Abstract
Background:
Grewia asiatica Linn. (Family: Tiliaceae), called Phalsa in Hindi is an Indian medicinal plant used for a variety of therapeutic and nutritional uses. The root bark of the plant is traditionally used in rheumatism (painful chronic inflammatory condition).
Aims:
The present study demonstrates the analgesic and anti-inflammatory activity of root bark of G. asiatica in rodents.
Settings and Design:
The methanolic extract of Grewia asiatica (MEGA) and aqueous extract of Grewia asiatica (AEGA) of the bark were prepared and subjected to phytochemical tests and pharmacological screening for analgesic and anti-inflammatory effect in rodents.
Materials and Methods:
Analgesic effect was studied using acetic acid-induced writhing in mice and hot plate analgesia in rats while anti-inflammatory activity was investigated using carrageenan-induced paw oedema in rats. The MEGA or AEGA was administered orally in doses of 200 and 400 mg/kg/day of body weight.
Statistical Analysis:
Data were analysed by one-way analysis of variance followed by Dunnett's test.
Results:
The extracts showed a significant inhibition of writhing response and increase in hot plate reaction time and also caused a decrease in paw oedema. The effects were comparable with the standard drugs used.
Conclusions:
The present study indicates that root bark of G. asiatica exhibits peripheral and central analgesic effect and anti-inflammatory activity, which may be attributed to the various phytochemicals present in root bark of G. asiatica.
KEY WORDS: Analgesic, anti-inflammatory, Grewia asiatica, root bark
INTRODUCTION
Alarge number of plants are being increasingly used in the treatment of pain and inflammatory conditions.[1,2] The preference to the herbal agents is more compared to conventional non-steroidal anti-inflammatory drugs (NSAIDs) because NSAIDs administration is associated with the risk of producing adverse effects such as gastric ulceration, thrombocytopenia, skin rashes.[3] Plant based medicines are widely used in Ayurvedic system of medicine as Vedana śāmaka or śothahara.[4] Thus, there is growing interest in the pharmacological evaluation of various plants used in the Indian traditional system of medicine for treatment of inflammatory conditions.
Grewia asiatica Linn. (Family: Tiliaceae), called Phalsa in Hindi, Alpāsthi in Sanskrit is cultivated on a commercial scale mainly in the Northern and Western states of India.[5,6] It is known for its medicinal properties. In this, the fruit possesses astringent, cooling and stomachic properties. It is reported that unripe Phalsa fruit alleviates inflammation and is administered in respiratory, cardiac and blood disorders. It is also used for fever reduction. The fruits are also documented to have antimalarial and anti-ulcer effect.[7] A polyherbal preparation-PD10, which contains fruits of G. asiatica as an important constituent has been demonstrated to possess antipyretic-analgesic activity in animals.[7] The leaves are applied on pustular skin eruptions and they are known to have antibiotic action.[8,9] The infusion of the bark is given as a demulcent, febrifuge and is also used in the treatment of diarrhoea. In traditional uses, the Santal tribes use the root-bark of G. asiatica for treating rheumatism.[10,11]
In view of the traditional use of root bark of G. asiatica in rheumatism, a painful chronic inflammatory condition, we thought it worthwhile to investigate analgesic and anti-inflammatory effect of methanolic extract of Grewia asiatica and aqueous extract of Grewia asiatica (MEGA and AEGA) root bark of G. asiatica in the experimental models of pain and inflammation.
MATERIALS AND METHODS
Plant material
The root barks of G. asiatica were collected from Jorasi village, Gwalior in the month of January and authenticated by a Taxonomist of the National Research Institute for Ayurveda-Siddha Human Resource Development, Gwalior.
Preparation of plant extracts
The barks were shade dried and coarsely powdered. The powder was subjected to defatting in soxhlet apparatus using petroleum ether at 60-80°C. After this, MEGA was collected using 95% methanol in soxhlet apparatus. The soxhlet extraction of bark powder with methanol was carried out until the complete extraction. The AEGA was collected using cold maceration for 72 h. The solvents were completely removed under reduced pressure using rotary evaporator to get the yield 12.69% and 13.01% w/w, respectively.
Phytochemical screening of the extracts
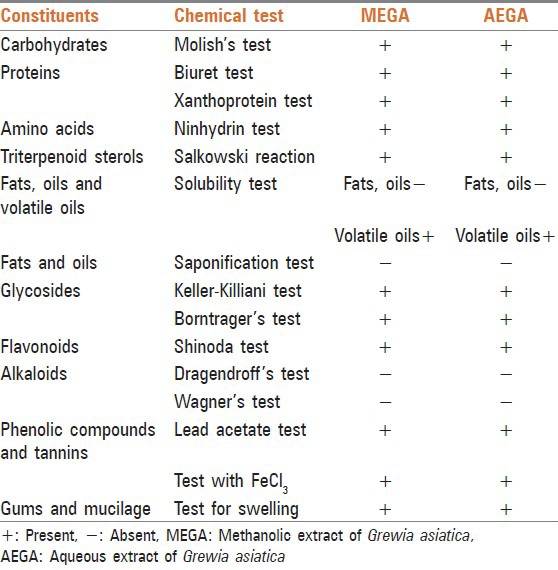
The extracts (MEGA and AEGA) were subjected to various tests to study the presence of different phytochemical constituents (carbohydrates, proteins, fats and oils, sterols, glycosides, flavonoids, alkaloids, phenolic compounds and tannins) using established methods [Table 1].[12]
Table 1.
Phytochemical screening of the MEGA and AEGA
Experimental animals
Male Swiss albino mice (22-25 g) and Wistar rats (220-250 g) were used for assessment of analgesic activity while male Wistar rats (220-250 g) were used for assessment of anti-inflammatory activity. Animals were bred and maintained at Central Animal Facility of the Institute under standard housing conditions of temperature 25 ± 2°C, relative humidity 55-65% and light and dark cycles of 12 h, respectively. Animals were provided with standard pellet diet (Ashirwad brand, Chandigarh) and water ad libitum. Principles of laboratory animal care guidelines were followed and prior permission was sought from the Institute Animal Ethics Committee for conducting the experiments (proposal no. CRI-GWL/IAEC/2010/03).
Drugs and chemicals
Indomethacin (IND) (Alfa remedies, Ambala) was obtained as gift sample. Sodium carboxymethylcellulose (Himedia, Mumbai) was procured from local market. Carrageenan was procured from Sigma Aldrich, USA. Pentazocine (Fortwin, Ranbaxy Laboratories Ltd., Ahmedabad) was purchased in injectable form. Drug solutions were freshly prepared and their doses are expressed in terms of their free bases.
Doses and treatments
Mice and rats were divided into six different groups (n = 5). MEGA was suspended in 0.5% sodium carboxymethylcellulose in normal saline while AEGA was solubilised as such for administration purpose. MEGA and AEGA were administered in graded doses of 200 and 400 mg/kg, orally. The doses selected were based on acute toxicity study and previous reports.[13,14] The extracts and standard drugs were given in volume of 10 ml/kg, orally and 5 ml/kg, intra-peritoneally (i.p.) All rodents were fasted for 12 h before starting the test.
Acute toxicity study
The acute oral toxicity study was carried out according to Organisation for Economic Cooperation and Development (OECD) guidelines 423. The limit test for acute toxicity of MEGA and AEGA was carried out in the dose of 2000 mg/kg, orally, in rats. The rats were observed for a period of 72 h and thereafter up to 14 days for behavioural, neurological, autonomic profiles and for any lethality, moribund state, or death.[15]
Assessment of analgesic activity
Assessment of peripheral analgesic effect of MEGA and AEGA
The peripheral analgesic effect of MEGA and AEGA was evaluated using acetic acid-induced writhing test model.[16] Writhing is defined as stretch, torsion to one side, retraction of the abdomen and opisthotonous so that the belly of the mouse touches to the floor. Abdominal writhing episodes were induced in mice by i.p. injection of acetic acid (10 ml/kg of 0.6%). IND (10 mg/kg, orally) was used as standard drug.[17] Mice were pre-treated with vehicle or MEGA or AEGA or IND 1 h prior to acetic acid injection and thereafter the observations were made. The number of stretchings occurred immediately within 30 min after the acetic acid injection was recorded. The results were evaluated by calculating the mean number of stretching per group and represented as % inhibition of stretching movement compared with the control group as below:
% Analgesic activity = (n − n′/n) ×100
where n is the average number of stretchings in the control group and n′ is the average number of stretching in the test group.
Assessment of central analgesic effect of MEGA and AEGA
The central analgesic effect of MEGA and AEGA was studied using Eddy's hot plate test.[18] The rats were placed on the hot plate maintained at 55 ± 1°C and time in seconds for the paw licking or jumping was recorded as the reaction time. Rats, which have shown basal reaction time period below 6 s, were selected and used for the screening. Pentazocine (5 mg/kg) was administered i.p. as standard drug. Rats were pre-treated with vehicle or MEGA or AEGA or pentazocine. The activity of the rats on the hot plate was closely observed and the reaction time (latency period) was recorded in seconds as the time taken for the animal to react to the thermal pain by licking its paw or attempting to jump out.
Assessment of anti-inflammatory activity
The anti-inflammatory activity was assessed using carrageenan-induced paw oedema in the rat model.[19] Acute paw oedema (acute inflammation) was induced by sub-plantar injection of carrageenan suspension (0.1 ml of 1% w/v freshly prepared suspension in normal saline) into the right hind paw of each rat. The left hind paw was injected with the same volume of 0.1 ml of normal saline. IND (10 mg/kg, orally) was used as standard drug.[20] Rats were pre-treated with vehicle or MEGA or AEGA or IND 1 h prior to carrageenan administration. The paw size was measured in mm using vernier caliper before (0 min) and at 1st and 3rd h after carrageenan administration. The percent inhibition was calculated by the following formula:
Percentage inhibition = (Ct − Co)control − (Ct − Co)treated/(Ct − Co)control
where Ct = Thickness of paw after carrageenan injection and Co = Thickness of paw before carrageenan injection
Statistical analysis
Data were analysed by one-way analysis of variance (ANOVA) and one-way repeat measure ANOVA, wherever applicable followed by Dunnett's post hoc test. The difference of P < 0.05 was considered as significant in all cases.
RESULTS
Phytochemical screening
The phytochemical screening of MEGA and AEGA revealed the presence of flavonoids, phenolic compounds, tannins, triterpenoid, sterols, mucilage, gum, protein, amino acid, carbohydrate and glycosides [Table 1].
Acute toxicity study
The limit test for acute toxicity showed that rats receiving MEGA and AEGA at 2000 mg/kg, orally, exhibited normal behavioural, neurological and autonomic profiles. No mortality or toxicity of any nature was observed during 72 h and thereafter up to 14 days observation. This indicated that approximate LD50 value of MEGA and AEGA is more than 2000 mg/kg, orally and it is relatively safe and non-toxic to rats.
Assessment of analgesic activity
Peripheral analgesic effect of MEGA and AEGA on acetic acid-induced writhing test in mice
One-way ANOVA revealed a significant (P < 0.0001) influence of MEGA and AEGA on acetic acid-induced writhing compared to control [Figure 1]. Post hoc Dunnett's test showed that MEGA and AEGA at 400 mg/kg, caused significant inhibition (P < 0.01) of writhing, but lower dose of the extracts did not exhibit inhibition of writhing (P > 0.05). The maximum inhibition for MEGA and AEGA at 400 mg/kg noted was 41.14% and 46.24%, respectively. The effects were comparable with that of reference standard, IND that also showed significant inhibition (36.04%) of writhing (P < 0.01) [Figure 1].
Figure 1.
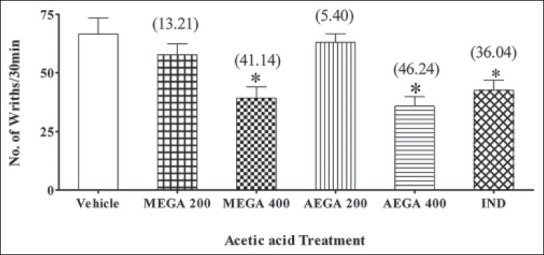
Mice were pre-treated with vehicle or methanolic extract of Grewia asiatica or aqueous extract of Grewia asiatica (200 and 400 mg/kg, orally) or indomethacin (10 mg/kg, orally) 1 h prior to acetic acid injection. The number of stretching's occurred immediately within 30 min after the acetic acid injection was recorded. The results are represented as % inhibition of stretching movement. Results are expressed as mean ± standard error of the mean. (n = 5 mice) *P < 0.01 versus control (one-way analysis of variance followed by post hoc Dunnett's test). Value in parenthesis shows percentage inhibition of writhings
Central analgesic effect of MEGA and AEGA on the hot plate test in mice
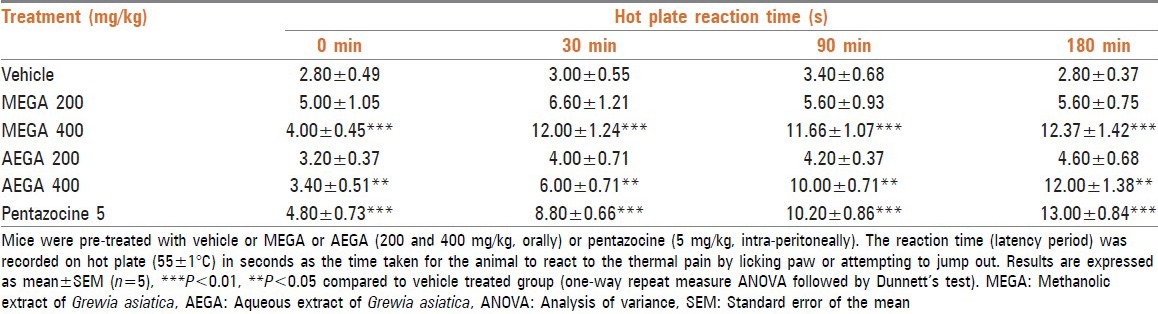
One-way repeat measure ANOVA analysis of the data generated showed significant (P < 0.0001) influence of MEGA and AEGA on the hot plate reaction time [Table 2]. Dunnett's test showed that MEGA and AEGA at 400 mg/kg, showed significant (P < 0.01, P < 0.05, respectively) increase in reaction time, but MEGA and AEGA at 200 mg/kg, did not show any significant effect on reaction time compared with the control group (P > 0.05). MEGA and AEGA at 400 mg/kg produced maximum effect and increased reaction time from 4.00 to 12.37 s and 3.40-12.00 s, respectively at the end of 180 min. The effect of MEGA and AEGA was comparable with pentazocine that showed a significant increase (P < 0.001) in reaction time from 4.80 s to 13.00 s at the end of 180 min [Table 2].
Table 2.
Assessment of central analgesic effect of MEGA and AEGA in hot plate test
Anti-inflammatory effect of MEGA and AEGA on carrageenan-induced rat paw oedema
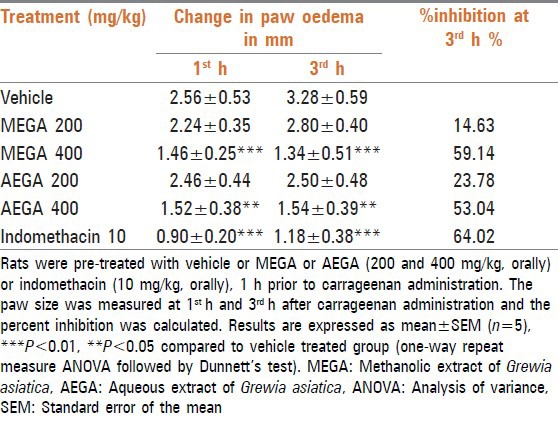
One-way repeat measure ANOVA analysis of the generated data showed significant (P < 0.0001) influence of MEGA and AEGA on carrageenan-induced inflammation [Table 3]. Dunnett's test indicated MEGA and AEGA at 400 mg/kg, caused significant (P < 0.01, P < 0.05 respectively) decrease in paw oedema as indicated by increased percent inhibition of paw oedema compared to vehicle. The percent inhibition was found to be 59.14 and 53.04%, respectively. MEGA and AEGA at 200 mg/kg did not show any significant effect (P > 0.05). IND at 10 mg/kg showed a significant and maximum reduction of paw oedema with 64.02% inhibition in comparison with the control group at 3rd h. The effects of MEGA and AEGA were comparable to IND that showed maximum decrease in paw oedema compared with vehicle [Table 3].
Table 3.
Assessment of anti-inflammatory activity of MEGA and AEGA in carrageenan-induced paw oedema
DISCUSSION
The results of the analgesic test experiments suggest that G. asiatica exhibits both peripheral and central analgesic properties. Acetic acid-induced writhing and hot plate test methods are useful techniques for the evaluation of peripherally and centrally acting analgesic drugs, respectively.[21,22] The MEGA and AEGA of the root bark of G. asiatica exhibited significant dose dependent inhibition of acetic acid-induced writhing in mice, comparable to IND while delayed the reaction times of rats in the ‘hot plate’ analgesia in a dose dependent fashion, comparable to pentazocine. The peripheral analgesic effect is generally mediated through inhibition of cyclooxygenase and/or lipoxygenase (and other inflammatory mediators) or inhibition of pain responses mediated by noiceptors peripherally[21,23] while the central analgesic action is mediated via inhibition of central pain receptors.[24,25] Therefore, it is possible that MEGA and AEGA may be showing analgesic effect through these mechanisms. An earlier study on the polyherbal formulation-PD-10 has shown it to possess antipyretic-analgesic activity in animals[7] and this formulation contains fruits of G. asiatica as an important constituent. In another ethnobotanical survey study,[26] it is mentioned that roots paste of G. asiatica is used in backache. Thus, the above studies support the analgesic effect of the extracts and strengthen the findings.
The carrageenan-induced rat paw oedema is a suitable test for evaluating anti-inflammatory drugs.[22] Development of the oedema in the paw of the rat after injection of carrageenan is a biphasic event. The initial phase observed during the 1st h is attributed to the release of histamine and serotonin while the second phase of oedema is due to the release of prostaglandins, proteases and lysosomes.[27,28] In carrageenan paw oedema model of acute inflammation, MEGA and AEGA showed significant dose dependent reduction in paw size both at 1st and 3rd h and elicited anti-inflammatory response comparable with IND. This might be due to the inhibition of the biphasic response induced by the carrageenan. It is possible that acute anti-inflammatory effect of MEGA and AEGA may involve multiple mechanisms like inhibition of either cyclo-oxygenase and/or lypo-oxygenase enzyme or inhibition of synthesis, release and action of above inflammatory mediators. This is in agreement with the generally believed thinking that herbal preparations may involve multiple sites in their actions.[29] In previous studies, several plants showing anti-inflammatory effects have been suggested to act through these mechanisms.[30,31,32] The recent review[33] on G. asiatica has also not cited any study indicating the analgesic and anti-inflammatory effect of root bark suggesting that these effects have not been studied yet. The present investigation is probably the first to report the analgesic and anti-inflammatory action of the root bark in experimental animals.
Several plant extracts showed analgesic and anti-inflammatory effect in animal models and their effects have been attributed to the presence of triterpenoids, alkaloids, glycosides, flavonoids, tannins, saponins and sterols.[34,35,36,37,38,39] The phytochemical screening of MEGA and AEGA revealed the presence of most of the above-mentioned phytochemicals. The previous studies have also reported the presence of these phytochemicals such as kaempherol, quercetin, naringenin and their glucosides, grewinol, taxasterol, sitosterol, citric acid, arabinose, etc., in one or different parts of G. asiatica.[7,33] Therefore, it is possible that the analgesic and anti-inflammatory activity of MEGA and AEGA can be due to the presence of these phytochemical constituents. As the extracts possess both analgesic and anti-inflammatory effect, it is suggested that roots bark may be very effective in rheumatism (painful inflammatory condition). However, the assessment of analgesic and anti-inflammatory needs to be carried out in animal models of rheumatism like Freund adjuvant or collagen induced arthritis to further substantiate its claim in treatment of rheumatism.
CONCLUSION
It can be concluded that the root bark of G. asiatica has potential analgesic and anti-inflammatory activity. This study substantiates the traditional use of bark of G. asiatica in rheumatism. Further bioactivity guided studies are required to determine the exact constituent responsible for the analgesic and anti-inflammatory effect of G. asiatica.
Footnotes
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- 1.Rathore B, Ali Mahdi A, Nath Paul B, Narayan Saxena P, Kumar Das S. Indian herbal medicines: Possible potent therapeutic agents for rheumatoid arthritis. J Clin Biochem Nutr. 2007;41:12–7. doi: 10.3164/jcbn.2007002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Singh A, Malhotra S, Subban R. Anti-inflammatory and analgesic agents from Indian medicinal plants. Int J Integr Biol. 2008;3:57–72. [Google Scholar]
- 3.Burke A, Smyth E, Fitzgerald GA. Analgesic-antipyretic agents: Pharmacotherapy of gout. In: Brunton LL, Lazo JS, Parker KL, editors. Goodman and Gilmans the Pharmacological Basis of Therapeutics. New York: McGraw Hill; 2006. pp. 671–85. [Google Scholar]
- 4.Mishra LC. Rheumatoid arthritis, osteoarthritis and gout. In: Mishra LC, editor. Scientific Basis for Ayurvedic Therapies. Boca Raton: CRC Press; 2004. pp. 167–83. [Google Scholar]
- 5.Hays WB. 2nd revised ed. Allahabad: Kitabistan; 1953. Fruit Growing in India. [Google Scholar]
- 6.Anonymous. Vol. 4. New Delhi: Council of Scientific and Industrial Research; 1956. The Wealth of India: Raw materials; pp. 260–6. [Google Scholar]
- 7.Gupta M, Shaw BP, Mukherjee A. Studies on antipyretic-analgesic and ulcerogenic activity of polyherbal preparation in rats and mice. Int J Pharmacol. 2008;4:88–94. [Google Scholar]
- 8.Morton JF. Miami, FL: Julia Morton; 1987. Phalsa. Fruits of Warm Climate; pp. 276–7. [Google Scholar]
- 9.Yadav AK. Phalsa: A potential new small fruit for Georgia. In: Janick J, editor. Perspectives on New Crops and New Uses. Alexandria: ASHS Press; 1999. pp. 348–52. [Google Scholar]
- 10.Kirtikar KR, Basu BD. 2nd ed. Dehradun: International Book Distributors; 1987. Indian Medicinal Plants; pp. 388–9. [Google Scholar]
- 11.Nadkarni KM. Mumbai: Popular Prakashan; 1976. Indian Materia Medica. Reprint of 3rd revised and enlarged ed; p. 593. [Google Scholar]
- 12.Kokate CK. 4th ed. Delhi: Vallabh Prakashan; 1997. Practical Pharmacognosy; pp. 107–11. [Google Scholar]
- 13.Singh S, Sharma KV, Ahaskar M, Sisodia R. Protective role of Grewia asiatica on blood after radiation exposure in mice. J Comp Integr Med. 2008;5:1–16. [Google Scholar]
- 14.Ahaskar M, Sharma KV, Singh S, Sisodia R. Radioprotective effect of the fruit extract of Grewia asiatica in Swiss albino mice against lethal dose of γ-irradiation. Asian J Exp Sci. 2007;21(Suppl 2):295–308. [Google Scholar]
- 15.Paris: Organization for Economic Cooperation and Development; 2001. OECD/OCDE Guidelines for Testing of Chemicals, Acute Oral Toxicity-Acute Toxic Class Method, No. 423; pp. 1–14. [Google Scholar]
- 16.Wu Y, Zhou C, Li X, Song L, Wu X, Lin W, et al. Evaluation of antiinflammatory activity of the total flavonoids of Laggera pterodonta on acute and chronic inflammation models. Phytother Res. 2006;20:585–90. doi: 10.1002/ptr.1918. [DOI] [PubMed] [Google Scholar]
- 17.Carvalho JC, Santos LS, Viana EP, de Almeida SS, Marciano E, Rodrigues M, et al. Anti-inflammatory and analgesic activities of the crude extracts from stem bark of Bauhinia guianensis. Pharm Biol. 1999;37:281–4. [Google Scholar]
- 18.Turner RA. Vol. 1. New York: Academic Press; 1965. Screening Methods in Pharmacology; pp. 114–5. [Google Scholar]
- 19.Winter CA, Risley EA, Nuss GW. Carrageenin-induced edema in hind paw of the rat as an assay for antiiflammatory drugs. Proc Soc Exp Biol Med. 1962;111:544–7. doi: 10.3181/00379727-111-27849. [DOI] [PubMed] [Google Scholar]
- 20.Barbieri EJ, Orzechowski RF, Rossi GV. Measurement of prostaglandin E2 in an inflammatory exudate: Effects of nonsteroidal anti-inflammatory agents. J Pharmacol Exp Ther. 1977;201:769–77. [PubMed] [Google Scholar]
- 21.Koster R, Anderson M, De Beer EJ. Acetic acid for analgesic screening. Fed Proc. 1959;18:418–20. [Google Scholar]
- 22.Adeyemi OO, Okpo SO, Okpaka O. The analgesic effect of the methanolic extract of Acanthus montanus. J Ethnopharmacol. 2004;90:45–8. doi: 10.1016/j.jep.2003.09.021. [DOI] [PubMed] [Google Scholar]
- 23.Duarte ID, Nakamura M, Ferreira SH. Participation of the sympathetic system in acetic acid-induced writhing in mice. Braz J Med Biol Res. 1988;21:341–3. [PubMed] [Google Scholar]
- 24.Sharma S, Jain NK, Kulkarni SK. Inhibition of COX-1 enzyme potentiates opoid induced antinociception in animal model of central noiception. Indian J Pharmacol. 2003;35:21–6. [Google Scholar]
- 25.Khanna N, Bhatia J. Antinociceptive action of Ocimum sanctum (Tulsi) in mice: Possible mechanisms involved. J Ethnopharmacol. 2003;88:293–6. doi: 10.1016/s0378-8741(03)00215-0. [DOI] [PubMed] [Google Scholar]
- 26.Tomar A. Folk medicinal uses of plant roots from Meerut District, Uttar Pradesh. Indian J Tradit Knowl. 2009;8:298–301. [Google Scholar]
- 27.Crunkhorn P, Meacock SC. Mediators of the inflammation induced in the rat paw by carrageenin. Br J Pharmacol. 1971;42:392–402. doi: 10.1111/j.1476-5381.1971.tb07124.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Vane JR. Inhibition of prostaglandin synthesis as a mechanism of action for aspirin-like drugs. Nat New Biol. 1971;231:232–5. doi: 10.1038/newbio231232a0. [DOI] [PubMed] [Google Scholar]
- 29.Huang TK, Ding ZZ, Zhao SX, Yan YQ, Xu GJ, Chen L, et al. Beijing: China Medico-Pharmaceutical Science and Technology Publishing House; 2001. Compendium of Modern Materia Medica; pp. 2473–4. [Google Scholar]
- 30.Telang RS, Chatterjee S, Varshneya C. Study on analgesic and anti-inflammatory activities of Vitex negundo Linn. Indian J Pharmacol. 1999;31:363–6. [Google Scholar]
- 31.Dhara AK, Suba V, Sen T, Pal S, Chaudhuri AK. Preliminary studies on the anti-inflammatory and analgesic activity of the methanolic fraction of the root extract of Tragia involucrata Linn. J Ethnopharmacol. 2000;72:265–8. doi: 10.1016/s0378-8741(00)00166-5. [DOI] [PubMed] [Google Scholar]
- 32.Zhang GQ, Huang XD, Wang H, Leung AK, Chan CL, Fong DW, et al. Anti-inflammatory and analgesic effects of the ethanol extract of Rosa multiflora Thunb. hips. J Ethnopharmacol. 2008;118:290–4. doi: 10.1016/j.jep.2008.04.014. [DOI] [PubMed] [Google Scholar]
- 33.Tripathi S, Chaurey M, Balasubramaniam A, Balakrishnan N. Grewia asiatica Linn. As a phytomedicine: A review. Res J Pharm Technol. 2010;3:1–3. [Google Scholar]
- 34.Seibert K, Zhang Y, Leahy K, Hauser S, Masferrer J, Perkins W, et al. Pharmacological and biochemical demonstration of the role of cyclooxygenase 2 in inflammation and pain. Proc Natl Acad Sci U S A. 1994;91:12013–7. doi: 10.1073/pnas.91.25.12013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Perazzo FF, Souza GH, Lopes W, Cardoso LG, Carvalho JC, Nanayakkara NP, et al. Anti-inflammatory and analgesic properties of water-ethanolic extract from Pothomorphe umbellata (Piperaceae) aerial parts. J Ethnopharmacol. 2005;99:215–20. doi: 10.1016/j.jep.2005.02.023. [DOI] [PubMed] [Google Scholar]
- 36.Gupta M, Mazumdar UK, Sivakumar T, Vamsi ML, Karki SS, Sambathkumar R, et al. Evaluation of anti-inflammatory activity of chloroform extract of Bryonia laciniosa in experimental animal models. Biol Pharm Bull. 2003;26:1342–4. doi: 10.1248/bpb.26.1342. [DOI] [PubMed] [Google Scholar]
- 37.Ferrándiz ML, Alcaraz MJ. Anti-inflammatory activity and inhibition of arachidonic acid metabolism by flavonoids. Agents Actions. 1991;32:283–8. doi: 10.1007/BF01980887. [DOI] [PubMed] [Google Scholar]
- 38.Della Loggia R, Tubaro A, Dri P, Zilli C, Del Negro P. The role of flavonoids in the antiinflammatory activity of Chamomilla recutita. Prog Clin Biol Res. 1986;213:481–4. [PubMed] [Google Scholar]
- 39.Ramaswamy S, Pillai NP, Gopalakrishnan V, Parmar NS, Ghosh MN. Analgesic effect of O-(beta-hydroxy ethyl) rutoside in mice. Indian J Exp Biol. 1985;23:219–20. [PubMed] [Google Scholar]


