–9 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kerkel K. et al. Genomic surveys by methylation-sensitive SNP analysis identify sequence-dependent allele-specific DNA methylation. Nat Genet 40, 904–8 (2008). [DOI] [PubMed] [Google Scholar]
- Lee S. M. et al. Prognostic significance of O6-methylguanine DNA methyltransferase and p57 methylation in patients with diffuse large B-cell lymphomas. APMIS 117, 87–94 (2009). [DOI] [PubMed] [Google Scholar]
- Hiraga J. et al. Promoter hypermethylation of the DNA-repair gene O6-methylguanine-DNA methyltransferase and p53 mutation in diffuse large B-cell lymphoma. Int J Hematol 84, 248–55 (2006). [DOI] [PubMed] [Google Scholar]
- Amara K. et al. Presence of simian virus 40 in diffuse large B-cell lymphomas in Tunisia correlates with germinal center B-cell immunophenotype, t(14;18) translocation, and P53 accumulation. Mod Pathol 21, 282–96 (2008). [DOI] [PubMed] [Google Scholar]
- Nakamichi I. et al. Correlation between promoter hypermethylation of GSTP1 and response to chemotherapy in diffuse large B cell lymphoma. Ann Hematol 86, 557–64 (2007). [DOI] [PubMed] [Google Scholar]
- Katzenellenbogen R. A., Baylin S. B. & Herman J. G. Hypermethylation of the DAP-kinase CpG island is a common alteration in B-cell malignancies. Blood 93, 4347–53 (1999). [PubMed] [Google Scholar]
- Reddy A. N. et al. Death-associated protein kinase promoter hypermethylation in normal human lymphocytes. Cancer Res 63, 7694–8 (2003). [PubMed] [Google Scholar]
- Eads C. A. et al. MethyLight: a high-throughput assay to measure DNA methylation. Nucleic Acids Res 28, E32 (2000). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kristensen L. S., Mikeska T., Krypuy M. & Dobrovic A. Sensitive Melting Analysis after Real Time- Methylation Specific PCR (SMART-MSP): high-throughput and probe-free quantitative DNA methylation detection. Nucleic Acids Res 36, e42 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hansen L. L., Andersen J., Overgaard J. & Kruse T. A. Molecular genetic analysis of easily accessible breast tumour DNA, purified from tissue left over from hormone receptor measurement. APMIS 106, 371–7 (1998). [DOI] [PubMed] [Google Scholar]
- Mikeska T., Felsberg J., Hewitt C. A. & Dobrovic A. Analysing DNA methylation using bisulphite pyrosequencing. Methods Mol Biol 791, 33–53 (2011). [DOI] [PubMed] [Google Scholar]
- Treppendahl M. B. et al. Allelic methylation levels of the noncoding VTRNA2-1 located on chromosome 5q31.1 predict outcome in AML. Blood 119, 206–16 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Information