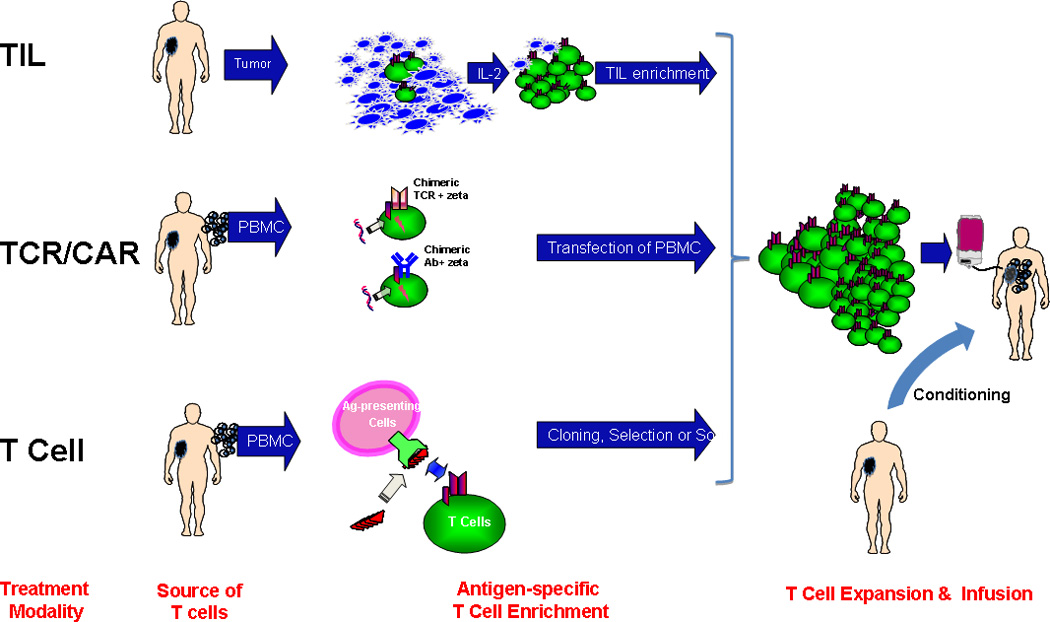
Adoptive Cell Therapy is represented by three general approaches:
Enrichment and expansion of tumor-infiltrating lymphocytes (TIL) from a disaggregated tumor biopsy sample
Genetic transfer of T Cell Receptor (TCR) recognizing tumor antigen-derived peptide-MHC target or Chimeric Antibody Receptor (CAR) recognizing surface tumor protein
Enrichment of endogenous antigen-specific T cells from peripheral blood mononuclear cells by in vitro stimulation followed by cell selection or cloning. PBMCs are a source of both antigen-presenting cells and T cells.
Following enrichment, the population of tumor-reactive T cells undergoes rapid expansion of 1000–5000 fold achieving 10 – 100 billion cells for adoptive transfer. Patients often receive a lymphodepleting conditioning regimen pre-infusion followed by exogenous IL-2. In the case of adoptive TIL therapy, patients receive high-dose near ablative or fully ablative conditioning pre-infusion and a course of high-dose IL-2 post-infusion. In Besser et al, ‘young’ TIL are generated using a shortened pre-expansion culture phase prior to rapid cell expansion, enabling production of an infusible T cell product within 5–7 weeks from time of tumor collection.