Abstract
Steroid synthesis and metabolic pathways play important roles in the pathophysiology of PCOS, but until now there have been no studies on the methylation profiles of specific genes in steroid synthesis pathways that are known to be associated with PCOS. Here we used MassARRAY quantitative methylation analysis to determine the methylation levels of each CpG site or cluster in the promoters of EPHX1, SRD5A1, and CYP11A1 in 64 peripheral blood samples. We further examined the methylation level of EPHX1 in an independent cohort consisting of 116 people. Finally, we investigated the role of EPHX1 in steroidogenesis in the KGN cell line. For SRD5A1 and CYP11A1, there was no significant difference in methylation level between patients and controls. For EPHX1, however, the methylation levels of a few consecutive CpG sites and clusters were found to be significantly associated with PCOS. The methylation levels of a number of CpG clusters or sites were significantly lower in patients than in controls in the first cohort consisting of 64 people, such as clusters 13–14 (P<0.05), 15–16 (P<0.001), and 19–24 (P<0.001) and sites CpG_53 (P<0.01) and CpG_54 (P<0.05). Among differentiated methylation sites and clusters, the methylation levels of the CpG cluster 13–14 and CpG cluster 19–24 in PCOS patients were significantly lower than in controls in the second cohort of 116 people (P<0.05 for both). In addition, knockdown and overexpression experiments in KGN cells showed that EPHX1 can regulate estradiol concentrations, and this indicates a role for EPHX1 in steroidogenesis. Our study has demonstrated that methylation of the EPHX1 promoter might be associated with PCOS. This study provides direct evidence that methylation plays an important role in PCOS and demonstrates a novel role for EPHX1 in female reproduction.
Introduction
Polycystic ovary syndrome (PCOS) is the most common endocrine disorder among reproductive-age women and has an estimated global prevalence of approximately 5%–10%. It is a leading cause of female infertility and is associated with polycystic ovaries, hirsutism, obesity, and insulin resistance [1], [2]. The etiology of PCOS is complex and is not well understood. Some evidence, however, suggests that genetic and environmental factors contribute to the etiology of PCOS [3].
Epigenetic regulation of gene expression is important in human phenotype expression and has been implicated in various diseases [4]–[6]. However, the role of epigenetics in PCOS is only now being elucidated. Demethylation of the luteinizing hormone receptor (LHR) gene has been identified in a mouse model of PCOS [7], and skewed X-chromosome inactivation has been demonstrated in human PCOS patients [8], [9]. Xu et al. performed pioneering work on the global DNA methylation status of peripheral blood from PCOS patients and healthy women. Although no significance differences in DNA methylation were seen in the peripheral blood in their study, further study of specific genes in specific tissue was recommended [10]. We recently investigated the methylation level of follistatin in the peripheral blood of patients with PCOS but did not find any difference compared to the methylation level in controls [11].
CYP11A1, EPHX1, HSD17B6, CYP17, CYP19a1, and SRD5A1 are key genes in steroid synthesis and metabolic pathways that play important roles in the pathophysiology of PCOS [12]–[14]. Studies have investigated whether polymorphisms in these key genes confer PCOS susceptibility [15]–[18]. Using a CpG island searcher (http://www.uscnorris.com/cpgislands2/cpg.aspx), we found that SRD5A1, CYP11A1, and EPHX1 possess obvious CpG islands in their promoter regions.
In this study, we used MassARRAY techniques to determine whether there is an association between the methylation levels of the promoters of SRD5A1, CYP11A1, and EPHX1 and the etiology of PCOS. In addition, we used knockdown and overexpression of EPHX1 in cells to investigate the function of this gene in estradiol (E2) synthesis. Our results provide new evidence for the important role of epigenetics in the etiology of PCOS and provide new insights into the function of EPHX1.
Materials and Methods
2.1 Patients and Peripheral Blood Collection
In the first cohort, 32 PCOS patients (mean age (SEM) = 26.8±0.2 years) and 32 healthy controls (mean age (SEM) = 27.2±0.1 years) were recruited from the outpatient clinic of the Xi’an Fourth Hospital. In the second cohort, 49 PCOS patients (mean age (SEM) = 28±0.2 years) and 67 controls (mean age (SEM) = 28±0.1 years) were recruited from the outpatient clinic of the Shanghai Ninth Hospital. The diagnosis of PCOS was based on the following revised Rotterdam diagnostic criteria [19]: (i) oligo-ovulation and/or anovulation; (ii) clinical and/or biochemical signs of hyperandrogenism; and (iii) polycystic ovaries. Women who met at least two of these criteria were defined as having PCOS. Oral contraceptives or other hormonal medications had not been taken by any of the patients within the previous 3 months, and blood samples were taken from all of the patients after a 12-hour fast. The Fudan University Ethics Review Committee approved the study, and written informed consent was obtained from all participants.
2.2 DNA Preparation and CT Conversion
Genomic DNA from the peripheral blood of PCOS patients and controls was isolated using the QIAamp DNA Mini Kit (Qiagen, Hilden, Germany). Bisulfite conversion of the genomic DNA was performed with the EZ DNA CT Conversion Reagent, Zymo Research Corporation (Irvine, CA, US) as recommended by the manufacturer.
2.3 MassARRAY Quantitative Methylation Analysis
MassARRAY is a novel EpiTYPER assay for high-throughput analysis of DNA methylation patterns using matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF MS). This assay is a tool for the detection and quantitative analysis of DNA methylation using MALDI-TOF MS and the MassCLEAVE reagent, which enables base-specific (C/T) cleavage reactions [20]. PCRs were carried out in a volume of 5 µL containing 1 µL (10 ng) bisulfite-treated template DNA using PCR polymerase (Sequenom, Inc., San Diego, CA, USA). The protocol consisted of a 4 minute denaturing step at 95°C; 45 cycles of 95°C for 20 seconds, 56°C for 30 seconds, and 72°C for 1 minute. There was a final elongation step of 3 minutes at 72°C. For partially methylated CpG sites identified by sequencing analysis, quantitative methylation was further measured using the MassARRAY Compact System following the MassCLEAVE training protocol (Sequenom). This system uses bisulfite-converted genomic DNA and combines MassCLEAVE base-specific cleavage with MALDI-TOF mass spectrometry. The resulting methylation calls were analyzed with the EpiTYPER software package (Sequenom) to generate quantitative CpG methylation results (http://www.sequenom.com). All the primers used were listed in Table S1.
2.4 Cell Culture
Steroidogenic human granulosa-like KGN tumor cells were kindly donated by Dr. Fei Sun from the University of Science and Technology (Hefei, China) [21]. The cells were grown in HyClone DMEM high-glucose medium (HyClone Laboratories, Inc., Utah, USA) with 10% fetal bovine serum (Life Technologies, Inc., Carlsbad, CA) and 1% antibiotics (100 U/mL penicillin and 100 µg/mL streptomycin; HyClone Laboratories, Inc.) at 37°C under a 5% CO2 atmosphere. The culture medium was changed every other day.
2.5 Plasmid and siRNA
The pMCB3 overexpression vector was kindly donated by Dr. Shimin Zhao from Fudan University (Shanghai, China). Full-length human EPHX1 cDNA was amplified from the cDNA of the KGN cells and subcloned into the pMCB3 vector through BamH1 and Xho1 restriction sites. All constructs were confirmed by DNA sequencing. Chemically synthesized EPHX1 siRNA and scrambled or control siRNA were purchased from Shanghai GenePharma (Shanghai, China). The sequence of the siRNA is shown in Table S2. Annealed siRNA duplexes were resuspended in RNAse-free solution buffered to pH 7.4.
2.6 Transient Transfection and Hormone Analysis
KGN cells were plated in 24-well plates and grown to 70%–80% confluence. The culture medium was changed 3 h to 4 h before transfection. Transient transfection with plasmid DNA was performed with Lipofectamine 2000 (Invitrogen) following the manufacturer’s protocol. Transient transfection of siRNA was performed with HiPerFect (Qiagen Sciences, Germantown, MD). The culture medium was replaced with serum-free medium 24 h after transfection, and 10 nM testosterone (Biodee BioTech Corporation, Beijing, China) was added to each well for another 24 h. After this final incubation, the culture medium was collected and centrifuged at 1000×g for 5 min to prepare it for hormone analysis.
The remaining cells on the 24-well plate were lysed with Ripo (Biocolor BioScience & Technology Company, Shanghai, China) and centrifuged at 12,000×g for 10 minutes at 4°C and the supernatant was collected. Total protein concentration was measured with a BCA-100 Protein Quantitative Analysis Kit (Biocolor BioScience & Technology Company) and an Infinite M200 PRO reader (Tecan, Switzerland).
Concentrations of E2 and progesterone (P) in the culture medium were measured with the UniCel® DxI 800 Immunoassay System (Beckman Coulter, Inc., Brea, CA), which is an automated random-access chemilluminescence-based assay. The intra-assay and inter-assay coefficients of variation were less than 10% and 15%, respectively. The E2 concentration was further normalized to the total protein concentration in each well.
2.7 Statistical Analysis
The percent methylation of each CpG site or CpG cluster was expressed as the mean ± SEM. The data were analyzed using Student’s t-test with statistical significance at the level of P<0.05. Multiple testing was controlled by Bonferroni correction.
Results
3.1 Clinical Parameter Evaluation
The clinical characteristics of the PCOS patients and healthy controls are shown in Table 1 and Table 2, respectively. The age, height, weight, and serum concentrations of E2, P, and prolactin were comparable between the two groups, but serum luteinizing hormone, follicle stimulating hormone, and testosterone concentrations were significantly different. The PCOS group had much higher serum levels of luteinizing hormone and testosterone than the control group, and the serum level of follicle stimulating hormone was much lower in the PCOS group than control group.
Table 1. Clinical characteristics of healthy controls and PCOS patients in the first cohort (Control n = 32, PCOS n = 32).
| Variable | Control | PCOS |
| Age (years) | 26.8±0.2 | 27.2±0.1 |
| BMI (kg/m2) | 20.6±0.3 | 21.3±0.5 |
| LH (mlU/mL) | 2.8±0.3 | 6.2±1.2a |
| FSH (mlU/mL) | 7.3±0.6 | 5.9±0.3a |
| E2 (pg/mL) | 41.7±6.1 | 51.6±7.7a |
| PRL (ng/mL) | 23.0±1.5 | 20.5±1.4 |
| P (ng/mL) | 1.0±0.1 | 1.1±0.1 |
| T (ng/mL) | 0.3±0.0 | 0.6±0.1a |
Table 2. Clinical characteristics of healthy controls and PCOS patients in the second cohort (Control n = 49, PCOS n = 67).
| Variables | Control | PCOS |
| Age (year) | 28.0±0.2 | 28.0±0.1 |
| BMI (kg/m2) | 20.8±0.2 | 22.2±0.3a |
| LH (mIU/mL) | 3.1±0.1 | 5.3±0.5a |
| FSH (mIU/mL) | 6.9±0.3 | 5.9±0.2a |
| E2 (pg/mL) | 44.4±4.7 | 48.0±3.6a |
| PRL (ng/mL) | 26.2±1.6 | 26.0±2.1 |
| P (ng/mL) | 1.2±0.2 | 1.5±0.4 |
| T (ng/mL) | 0.3±0.0 | 0.4±0.0a |
Data represent the mean ± SEM;
BMI: body mass index, LH: luteinizing hormone, FSH: follicle stimulating hormone, E2: estradiol, PRL: prolactin, P: progesterone, T: testosterone;
p<0.001 compared with the control group, p values are determined by Student’s t-test.
3.2 MassARRAY Quantitative Methylation Analysis of the EPHX1, SRD5A1, and CYP11A1 Promoters
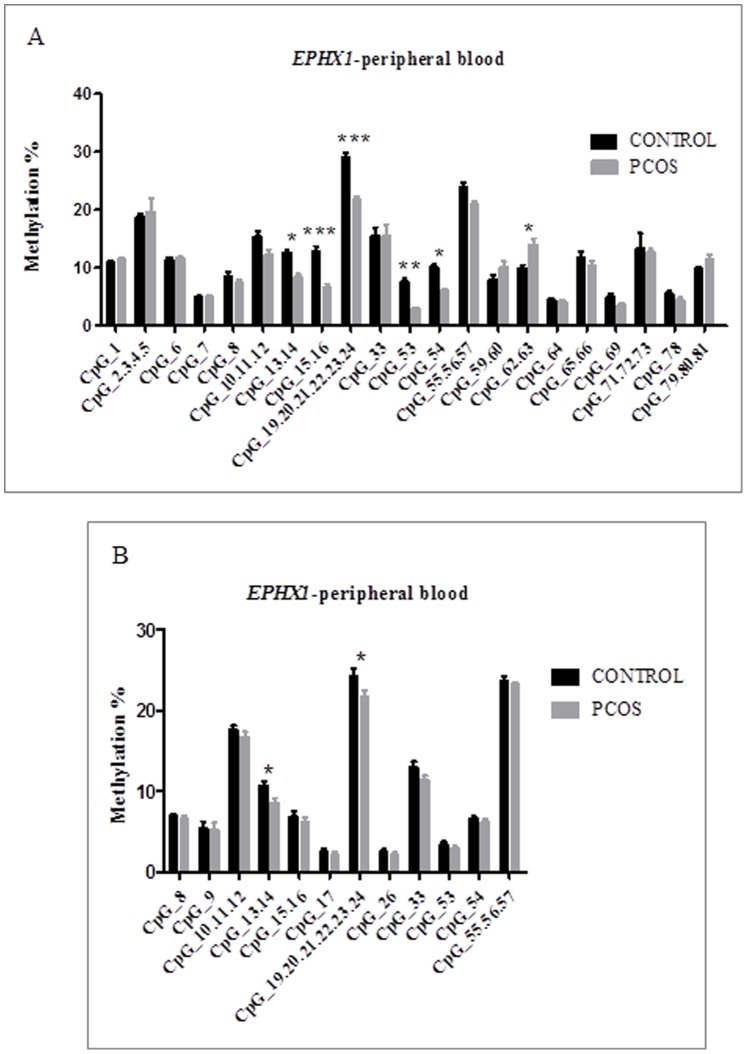
We first investigated the methylation level of each CpG site or cluster in the promoters of EPHX1, SRD5A1, and CYP11A1 in 64 peripheral blood DNA samples (32 PCOS patients and 32 healthy controls). For SRD5A1 and CYP11A1, there were no significant difference in methylation of any promoter CpG site or cluster between PCOS patients and healthy women (Figure S1). The promoter of EPHX1 contained 81 CpG sites (Figure S3), and the EpiTYPER software was used to design three pairs of primers as shown in Figure 1 and Table S1. There were significant differences in methylation levels of some CpG sites or clusters in the EPHX1 promoter between PCOS patients and controls. As shown in Figure 2, the average methylation level of the CpG clusters 13–14, 15–16, and 19–24 was 18% in controls and 12% in PCOS patients, and the average methylation level of the CpG sites 53 and 54 and CpG cluster 55–57 was 14% in controls and 12% in PCOS patients (P<0.05) (Table 3 and Figure 2A).
Figure 1. The schematic diagram of the CpG sites in the EPHX1 promoter.
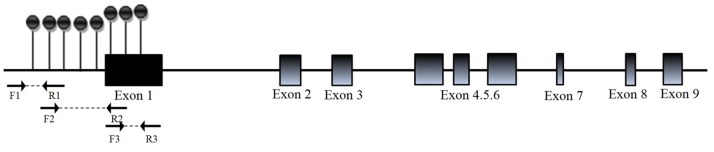
CpG sites are depicted by lollipop markers. Binding sites for the forward and reverse primers are shown as arrows below the diagram. The number of lollipop markers is not indicative of the number of detected CpG sites, and 47 informative CpG sites were identified in the EPHX1 promoter region.
Figure 2. The mean methylation levels for each CpG site of the EPHX1 promoter in PCOS patients and healthy women in the first cohort consisting of 64 peripheral blood samples (A) and in the second cohort consisting of 116 peripheral blood samples (B).
Values are mean ± SEM. *** represents p<0.001, ** represents p<0.01, * represents p<0.05.
Table 3. The methylation status (methylation %) of the CpG sites in the promoter region of EPHX1 between healthy women and PCOS patients in peripheral blood samples from the first cohort (Control n = 32, PCOS n = 32).
| CpG sites | Control | PCOS | Delta value | p-value |
| EPHX1 _CpG_1 | 10.8±0.4 | 11.4±0.3 | 0.6 | 0.30 |
| EPHX1 _CpG_2.3.4.5 | 18.7±0.5 | 19.6±2.4 | 0.9 | 0.71 |
| EPHX1 _CpG_6 | 11.3±0.5 | 11.7±0.3 | 0.4 | 0.52 |
| EPHX1 _CpG_8 | 4.9±0.2 | 5.0±0.3 | 0.1 | 0.63 |
| EPHX1 _CpG_9 | 8.5±0.9 | 7.5±0.4 | 1.0 | 0.29 |
| EPHX1 _CpG_10.11.12 | 15.3±0.9 | 12.3±0.8 | 3.0 | 0.12 |
| EPHX1 _CpG_13.14 | 12.5±0.5 | 8.4±0.5 | 4.1 | <0.05 |
| EPHX1 _CpG_15.16 | 12.8±0.8 | 6.6±0.5 | 6.2 | <0.001 |
| EPHX1 _CpG_19-24 | 28.9±0.9 | 21.6±0.6 | 7.3 | <0.001 |
| EPHX1 _CpG_25 | 0.7±0.4 | 0.7±0.2 | 0.0 | 0.85 |
| EPHX1 _CpG_27 | 2.2±0.6 | 2.1±0.4 | 0.1 | 0.80 |
| EPHX1 _CpG_34 | 14.9±1.5 | 18.0±3.2 | 3.1 | 0.38 |
| EPHX1 _CpG_53 | 7.3±0.6 | 2.8±0.3 | 4.5 | <0.01 |
| EPHX1 _CpG_54 | 10±0.6 | 6.0±0.3 | 4.0 | <0.05 |
| EPHX1 _CpG_55.56.57 | 24±0.7 | 21.0±0.4 | 3.0 | 0.16 |
| EPHX1 _CpG_59.60 | 8±0.8 | 10.0±1.0 | 2.0 | 0.13 |
| EPHX1 _CpG_61 | 4.7±0.3 | 4.9±0.6 | 0.2 | 0.73 |
| EPHX1 _CpG_62.63 | 9.9±0.5 | 13.8±1.2 | 3.9 | <0.05 |
| EPHX1 _CpG_64 | 4.5±0.3 | 4.1±0.4 | 0.4 | 0.39 |
| EPHX1 _CpG_65.66 | 11.7±0.9 | 10.4±0.7 | 1.3 | 0.27 |
| EPHX1 _CpG_67.68 | 2.6±0.2 | 2.8±0.4 | 0.2 | 0.71 |
| EPHX1 _CpG_69 | 4.9±0.5 | 3.5±0.3 | 1.4 | 0.19 |
| EPHX1 _CpG_71.72.73 | 13.3±2.6 | 12.7±0.6 | 0.6 | 0.81 |
| EPHX1 _CpG_78 | 5.4±0.6 | 4.2±0.6 | 1.2 | 0.15 |
| EPHX1 _CpG_79.80.81 | 9.7±0.5 | 11.5±0.7 | 1.8 | 0.22 |
Data represent the mean ± SEM, p values are determined by two-way ANOVA with Bonferroni post-tests.
To confirm this difference, we further validated the significance of the results in a second cohort consisting of 67 PCOS patients and 49 controls. We found that there were still two CpG clusters (CpG cluster 13–14 and the CpG cluster 19–24) that were significantly different between PCOS patients and controls. The average methylation levels of CpG cluster 13–14 were 10.7% in controls and 8.5% in PCOS patients (P<0.05), and the average methylation levels of CpG cluster 19–24 were 24.3% in controls and 21.5% in the PCOS patients (P<0.05) (Table 4 and Figure 2B).
Table 4. The methylation status (methylation %) of the promoter region covered by the second EPHX1 primer set in the peripheral blood samples from healthy women and PCOS patients in the second cohort (Control = 49, PCOS = 67).
| CpG sites | Control | PCOS | Delta value | p-value |
| EPHX1 _CpG_8 | 6.9±0.3 | 6.7±0.4 | 0.2 | 0.72 |
| EPHX1 _CpG_9 | 5.2±0.8 | 5.5±0.7 | 0.3 | 0.89 |
| EPHX1 _CpG_10.11.12 | 17.6±0.5 | 16.8±0.6 | 0.8 | 0.31 |
| EPHX1 _CpG_13.14 | 10.7±0.5 | 8.5±0.5 | 2.2 | <0.05 |
| EPHX1 _CpG_15.16 | 6.9±0.7 | 6.5±0.5 | 0.4 | 0.71 |
| EPHX1 _CpG_17 | 2.4±0.4 | 2.1±0.3 | 0.3 | 0.44 |
| EPHX1 _CpG_19-24 | 24.3±0.9 | 21.4±0.8 | 2.9 | <0.05 |
| EPHX1 _CpG_26 | 2.4±0.4 | 2.1±0.3 | 0.3 | 0.44 |
| EPHX1 _CpG_33 | 12.9±0.7 | 11.6±0.5 | 1.3 | 0.12 |
| EPHX1 _CpG_53 | 3.4±0.3 | 3.2±0.3 | 0.2 | 0.64 |
| EPHX1 _CpG_54 | 6.6±0.3 | 6.2±0.3 | 0.4 | 0.27 |
| EPHX1 _CpG_55.56.57 | 23.7±0.5 | 23±0.3 | 0.7 | 0.22 |
Data represent the mean ± SEM, p values are determined by two-way ANOVA with Bonferroni post-tests.
3.3 Role of EPHX1 in E2 Synthesis in KGN Cells
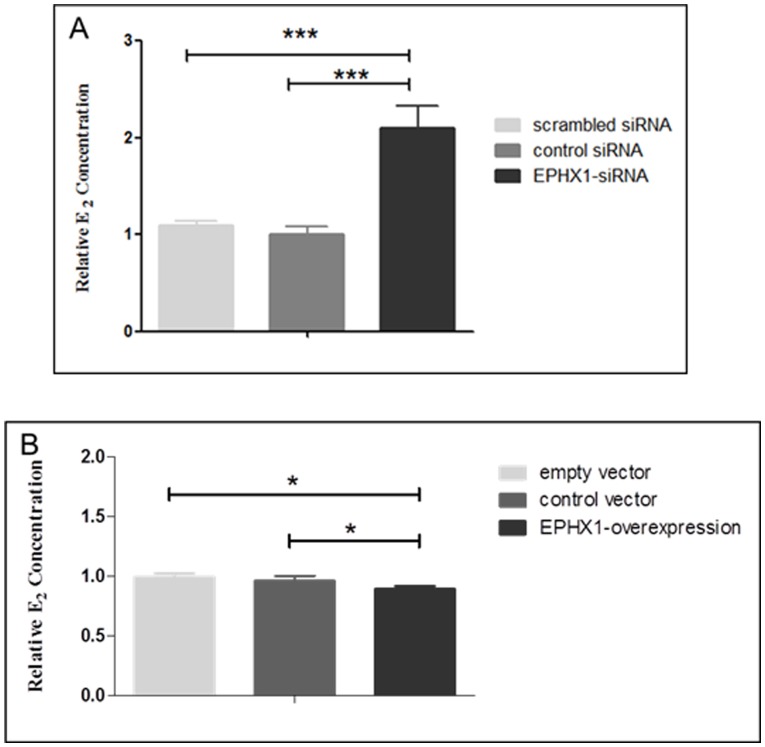
To investigate the function of EPHX1, KGN cells were transfected with either siRNA against EPHX1 or with a plasmid carrying the cDNA for EPHX1. As shown in Figure S2, the EPHX1 mRNA expression level was decreased when cells were transfected with the siRNA, and its mRNA expression level was elevated when the cells were transfected with the EPHX1 cDNA plasmid. Compared to cells transfected with negative control or scrambled siRNA, cells transfected with siRNA against EPHX1 had significantly higher E2 concentrations. On the contrary, when KGN cells were transfected with the EPHX1 cDNA plasmid, the E2 level was significantly lower than in cells transfected with control vector or empty vector ( Figure 3 ).
Figure 3. EPHX1 affects E2 synthesis in the KGN cell line.
The E2 level is increased when cells are transfected with EPHX1 siRNA (A). The E2 level is decreased when cells are transfected with the EPHX1 cDNA plasmid (B). *** represents p<0.001 and * represents p<0.05.
Discussion
Epigenetic changes have been found to be associated with common diseases such as type 2 diabetes, various kinds of cancers, and mental disorders such as schizophrenia and depression. Recent advances in epigenetics and epigenomics have provided evidence that epigenetic mechanisms may function as an interface between environmental factors and the genome [22].
PCOS is a multigenic disorder, and both genetic and environmental factors play a role in its etiology and pathophysiology. Xu et al conducted a pilot epigenetic study of DNA methylation in PCOS patients by comparing the global methylation levels between these patients and age-matched healthy controls [10]. Although there was no significant difference in the global methylation of peripheral leukocyte DNA between PCOS patients and controls, the authors of that study suggested that the methylation levels in specific genes and key tissues were worthy of further investigation. More recently, they have performed a genome-wide methylation analysis and identified the first specific alterations in the epigenome of infant and adult rhesus monkeys that have been exposed to androgenization. These results support the potential contributory role of epigenomic perturbations in the etiology of PCOS [23].
In the current study, we focused on three critical genes in steroid synthesis and metabolic pathways and systematically analyzed the methylation status of their promoters in the peripheral blood of PCOS patients and healthy controls. We found that the methylation level of a subset of CpG clusters in the promoter region of the EPHX1 gene was lower in PCOS patients than in controls. We also found that EPHX1 regulated E2 levels in KGN cells. Our study is the first to show that EPHX1 plays an important role in steroidogenesis in vitro and that the methylation status of its promoter has a close relationship with PCOS.
EPHX1 encodes epoxide hydrolase 1, which is a critical biotransformation enzyme that converts epoxides from the degradation of aromatic compounds into trans-dihydrodiols that can be conjugated and excreted from the body [24]. This protein plays a key role in detoxification processes and in the metabolism of endogenous and exogenous compounds. Several studies have shown that EPHX1 has an important effect on the female reproduction system and influences susceptibility to spontaneous abortion [25], ovarian cancer [26], and preeclampsia [27]. Furthermore, Korhonen et al demonstrated that two exonic single nucleotide polymorphisms of EPHX1 are associated with PCOS [28] suggesting that EPHX1 has a role in the etiology of PCOS.
This is direct evidence that EPHX1 can regulate E2 concentrations in vitro. The ability of granulosa cells to convert testosterone to E2 is an important indicator for PCOS because this process contributes to hyperandrogenism, which is the most important clinical symptom for PCOS diagnosis [29]–[32]. Taken together, our results suggest that the reduced methylation level of the EPHX1 promoter region in PCOS patients might activate EPHX1 expression, which in turn could suppress E2 production from testosterone and increase the risk of PCOS. Contrary to our in vitro findings, the in vivo E2 level is higher in PCOS patients than in the control. We speculate, therefore, that the in vivo E2 level might not be determined simply by EPHX1 gene expression. Instead, it might be affected by multiple factors, such as higher testosterone level or abnormalities in other metabolic traits.
Because RNA from peripheral blood was not obtained, we could not directly evaluate the EPHX1 expression level and this is a limitation of the current study. There is also evidence that methylation profiles might be different in different cell types and tissues. In this study, methylation of peripheral blood DNA was investigated. Although peripheral blood is not the target tissue for PCOS, it might provide useful clues for elucidating the disease mechanism and the changes in methylation that we have identified might also stimulate future research. Methylation levels in peripheral blood DNA have been widely investigated in various diseases, including diabetes [33], obesity [34], major depression [35], and various cancers [36]–[38]. This evidence for the role of methylation in other diseases strengthened the rationale for studying methylation in peripheral blood DNA in relation to PCOS.
Glossop et al demonstrated that DNA methylation pattern vary dramatically between blood cell types [39]. In our study, we cannot distinguish DNA from specific cell types in the whole blood. Thus, we cannot exclude the possibility that the differentiated methylation level might reflect differences in different cell types. However, the differentiated methylation level of the promoter region of the gene was validated by the two independent cohorts, and this increases the possibility that differentiated methylation levels in regions of the gene might reflect the difference between controls and PCOS patients.
Although differences observed in the study at significant loci in the larger cohort are low (<3%), the significant loci associated with PCOS are in the proximity of the transcription start site (TSS), which might have an important impact on corresponding gene expression. For example, several transcriptional factors such as GAGA-3, Adf-1, and StuAp might bind to differentially methylated CpG cluster 19–24. The change in methylation level of the CpG clusters might affect binding of those transcription factors, which in turn might have roles in the development and progression of PCOS. This, along with our results showing that E2 levels were affected by EPHX1 expression, provides convincing evidence that the methylation level of EPHX1 is associated with PCOS.
In summary, we have found significant differences in the methylation level of the EPHX1 promoter region in peripheral blood between PCOS patients and healthy controls. We have also shown that EPHX1 can regulate estradiol levels in granulosa cells. This is the first study to provide direct evidence that the methylation level of EPHX1 is associated with PCOS. More in-depth studies of the molecular mechanisms of methylation of EPHX1 in PCOS, and analysis of the methylation levels of other related genes, might yield new insights into the pathophysiology of this disorder.
Supporting Information
Comparison of mean methylation levels for each CpG site in the CYP11A1 (A) and SRD5A1 (B) promoters between PCOS patients and healthy women. Values are the mean ± SEM.
(TIF)
Relative mRNA expression of the EPHX1 gene in KGN cells transfected with EPHX1 siRNA and EPHX1 cDNA plasmid. The final data were normalized to human GAPDH. ** represents p<0.01 and * represents p<0.05.
(TIF)
Positions of the CpGs in the individual CpG clusters that we detected by MassARRAY. CpG clusters are labeled with yellow or green. Base C in CpGs are labeled with red.
(TIF)
The primer sets of methylation analysis used in this study.
(DOCX)
Sequence of EPHX1 -targeted siRNA.
(DOCX)
Funding Statement
This work was supported by the National Natural Science Foundation of China [grant numbers 81100467 and 81270747]; the Shanghai City Board of Education Scientific Research Innovation Key Projects [grant number 13ZZ001]; and the Fundamental Research Fund for Central University. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Teede H, Deeks A, Moran L (2010) Polycystic ovary syndrome: a complex condition with psychological, reproductive and metabolic manifestations that impacts on health across the lifespan. BMC medicine 8: 41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Dunaif A (1997) Insulin resistance and the polycystic ovary syndrome: mechanism and implications for pathogenesis. Endocrine reviews 18: 774–800. [DOI] [PubMed] [Google Scholar]
- 3. Ehrmann DA (2005) Polycystic ovary syndrome. The New England journal of medicine 352: 1223–1236. [DOI] [PubMed] [Google Scholar]
- 4. Esteller M (2008) Epigenetics in cancer. The New England journal of medicine 358: 1148–1159. [DOI] [PubMed] [Google Scholar]
- 5. Mandrekar P (2011) Epigenetic regulation in alcoholic liver disease. World journal of gastroenterology : WJG 17: 2456–2464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.North ML, Ellis AK (2011) The role of epigenetics in the developmental origins of allergic disease. Annals of allergy, asthma & immunology: official publication of the American College of Allergy, Asthma, & Immunology 106: 355–361; quiz 362. [DOI] [PubMed]
- 7. Zhu JQ, Zhu L, Liang XW, Xing FQ, Schatten H, et al. (2010) Demethylation of LHR in dehydroepiandrosterone-induced mouse model of polycystic ovary syndrome. Molecular human reproduction 16: 260–266. [DOI] [PubMed] [Google Scholar]
- 8. Hickey TE, Legro RS, Norman RJ (2006) Epigenetic modification of the X chromosome influences susceptibility to polycystic ovary syndrome. The Journal of clinical endocrinology and metabolism 91: 2789–2791. [DOI] [PubMed] [Google Scholar]
- 9. Calvo RM, Asuncion M, Sancho J, San Millan JL, Escobar-Morreale HF (2000) The role of the CAG repeat polymorphism in the androgen receptor gene and of skewed X-chromosome inactivation, in the pathogenesis of hirsutism. The Journal of clinical endocrinology and metabolism 85: 1735–1740. [DOI] [PubMed] [Google Scholar]
- 10. Xu N, Azziz R, Goodarzi MO (2010) Epigenetics in polycystic ovary syndrome: a pilot study of global DNA methylation. Fertility and sterility 94: 781–783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Sang Q, Zhang S, Zou S, Wang H, Feng R, et al. (2013) Quantitative analysis of follistatin (FST) promoter methylation in peripheral blood of patients with polycystic ovary syndrome. Reproductive biomedicine online 26: 157–163. [DOI] [PubMed] [Google Scholar]
- 12. Gharani N, Waterworth DM, Batty S, White D, Gilling-Smith C, et al. (1997) Association of the steroid synthesis gene CYP11a with polycystic ovary syndrome and hyperandrogenism. Hum Mol Genet 6: 397–402. [DOI] [PubMed] [Google Scholar]
- 13. Hattori N, Fujiwara H, Maeda M, Fujii S, Ueda M (2000) Epoxide hydrolase affects estrogen production in the human ovary. Endocrinology 141: 3353–65. [DOI] [PubMed] [Google Scholar]
- 14. Simoni M, Tempfer CB, Destenaves B, Fauser BC (2008) Functional genetic polymorphisms and female reproductive disorders: Part I: Polycystic ovary syndrome and ovarian response. Hum Reprod Update 14: 459–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Luense LJ, Veiga-Lopez A, Padmanabhan V, Christenson LK (2011) Developmental programming: gestational testosterone treatment alters fetal ovarian gene expression. Endocrinology 152: 4974–4983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Jones MR, Mathur R, Cui J, Guo X, Azziz R, et al. (2009) Independent confirmation of association between metabolic phenotypes of polycystic ovary syndrome and variation in the type 6 17 beta-hydroxysteroid dehydrogenase gene. The Journal of clinical endocrinology and metabolism 94: 5034–5038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Popp SL, Abele IS, Buck MB, Stope MB, Blok LJ, et al. (2010) Microsomal epoxide hydrolase expression in the endometrial uterine corpus is regulated by progesterone during the menstrual cycle. Journal of molecular histology 41: 111–119. [DOI] [PubMed] [Google Scholar]
- 18. Goodarzi MO, Shah NA, Antoine HJ, Pall M, Guo X, et al. (2006) Variants in the 5 alpha-reductase type 1 and type 2 genes are associated with polycystic ovary syndrome and the severity of hirsutism in affected women. The Journal of clinical endocrinology and metabolism 91: 4085–4091. [DOI] [PubMed] [Google Scholar]
- 19. March WA, Moore VM, Willson KJ, Phillips DI, Norman RJ, et al. (2010) The prevalence of polycystic ovary syndrome in a community sample assessed under contrasting diagnostic criteria. Human reproduction 25: 544–551. [DOI] [PubMed] [Google Scholar]
- 20. Ehrich M, Nelson MR, Stanssens P, et al. (2005) Quantitative high-throughput analysis of DNA methylation patterns by base-specific cleavage and mass spectrometry. Proc Natl Acad Sci USA 102: 15785–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Yao G, Yin M, Lian J, Tian H, Liu L, et al. (2010) MicroRNA-224 is involved in transforming growth factor-beta-mediated mouse granulosa cell proliferation and granulosa cell function by targeting Smad4. Molecular Endocrinology 24(3): 540–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Herceg Z, Vaissiere T (2011) Epigenetic mechanisms and cancer: an interface between the environment and the genome. Epigenetics: official journal of the DNA Methylation Society 6: 804–819. [DOI] [PubMed] [Google Scholar]
- 23. Xu N, Kwon S, Abbott DH, Geller DH, Dumesic DA, et al. (2011) Epigenetic mechanism underlying the development of polycystic ovary syndrome (PCOS)-like phenotypes in prenatally androgenized rhesus monkeys. PloS one 6: e27286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Hartsfield JK Jr, Sutcliffe MJ, Everett ET, Hassett C, Omiecinski CJ, et al. (1998) Assignment1 of microsomal epoxide hydrolase (EPHX1) to human chromosome 1q42.1 by in situ hybridization. Cytogenetics and cell genetics 83: 44–45. [DOI] [PubMed] [Google Scholar]
- 25. Wang X, Wang M, Niu T, Chen C, Xu X (1998) Microsomal epoxide hydrolase polymorphism and risk of spontaneous abortion. Epidemiology 9: 540–544. [PubMed] [Google Scholar]
- 26. Lancaster JM, Brownlee HA, Bell DA, Futreal PA, Marks JR, et al. (1996) Microsomal epoxide hydrolase polymorphism as a risk factor for ovarian cancer. Molecular carcinogenesis 17: 160–162. [DOI] [PubMed] [Google Scholar]
- 27. Zusterzeel PL, Peters WH, Visser W, Hermsen KJ, Roelofs HM, et al. (2001) A polymorphism in the gene for microsomal epoxide hydrolase is associated with pre-eclampsia. Journal of medical genetics 38: 234–237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Korhonen S, Romppanen EL, Hiltunen M, Helisalmi S, Punnonen K, et al. (2003) Two exonic single nucleotide polymorphisms in the microsomal epoxide hydrolase gene are associated with polycystic ovary syndrome. Fertility and sterility 79: 1353–1357. [DOI] [PubMed] [Google Scholar]
- 29. Amsterdam A, Keren-Tal I, Aharoni D, Dantes A, Land-Bracha A, et al. (2003) Steroidogenesis and apoptosis in the mammalian ovary. Steroids 68: 861–867. [DOI] [PubMed] [Google Scholar]
- 30. Goodarzi MO, Dumesic DA, Chazenbalk G, Azziz R (2011) Polycystic ovary syndrome: etiology, pathogenesis and diagnosis. Nature reviews Endocrinology 7: 219–231. [DOI] [PubMed] [Google Scholar]
- 31. Rosenfield RL (1999) Ovarian and adrenal function in polycystic ovary syndrome. Endocrinology and metabolism clinics of North America 28: 265–293. [DOI] [PubMed] [Google Scholar]
- 32. Franks S, Gharani N, Waterworth D, Batty S, White D, et al. (1998) Current developments in the molecular genetics of the polycystic ovary syndrome. Trends in endocrinology and metabolism: TEM 9: 51–54. [DOI] [PubMed] [Google Scholar]
- 33. Zhao J, Goldberg J, Bremner JD, Vaccarino V (2012) Global DNA methylation is associated with insulin resistance: a monozygotic twin study. Diabetes 61: 542–546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Xu X, Su S, Barnes VA, De Miguel C, Pollock J, et al. (2013) A genome-wide methylation study on obesity: Differential variability and differential methylation. Epigenetics 8: 522–533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Fuchikami M, Morinobu S, Segawa M, Okamoto Y, Yamawaki S, et al. (2011) DNA methylation profiles of the brain-derived neurotrophic factor (BDNF) gene as a potent diagnostic biomarker in major depression. PLoS One 6: e23881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Walters RJ, Williamson EJ, English DR, Young JP, Rosty C, et al. (2013) Association between hypermethylation of DNA repetitive elements in white blood cell DNA and early-onset colorectal cancer. Epigenetics 8: 748–755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Chimonidou M, Strati A, Malamos N, Georgoulias V, Lianidou ES (2013) SOX17 promoter methylation in circulating tumor cells and matched cell-free DNA isolated from plasma of patients with breast cancer. Clin Chem 59: 270–279. [DOI] [PubMed] [Google Scholar]
- 38. Woo HD, Kim J (2012) Global DNA hypomethylation in peripheral blood leukocytes as a biomarker for cancer risk: a meta-analysis. PLoS One 7: e34615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Glossop JR, Nixon NB, Emes RD, Haworth KE, Packham JC, et al.. (2013) Epigenome-wide profiling identifies significant differences in DNA methylation between matched-pairs of T- and B-lymphocytes from healthy individuals. Epigenetics 8: Epub ahead of print. [DOI] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Comparison of mean methylation levels for each CpG site in the CYP11A1 (A) and SRD5A1 (B) promoters between PCOS patients and healthy women. Values are the mean ± SEM.
(TIF)
Relative mRNA expression of the EPHX1 gene in KGN cells transfected with EPHX1 siRNA and EPHX1 cDNA plasmid. The final data were normalized to human GAPDH. ** represents p<0.01 and * represents p<0.05.
(TIF)
Positions of the CpGs in the individual CpG clusters that we detected by MassARRAY. CpG clusters are labeled with yellow or green. Base C in CpGs are labeled with red.
(TIF)
The primer sets of methylation analysis used in this study.
(DOCX)
Sequence of EPHX1 -targeted siRNA.
(DOCX)