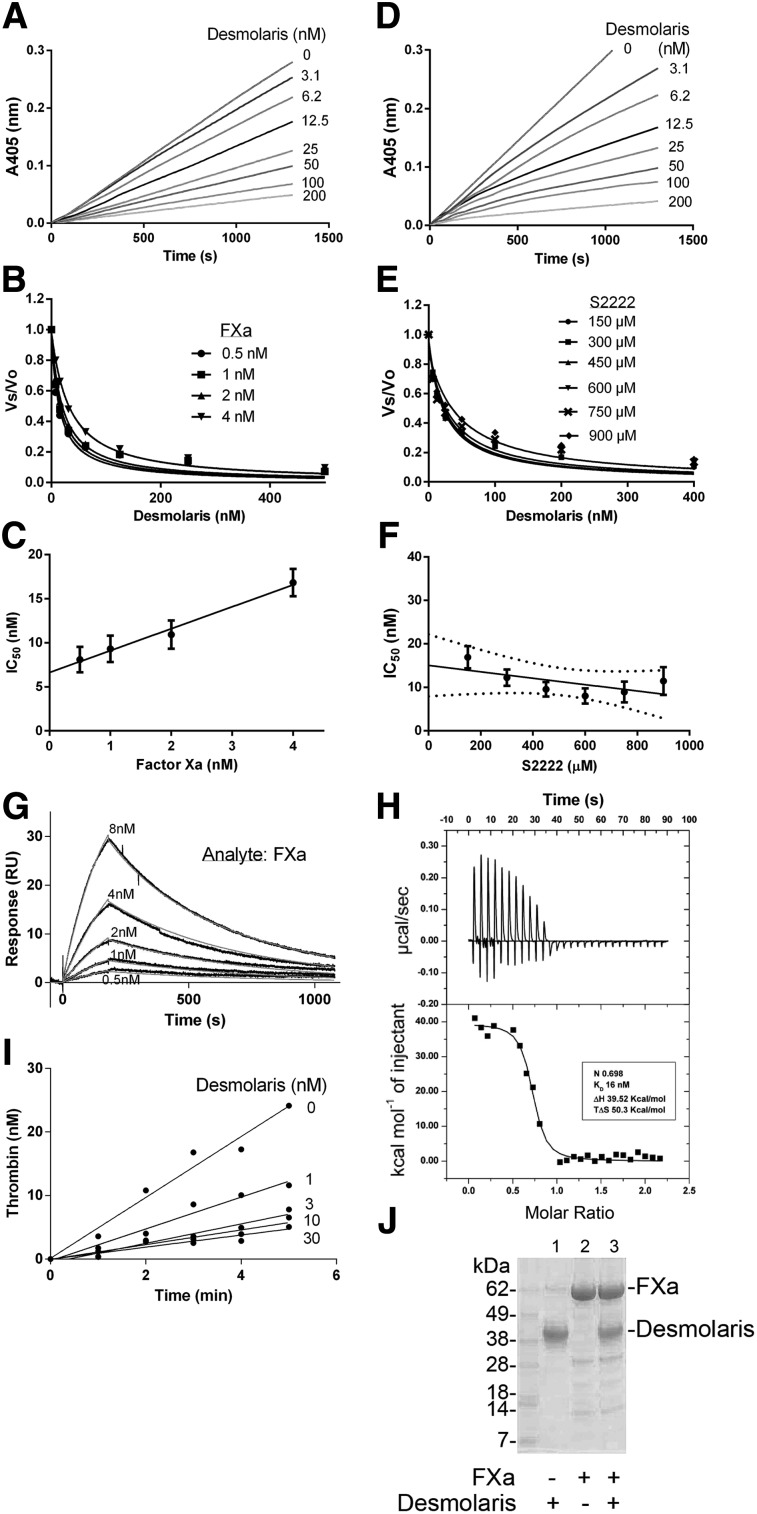
Figure 3.
Desmolaris is a slow and tight inhibitor of FXa. (A) Inhibition of the catalytic activity of FXa. Reactions started with addition of S2222 (250 μM) to a mixture containing Desmolaris incubated for 1 hour with FXa (1 nM). Substrate hydrolysis was followed for 2 hours at 405 nm. (B) Tight inhibition. Experiments were performed as in (A) but at 0.5, 1, 2, and 4 nM FXa. The rate of substrate hydrolysis between 30 and 60 minutes (Vmax mode) was transformed as Vs/Vo and plotted against Desmolaris concentration. Data points were fitted with the Morrison equation to calculate the IC50 at each enzyme concentration. (C) Plot of IC50 and FXa produces a straight line typical of tight inhibitors. (D) Slow-type inhibition. Reactions started with addition of FXa (1 nM) to a mixture containing Desmolaris and S2222 (250 μM). Substrate hydrolysis was followed for 2 hours at 405 nm. (E) IC50 determination. FXa was added to Desmolaris in the presence of S2222 (150, 300, 450, 600, 750, and 900 μM). The ratio Vs/Vo obtained (Vmax mode) between 30 and 60 minutes was plotted against S2222 concentrations. Data were fitted with the Morrison equation to calculate the IC50 values at each substrate concentration. Six experiments were performed, and each data point is the average of duplicate determinations. (F) Determination of the Ki. Plot of IC50 vs S2222 concentrations was fitted by linear regression and the y intercept equals the Ki. Confidence interval is shown as dotted lines. (G) SPR experiments. Factor Xa at the indicated concentrations was injected over immobilized Desmolaris for 180 seconds. Dissociation of the Desmolaris-FXa complex was monitored for 900 seconds. Representative sensorgrams are shown in black lines; global fitting of the data points using the Langmuir equation is depicted in red lines. (H) Solution binding of Desmolaris to FXa by ITC. (Upper panel) Baseline-adjusted heats per injection of FXa (10 μM) into Desmolaris (1.0 μM). (Lower panel) Molar enthalpies per injection for FXa interaction with Desmolaris. Filled squares, measured enthalpies; solid line, fit of experimental data to a single site-binding model. Thermodynamic parameters: ΔH in kcal/mol, TΔS in kcal/mol, and KD are indicated in the inset. (I) Effects of Desmolaris in the prothrombinase complex. FXa was incubated with Desmolaris (0-30 nM) followed by addition of FVa, phosphatidylcholine/phosphatidylserine, and prothrombin in the presence of Ca2+. Aliquots were taken to estimate thrombin generation. Data points are the average for triplicate determination. (J) Cleavage of Desmolaris. Desmolaris (7.5 μM) was incubated with FXa (1.7 μM) for 8 hours. The mixture was loaded in a NuPAGE gel. The bands corresponding to FXa or Desmolaris are indicated. Lane 1, Desmolaris; lane 2, FXa; lane 3, FXa plus Desmolaris. No cleavage is observed.