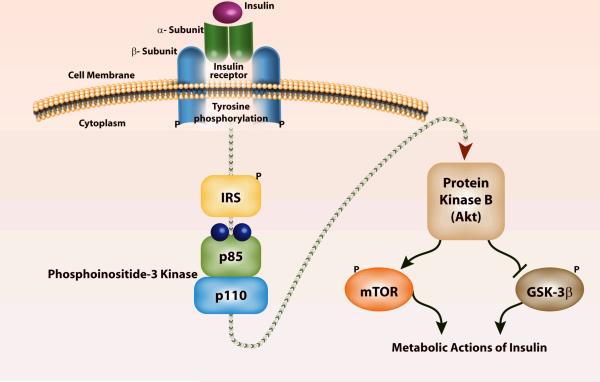
Figure 1. Signal transduction via insulin receptor (IR) and its downstream signaling proteins.
The IR is a kinase, an enzyme that catalyzes the transfer of phosphate from adenosine triphosphate (ATP) to another substrate. When insulin binds to the IR it undergoes autophosphorylation and catalyzes the tyrosine phosphorylation of insulin receptor substrates-1 and -2 (IRS). These IRS proteins interact with diverse signaling molecules including phosphoinositide-3 kinase (PI3K), which in turn activates protein kinase B (a.k.a. Akt). The downstream proteins controlled by Akt/PKB include mammalian target of rapamycin (mTOR) and glycogen synthase kinase-3β (GSK-3β). The metabolic and potent anabolic actions of insulin include glucose metabolism, glycogen/lipid/protein synthesis, cell growth and survival, and anti-inflammation. These pleiotropic effects of insulin are mediated by specific gene expression, translation of proteins, and enhanced mitochondrial function.