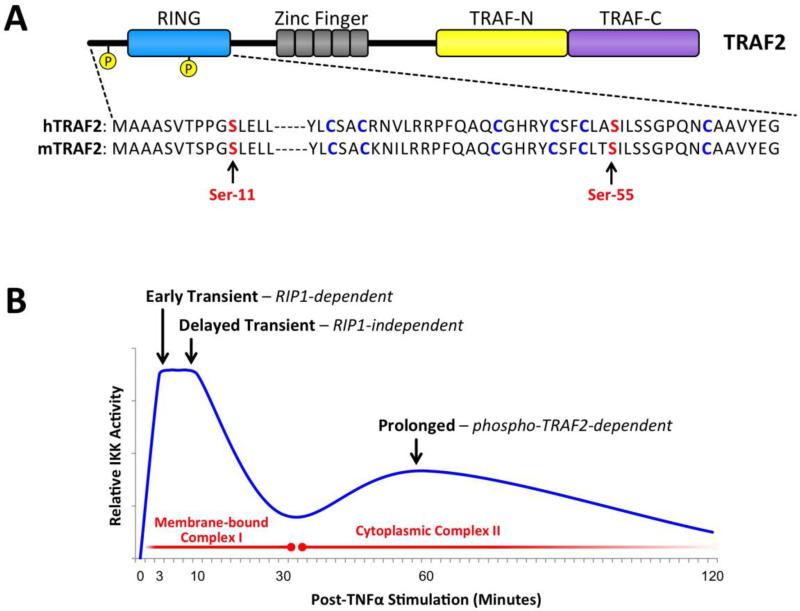
Fig. 2.
TRAF2 phosphorylation regulates the prolonged phase of IKK activation. (A) Schematic illustration of the TRAF2 protein domains, phosphorylation sites, consensus C and H residues in the RING domain. (B) Upon TNFR1 ligation, the membrane bound complex-I activates the early and delayed phases of IKK through RIP1-dependent and -independent pathways. Thereafter, TRAF2 is phosphorylated at Ser-11 and Ser-55, which triggers the translocation of the TRADD/RIP1/TRAF2/cIAP1 complex from TNFR1 to the cytoplasm to form complex-IIA. This leads to activation of the prolonged phases of IKK in a TRAF2 phosphorylation-dependent manner, which is also required for the efficient expression of some NF-κB target genes.