Abstract
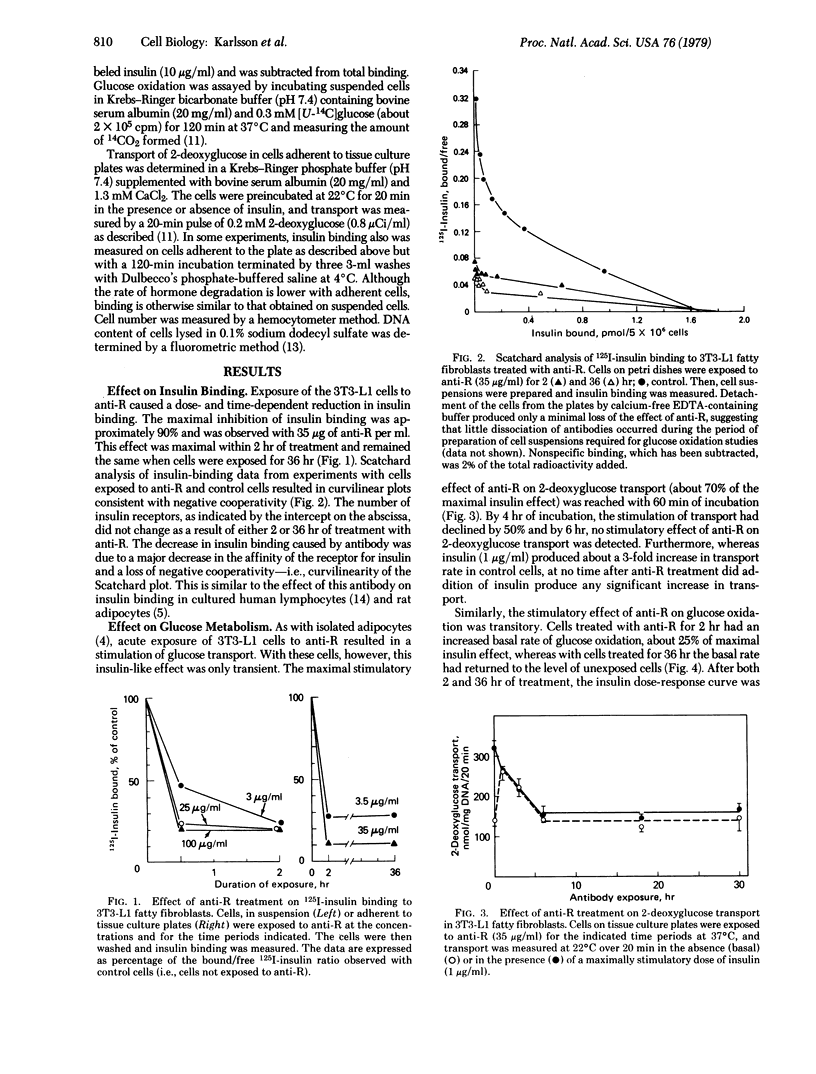
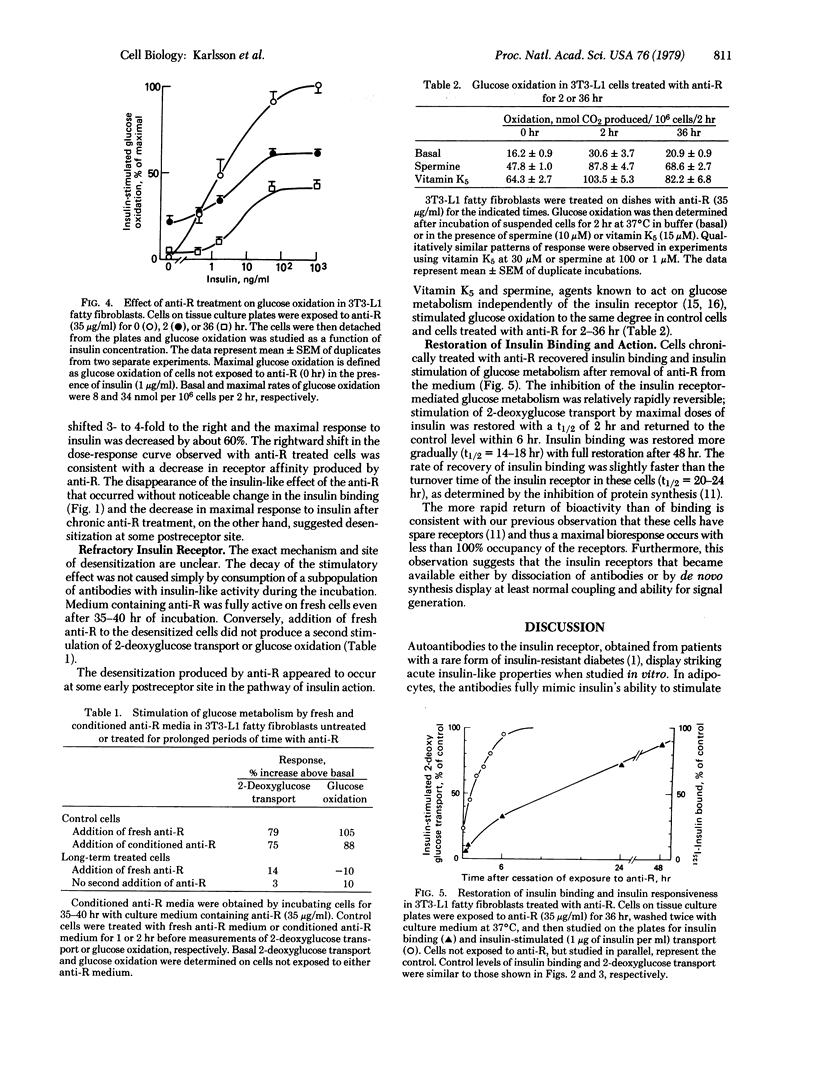
We have used an adipocyte-like cell line, the 3T3-L1 fatty fibroblasts, to compare acute and chronic effects of autoantibodies directed against the insulin receptor. Acute exposure of the cells in tissue culture to the antibodies resulted in a blockade of insulin binding and stimulation of 2-deoxy-glucose transport and glucose oxidation. Maximal acute effects were reached within 30-120 min. Subsequently, the stimulatory response decayed and, after 6 hr in the continuous presence of the antibodies, basal glucose metabolism had returned to the level observed with unexposed cells and a state of severe insulin resistance prevailed. In contrast to the decay of bioresponse, no change in insulin binding was detectable over the same time period. The mechanism of desensitization seemed to involve events early after insulin binding to receptor because cells exposed to antibody for prolonged periods of time, although unresponsive to insulin and antireceptor antibodies, responded normally to both spermine and vitamin K5, agents that stimulate glucose metabolism independently of the insulin receptor. These data suggest that prolonged or continuous occupancy of the insulin receptor by a ligand, in this case antireceptor antibodies, does not produce a continuous biological response. Instead, there is desensitization at some early step in the pathway for insulin action. These observations have important implications with respect to the mechanism of insulin action and to other situations in which there is long-term exposure of cells to antibodies that react with membrane components.
Keywords: hormone action, insulin resistance, 3T3-L1 fatty fibroblasts
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Czech M. P. Cellular basis of insulin insensitivity in large rat adipocytes. J Clin Invest. 1976 Jun;57(6):1523–1532. doi: 10.1172/JCI108422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Czech M. P., Lawrence J. C., Jr, Lynn W. S. Hexose transport in isolated brown fat cells. A model system for investigating insulin action on membrane transport. J Biol Chem. 1974 Sep 10;249(17):5421–5427. [PubMed] [Google Scholar]
- Drachman D. B. Myasthenia gravis (first of two parts). N Engl J Med. 1978 Jan 19;298(3):136–142. doi: 10.1056/NEJM197801192980305. [DOI] [PubMed] [Google Scholar]
- Flier J. S., Kahn C. R., Jarrett D. B., Roth J. Autoantibodies to the insulin receptor. Effect on the insulin-receptor interaction in IM-9 lymphocytes. J Clin Invest. 1977 Oct;60(4):784–794. doi: 10.1172/JCI108832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flier J. S., Kahn C. R., Jarrett D. B., Roth J. Characterization of antibodies to the insulin receptor: a cause of insulin-resistant diabetes in man. J Clin Invest. 1976 Dec;58(6):1442–1449. doi: 10.1172/JCI108600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flier J. S., Kahn C. R., Roth J., Bar R. S. Antibodies that impair insulin receptor binding in an unusual diabetic syndrome with severe insulin resistance. Science. 1975 Oct 3;190(4209):63–65. doi: 10.1126/science.170678. [DOI] [PubMed] [Google Scholar]
- Gavin J. R., 3rd, Roth J., Neville D. M., Jr, de Meyts P., Buell D. N. Insulin-dependent regulation of insulin receptor concentrations: a direct demonstration in cell culture. Proc Natl Acad Sci U S A. 1974 Jan;71(1):84–88. doi: 10.1073/pnas.71.1.84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hinegardner R. T. An improved fluorometric assay for DNA. Anal Biochem. 1971 Jan;39(1):197–201. doi: 10.1016/0003-2697(71)90476-3. [DOI] [PubMed] [Google Scholar]
- Kahn C. R., Baird K. L., Jarrett D. B., Flier J. S. Direct demonstration that receptor crosslinking or aggregation is important in insulin action. Proc Natl Acad Sci U S A. 1978 Sep;75(9):4209–4213. doi: 10.1073/pnas.75.9.4209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kahn C. R., Baird K., Filier J. S., Jarrett D. B. Effects of autoantibodies to the insulin receptor on isolated adipocytes. Studies of insulin binding and insulin action. J Clin Invest. 1977 Nov;60(5):1094–1106. doi: 10.1172/JCI108861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kahn C. R., Flier J. S., Bar R. S., Archer J. A., Gorden P., Martin M. M., Roth J. The syndromes of insulin resistance and acanthosis nigricans. Insulin-receptor disorders in man. N Engl J Med. 1976 Apr 1;294(14):739–745. doi: 10.1056/NEJM197604012941401. [DOI] [PubMed] [Google Scholar]
- Kahn C. R., Megyesi K., Bar R. S., Eastman R. C., Flier J. S. Receptors for peptide hormones. New insights into the pathophysiology of disease states in man. Ann Intern Med. 1977 Feb;86(2):205–219. doi: 10.7326/0003-4819-86-2-205. [DOI] [PubMed] [Google Scholar]
- Kasuga M., Akanuma Y., Tsushima T., Suzuki K., Kosaka K., Kibata M. Effects of antiinsulin receptor autoantibody on the metabolism of rat adipocytes. J Clin Endocrinol Metab. 1978 Jul;47(1):66–77. doi: 10.1210/jcem-47-1-66. [DOI] [PubMed] [Google Scholar]
- Le Marchand-Brustel Y., Gorden P., Flier J. S., Kahn C. R., Freychet P. Anti-insulin receptor antibodies inhibit insulin binding and stimulate glucose metabolism in skeletal muscle. Diabetologia. 1978 May;14(5):311–317. doi: 10.1007/BF01223022. [DOI] [PubMed] [Google Scholar]
- Lefkowitz R. J., Mullikin D., Williams L. T. A desensitized state of the beta adrenergic receptor not associated with high-affinity agonist occupancy. Mol Pharmacol. 1978 Mar;14(2):376–380. [PubMed] [Google Scholar]
- Lin M. C., Nicosia S., Lad P. M., Rodbell M. Effects of GTP on binding of (3H) glucagon to receptors in rat hepatic plasma membranes. J Biol Chem. 1977 Apr 25;252(8):2790–2792. [PubMed] [Google Scholar]
- Livingston J. N., Gurny P. A., Lockwood D. H. Insulin-like effects of polyamines in fat cells. Mediation by H2O2 formation. J Biol Chem. 1977 Jan 25;252(2):560–562. [PubMed] [Google Scholar]
- Livingston J. N., Purvis B. J., Lockwood D. H. Insulin-dependent regulation of the insulin-sensitivity of adipocytes. Nature. 1978 Jun 1;273(5661):394–396. doi: 10.1038/273394a0. [DOI] [PubMed] [Google Scholar]
- Olefsky J. M. The insulin receptor: its role in insulin resistance of obesity and diabetes. Diabetes. 1976 Dec;25(12):1154–1162. doi: 10.2337/diab.25.12.1154. [DOI] [PubMed] [Google Scholar]
- Roth J. Methods for assessing immunologic and biologic properties of iodinated peptide hormones. Methods Enzymol. 1975;37:223–233. doi: 10.1016/s0076-6879(75)37018-3. [DOI] [PubMed] [Google Scholar]
- Roth J. Peptide hormone binding to receptors: a review of direct studies in vitro. Metabolism. 1973 Aug;22(8):1059–1073. doi: 10.1016/0026-0495(73)90225-4. [DOI] [PubMed] [Google Scholar]
- Rubin C. S., Lai E., Rosen O. M. Acquisition of increased hormone sensitivity during in vitro adipocyte development. J Biol Chem. 1977 May 25;252(10):3554–3557. [PubMed] [Google Scholar]
- Williams L. T., Lefkowitz R. J. Slowly reversible binding of catecholamine to a nucleotide-sensitive state of the beta-adrenergic receptor. J Biol Chem. 1977 Oct 25;252(20):7207–7213. [PubMed] [Google Scholar]


