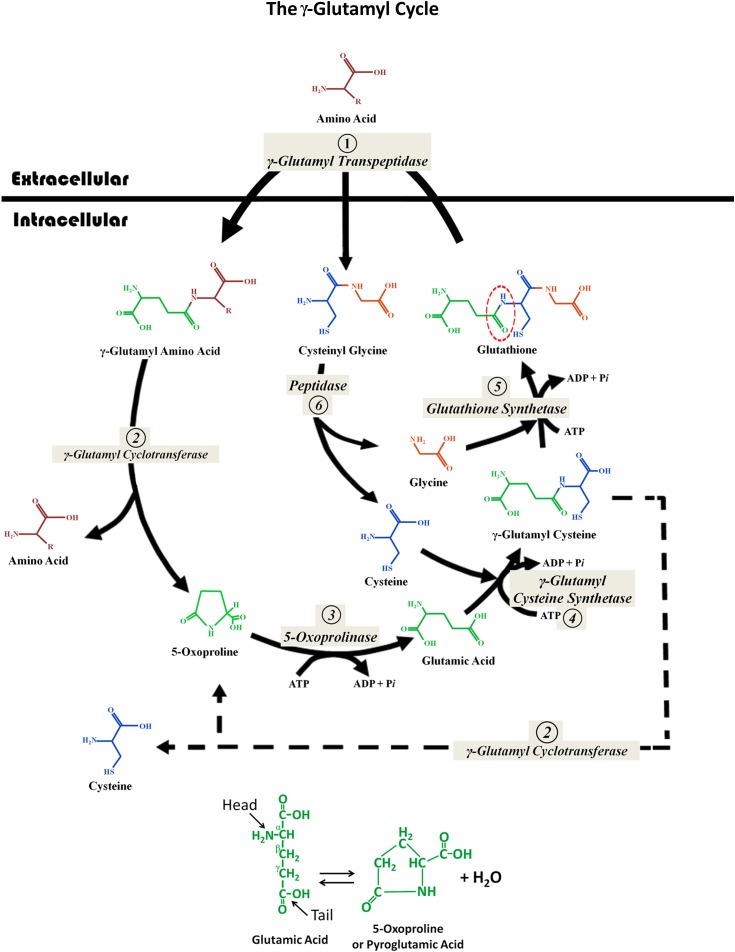
Figure 2.
The γ-glutamyl cycle. This cycle consists of six enzymatic reactions composed of two synthetic reactions and four degradation steps. (Step 1) Glutathione crosses the cell membrane where the enzyme γ-glutamyl transpeptidase, which is bound to the outer cell membrane, splits the molecule at the glutamyl γ-linkage (dotted circle). The released glutamic acid is then attached to another extracellular amino acid (or a peptide or water), again via the unusual γ-linkage. The γ-glutamyl-amino acid dipeptide and the cysteinylglycine dipeptide are both transported into the cell. (Step 2) The reaction catalyzed by γ-glutamyl cyclotransferase releases the amino acid and also “cyclizes” the glutamic acid to form 5-oxoproline (pyroglutamic acid). (Step 3) The 5-oxoproline ring is broken open by 5-oxoprolinase (pyroglutamate hydrolase) with energy input from ATP hydrolysis. (Step 4) Cysteine is bound to glutamic acid via a γ-linkage by the enzyme γ-glutamyl cysteine synthetase with energy input from ATP hydrolysis. This forms γ-glutamyl cysteine. (Step 5) Glutathione synthetase, with energy input from ATP hydrolysis, adds a glycine to reform glutathione. (Note steps 4 and 5 are the same as shown in Figure 2.) (Step 6) Cysteinylglycine is split by dipeptidase into cysteine and glycine, which are utilized in steps 4 and 5. Each complete rotation of this cycle requires the energy input derived from the hydrolysis of three molecules of ATP (steps 3–5). The net “work” produced by each turn of this cycle is the transport of one amino acid molecule across the cell membrane from the extracellular fluid into the cytoplasm. The interrupted line shows that under certain conditions (e.g., glycine deficiency), γ-glutamylcysteine can also be split by the enzyme γ-glutamyl cyclotransferase into cysteine and 5-oxoproline. The interconversion of glutamic acid and 5-oxoproline is shown below the cycle.