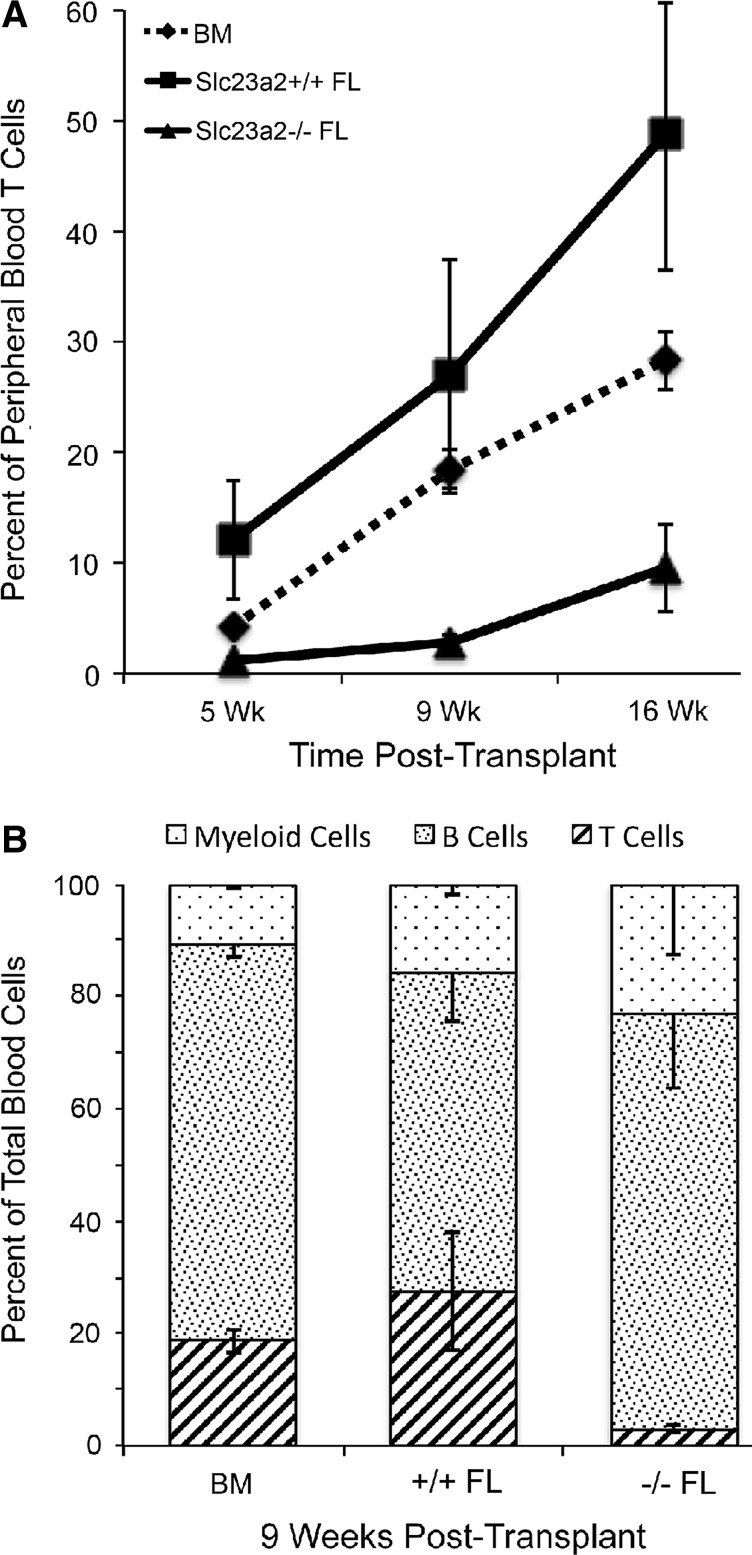
FIG. 4.
Fetal liver cells lacking the ability to accumulate ascorbic acid due to targeted mutation of Slc23a2 have defective T-cell maturation when transplanted into irradiated recipients. Fetal liver cells (Ly-5.2) identified as Slc23a2+/+ or Slc23a2−/− by polymerase chain reaction (PCR) were mixed with equal numbers of normal bone marrow cells (Ly-5.1/Ly-5.2) for injection at a dose of 2×106 total cells into four lethally irradiated recipient mice (Ly-5.1). Fetal liver-derived peripheral blood cells were identified by flow cytometry based on Ly-5.1 and Ly-5.2 antibody staining and phenotyped as myeloid, T-cell, or B-cell lineages at the indicated times after transplantation. Panel (A) shows T-lineage engraftment at three time points post-transplantation. Panel (B) shows myeloid, T, and B lineages 9 weeks post-transplantation. BM indicates the lineage contributions by the Ly-5.1/Ly-5.2 bone marrow cells in the same recipient mice, pooling data from both Slc23a2+/+ and Slc23a2−/− FL recipients. At 16 weeks post-transplantation, the frequency of Slc23a2−/− T cells was 20% of that seen with Slc23a2+/+ T cells (p=0.023 by two-tailed t-test). Values are mean±standard error.