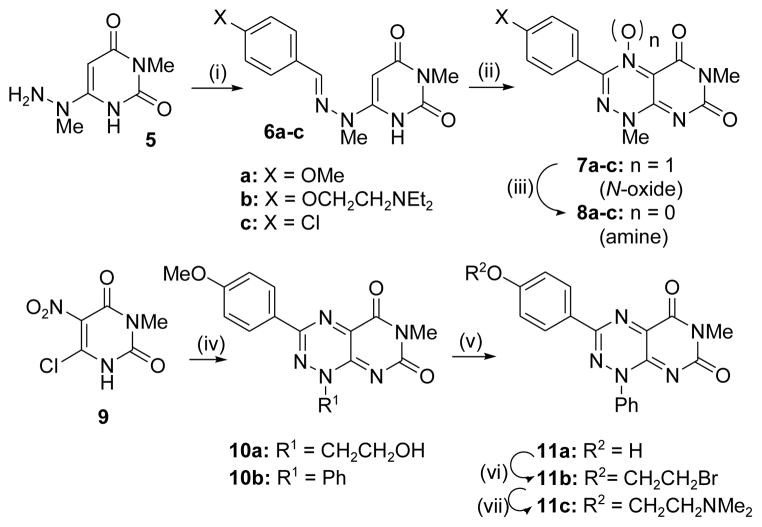
Scheme 1.
Synthesis of analogues in the xanthothricin series. (i) 4-substituted benzaldehyde, EtOH, reflux (31–87% yield); (ii) NaNO2, aq. AcOH, 0 °C – 25 °C (~80% yield); (iii) DTT, EtOH (47 – 87% yield); (iv) see reference 20; (v) for 10b, BBr3, DCM (63% yield); (vi) BrCH2CH2Br, Cs2CO3, DMF (47% yield); (vii) NHMe2, ACN, 80 °C (33% yield). See Supplemental Materials for experimental details.