Abstract
Background:
Cancer-associated fibroblasts (CAFs) activated by tumour cells are the predominant type of stromal cells in breast cancer tissue. The reciprocal effect of CAFs on breast cancer cells and the underlying molecular mechanisms are not fully characterised.
Methods:
Stromal fibroblasts were isolated from invasive breast cancer tissues and the conditioned medium of cultured CAFs (CAF-CM) was collected to culture the breast cancer cell lines MCF-7, T47D and MDA-MB-231. Neutralising antibody and small-molecule inhibitor were used to block the transforming growth factor-β (TGF-β) signalling derived from CAF-CM, which effect on breast cancer cells.
Results:
The stromal fibroblasts isolated from breast cancer tissues showed CAF characteristics with high expression levels of α-smooth muscle actin and SDF1/CXCL12. The CAF-CM transformed breast cancer cell lines into more aggressive phenotypes, including enhanced cell–extracellular matrix adhesion, migration and invasion, and promoted epithelial–mesenchymal transition (EMT). Cancer-associated fibroblasts secreted more TGF-β1 than TGF-β2 and TGF-β3, and activated the TGF-β/Smad signalling pathway in breast cancer cells. The EMT phenotype of breast cancer cells induced by CAF-CM was reversed by blocking TGF-β1 signalling.
Conclusion:
Cancer-associated fibroblasts promoted aggressive phenotypes of breast cancer cells through EMT induced by paracrine TGF-β1. This might be a common mechanism for acquiring metastatic potential in breast cancer cells with different biological characteristics.
Keywords: CAFs, TGF-β, EMT, breast cancer
Breast cancer is the most common malignancy in women and metastasis is the leading cause of death in breast cancer patients (Siegel et al, 2012). Owing to the heterogeneous nature of breast cancer, tumours with the same clinical and pathological status may differ in metastatic potential, and therefore lead to different prognoses (Fan et al, 2006). Metastasis of breast cancer cells requires decreased cell–cell adhesion, but increased cell–extracellular matrix (ECM) adhesion, and enhanced invasiveness and motility via secretion of matrix-degrading enzymes and cytoskeletal reorganisation (Kalluri and Weinberg, 2009). A well-recognised mechanism for initiating tumour cell invasive and metastatic behaviour is epithelial–mesenchymal transition (EMT), in which polarised epithelial breast cancer cells acquire a motile mesenchymal phenotype (Kalluri and Weinberg, 2009). However, the common mechanisms of induction of metastasis in breast cancers with different biological characteristics remain undefined. A better understanding of such mechanisms could lead to find universal molecular targets for anti-metastasis therapy.
The metastatic potential of breast cancer cells is closely dependent on the tumour microenvironment (Schedin and Borges, 2009). The fibroblast is the predominant stromal cell type in tumour microenvironment of heterogeneous breast cancer tissues. Tumour cells activate stromal fibroblast cells into cancer-associated fibroblasts (CAFs) through the stimulation of paracrine growth factors (Kalluri and Zeisberg, 2006; Tejada et al, 2006), and CAFs are more effective than normal stromal fibroblasts in promoting the cell survival (Martinez-Outschoorn et al, 2010), growth (Orimo et al, 2005) and progression (Giannoni et al, 2010) of cancer cells. Cancer-associated fibroblasts secrete a number of growth factors, including transforming growth factor-β1 (TGF-β1; Ao et al, 2007; Kojima et al, 2010), which is a strong extracellular signal regulating EMT in cancer cells (Sotgia et al, 2009; Wendt et al, 2010). Cancer-associated fibroblasts also secrete chemokine (C-X-C motif) ligand 12 (CXCL12)/stromal derived factor1 (SDF-1) to induce EMT of cancer cells (Al-Ansari et al, 2012). In addition, ECM, the non-cellular composition of the tumour microenvironment, is mainly synthesised by CAFs. It functions as a mediator of invasion and migration of cancer cells through ECM remodelling by ECM-degrading proteases secreted from cancer cells with the EMT phenotype (Giannoni et al, 2010; Qiao et al, 2010).
There is a consensus that CAFs can affect the metastatic behaviour of breast cancer cells (Lebret et al, 2007; Gao et al, 2010; Soon et al, 2013). However, it is unclear how CAFs affect the metastatic potential of breast cancers with different biological characteristics. In the present study, we demonstrated that CAFs could enhance the metastatic potential of breast cancer cells with different characteristics through EMT induced by paracrine TGF-β signalling. Different EMT programmes were activated in different breast cancer cells because of the different responses to CAF paracrine extracellular signalling.
Materials and Methods
Isolation and culture of stromal fibroblasts
To isolate stromal fibroblasts, primary cancer tissues were obtained from three female breast cancer patients at Tianjin Medical University Cancer Institute and Hospital (TMUCIH; Tianjin, China). These patients had undergone mastectomy but had not been treated with preoperative chemotherapy. The tissue specimens were divided into three parts for histopathological diagnosis, mRNA and protein extraction and isolation of stromal fibroblasts. The investigation and the use of specimens were approved by the Institutional Review Board of TMUCIH and written consent was obtained from participants.
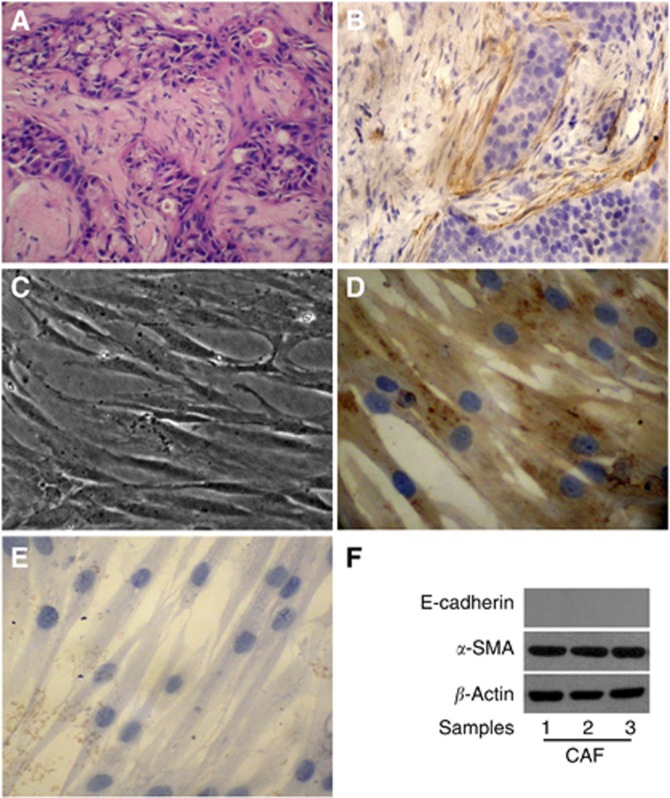
The breast cancer tissue specimens used for isolation of stromal fibroblasts were diagnosed as invasive ductal carcinoma with histological grade II and classified as luminal A subtype with oestrogen receptor-positive/progesterone receptor-positive/human epidermal growth factor receptor 2-negative. Importantly, the specimens were assessed by haematoxylin-eosin staining and immunohistochemical staining for α-smooth muscle actin (α-SMA) to confirm that there were stromal cells surrounding the cancer nests and distributed among the invasive cancer cells in the heterogeneous cancer tissue (Figure 1A and B).
Figure 1.
Stromal fibroblasts isolated from breast cancer tissues exhibit characteristics of CAFs. (A) H&E staining of paraffin-embedded breast cancer tissue sections ( × 400). There were stromal cells surrounding the cancer nests and distributed among the invasive cancer cells in the heterogeneous cancer tissue. (B) Activated fibroblast marker α-SMA expression in paraffin-embedded primary breast cancer tissue sections was detected by immunohistochemistry ( × 400). Stromal fibroblasts surrounding cancer cells highly expressed α-SMA, present characteristics of CAFs. (C) Morphological features of primary cultured stromal fibroblasts isolated from the primary breast cancer tissue ( × 200). (D) Mesenchymal marker vimentin expression in stromal fibroblasts grown on a coverslip was detected by immunohistochemistry ( × 400). The stromal fibroblasts expressed vimentin highly. (E) Epithelial marker E-cadherin expression in the stromal fibroblasts was detected by immunohistochemistry ( × 400). The stromal fibroblasts did not express E-cadherin. (F) The expression of α-SMA and E-cadherin in stromal fibroblasts isolated from different primary breast cancer tissues was detected by immunobotting. All the primary cultured stromal fibroblasts expressed α-SMA highly but did not express E-cadherin, presenting characteristics of CAFs.
The fresh tissues were sliced and digested with 160 μg ml−1 collagenase A (Sigma, St Louis, MO, USA) and 25 μg ml−1 hyaluronidase (Sigma) at 37 °C for 3 h. Then the cells were collected and cultured in Dulbecco's modified Eagle's medium nutrient mixture F12 (DMEM/F12; Invitrogen, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (FBS; Invitrogen) until cells grew into a confluent monolayer. After 2–3 passages, a unique homogeneity of stromal fibroblasts was formed. All the stromal fibroblasts used in the experiments were at less than 10 passages. To prepare conditioned medium of cultured CAFs (CAF-CM) for breast cancer cell treatments, stromal fibroblasts isolated from three tissue specimens were mixed and cultured for 48 h, then collected and centrifuged for 10 min at 5000 r.p.m. to remove cell debris.
Culture of breast cancer cell lines
Breast cancer cell lines MCF-7, T47D and MDA-MB-231, each with different biological characteristics and metastatic potential, were obtained from the American Type Culture Collection (ATCC; Manassas, VA, USA). Cells were cultured in RPMI-1640 (MCF-7 and MDA-MB-231) or DMEM/F12 (T47D) medium supplemented with 10% FBS, 100 units per ml penicillin and 100 μg ml−1 streptomycin at 37 °C. Cells in logarithmic growth phase or at 80% confluence were used for experiments. Breast cancer cell lines were treated with the CM of stromal fibroblasts, diluted with an equal volume of fresh medium for 2 days. An equal volume of complete culture medium was used as the control.
Cell–ECM adhesion assay
Breast cancer cells were plated in Matrigel-coated 12-well plates at 1 × 105 cells per well. The non-adherent cells were counted after being cultured for the appropriate time and the percentage of adherent cells was calculated as (1–non-adherent cells/total inoculated cells) × 100%.
Wound-healing assay
Breast cancer cells were plated in six-well plates at 5 × 105 cells per well to grow into a monolayer. A linear scratch/wound was made on cell monolayers with a sterile pipette. Photomicrographs were taken of live cells at × 100 magnification and the distance migrated was observed within an appropriate time.
Cell migration and invasion assay
The invasion and migration abilities of breast cancer cells in vitro were evaluated by Matrigel-coated Transwell and Transwell inserts (BD Biosciences, San Diego, CA, USA). 5 × 104 cells in 500 μl serum-free medium were added to the upper chamber, and medium containing 20% FBS was added into the lower chamber. The cells were left to invade the Matrigel for the appropriate time, the non-invading cells on the upper surface of the membrane were removed by wiping, and the invading cells were fixed and stained with a three-step stain set kit (Richard-Allan Scientific, Waltham, MA, USA). The number of invading or migrating cells was counted under a microscope in five predetermined fields for each membrane at × 400 magnification.
TGF-β signalling analysis
Breast cancer cells were cultured with CM of stromal fibroblasts containing 50 μg ml−1 TGF-β1 antibody (R&D Systems, Minneapolis, MN, USA) to neutralise TGF-β1, or 1.0 μmol ml−1 SB-431542 (Sigma) to block the TGF-β signalling of breast cancer cells.
Reverse transcription-quantitative PCR
Total RNA of tissues or cultured cells was isolated using TRIzol reagent (Invitrogen). Reverse transcription was performed using a First-strand cDNA Synthesis System (Invitrogen) according to the manufacturer's instructions. We quantified the transcripts of the housekeeping gene glyceraldehyde 3-phosphate dehydrogenase (GAPDH) as an internal mRNA quantity control, as previously described (Du et al, 2013). All primers and TaqMan probes for cDNA amplification of various target genes were designed and optimised using Oligo 6.0 software (Molecular Biology Insights, West Cascade, USA), and synthesised by Sangon Biological Engineering Technology & Services (Shanghai, China). Triplicate quantitative PCR reactions were performed for both target genes and the housekeeping gene using Platinum Quantitative PCR SuperMix-UDG System (Invitrogen) according to the manufacturer's instructions. The relative expression level of the target gene was calculated by normalising the cycle threshold (Ct) values of the target gene to the Ct values of GAPDH (ΔCt), and determined as 2−ΔCt (Du et al, 2013).
Immunohistochemistry
Breast cancer cells growing on sterile coverslips were fixed with 4% paraformaldehyde. Tissue specimens were formalin fixed and paraffin embedded. Immunohistochemistry staining of specimens was carried out using the primary antibodies anti-α-SMA (1A4; DAKO, Copenhagen, Denmark), anti-Vimentin (V9; Invitrogen) and anti-E-cadherin (4A2C7; Invitrogen); the secondary antibodies (GE Healthcare, Piscataway, NJ, USA) were used at appropriate concentration. Staining was performed with diaminobenzidine; hematoxylin was used for counterstaining.
Immunoblotting
Harvested cells were lysed with protein lysis buffer containing 20 mM Tris-HCl pH 7.4, 5 mM EDTA acid, 1% Triton X-100, 150 mM NaCl, 1% dithiothreitol and 1% protease inhibitor cocktail (Sigma). The proteins in the lysate were separated by SDS–PAGE and transferred to a polyvinyldifluoride membrane (Pierce, Rockford, IL, USA). The primary antibodies used were anti-Vimentin, anti-E-cadherin, anti-TGF-βRII (K105; Cell Signaling Technology, Danvers, Massachusetts, USA), anti-Smad2 (86F7; Cell Signaling Technology) and anti-pSmad2 (Phospho-Ser245/250/255; Cell Signaling Technology) at a dilution of 1 : 1000, β-actin (Sigma-Aldrich, St Louis, MO, USA) at a dilution of 1 : 3000, and the secondary antibody (GE Healthcare, Piscataway, NJ, USA) was used at a dilution of 1 : 2500. The protein blotting on the membranes was performed as previously described (Tan et al, 2011). The immunoreactive protein bands on the membranes were visualised using enhanced chemiluminescence reagents (GE Healthcare).
Immunofluorescence staining
Cells were seeded on the coverslips and cultured for 48 h, and then fixed in 4% paraformaldehyde for 20 min. After washing with PBS, the cells were permeabilised in 0.1% Triton X-100 for 15 min, washed with PBS and incubated with primary antibodies overnight at 4 °C. The cells were then washed with PBS and incubated with fluorescein isothiocyanate or phycoerythrin-conjugated secondary antibodies. Nuclei were stained using DAPI permamount. The protein expression was visualised using a fluorescence microscope.
Cytokine antibody array analysis
The TGFβs secreted from CAFs were analysed using a Cytokine Antibody Array (R&D Systems) in accordance with the manufacturer's instructions. Briefly, membranes were incubated with 1.0 ml of CAF-CM at room temperature for 1–2 h. Diluted biotin-conjugated antibodies were added to each membrane, which was then incubated at room temperature for 1–2 h. Diluted horseradish peroxidase-conjugated streptavidin was added to each membrane and incubated at room temperature for 2 h. The membranes were placed into the mixed Detection Buffer and exposed to X-ray film. The intensities of signals were quantified by ScanAlyze software (Michael Eisen, Lawrence Berkeley National Laboratory, Stanford University, Stanford, CA, USA). A positive control was used to normalise the results from the different membranes being compared. All samples were performed in duplicates.
Statistical analysis
Data are presented as mean±standard deviation. Student's t-test and rank sum test were used to compare the differences between the experimental group and control group. Statistical significance was defined as P<0.05.
Results
The stromal fibroblasts isolated from breast cancer tissues exhibit characteristics of CAFs
To characterise stromal fibroblasts in the microenvironment of breast cancer, we isolated and cultured stromal fibroblasts from primary breast cancer tissues. In the primary breast cancer tissue, we observed that there were stromal cells surrounding the cancer nests and distributed among the invasive cancer cells (Figure 1A), and some of them were active fibroblasts with positive α-SMA staining (Figure 1B). The stromal cells separated from primary breast cancer tissues were a heterogeneous mixture of a variety of cells in the primary culture, with fibroblasts being the main component. After passaging the primary culture cells, uniform fibroblasts started to grow (Figure 1C). The stromal fibroblasts showed positive staining for the mesenchymal marker vimentin (Figure 1D) and negative staining for the epithelial marker E-cadherin (Figure 1E). In particular, they expressed the activated myofibroblast marker α-SMA (Figure 1F) and CXCL12 (Supplementary Figure 1A). These results indicated that the isolated and cultured fibroblasts in vitro from breast cancer tissues maintained the features of CAFs.
To investigate whether the fibroblats at low passages cultured in vitro are retained features of CAFs, we detected the expression of E-cadherin, α-SMA and CXCL12 in CAFs at different low passages. The results showed that the expression levels of α-SMA and CXCL12 were similar in all the CAFs at different passages, and E-cadherin was not expressed in any CAFs at different passages (Supplementary Figure 1B and C), which indicated that the fibroblats at low passages cultured in vitro retained the features of CAFs.
CAFs enhanced aggressive behaviour of breast cancer cells
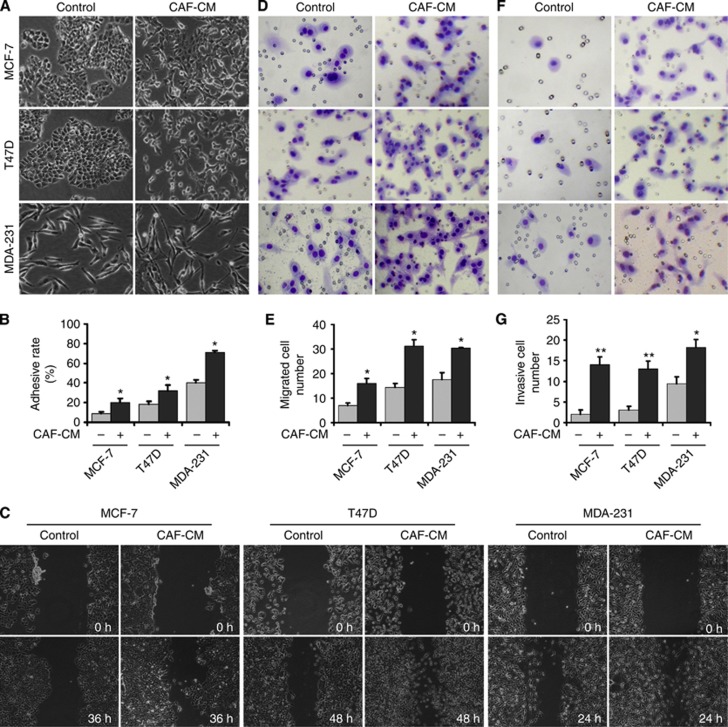
To investigate the effects of CAFs on breast cancer cells with different intrinsic characteristics, the CAF-CM was collected and used to culture breast cancer cell lines MCF-7, T47D and MDA-MB-231. The epithelial MCF-7 and T47D cells cultured with CAF-CM showed more spindle-like shape and cell scattering. The mesenchymal MDA-MB-231 cells cultured with CAF-CM were also changed to more fibroblast-like morphology (Figure 2A). All the three cell lines cultured with CAF-CM had enhanced cell–ECM adhesion (Figure 2B), migration (Figure 2C–E) and invasion (Figure 2F and G) compared with the control cells. All the above results suggested that CAF-secreted proteins could stimulate these different breast cancer cell lines to change their morphologies and phenotypes to have more metastatic potential.
Figure 2.
CAF-CM enhances the abilities of migration and invasion of breast cancer cell lines with different characteristics. (A) Morphological features of breast cancer cells. Compared with untreated control cells, the MCF-7 and T47D cells cultured in CAF-CM had fewer cell junctions, and scattered cells had elongated pseudopodia; pseudopodia in MDA-MB-231 cells in particular were significantly elongated. (B) Cell adhesion ability was measured using an cell–ECM adhesion assay. Compared with control cells, the adhesion rates of all the three cell lines cultured with CAF-CM were higher. (C) Cell migration ability was measured by a wound-healing assay. Compared with control cells, the migration distances of all the three cell lines cultured with CAF-CM were increased. (D, E) Cell migration ability was measured using a Transwell cell migration assay. The migration ability of all the three cell lines cultured with CAF-CM was significantly greater than that of the corresponding control cells. (F, G) Cell invasion ability was measured using a Transwell cell invasion assay. The invasion ability of all the three cell lines cultured with CAF-CM was significantly greater than that of the corresponding control cells. *P<0.05, **P<0.01.
CAFs induced EMT programming and phenotype in breast cancer cells
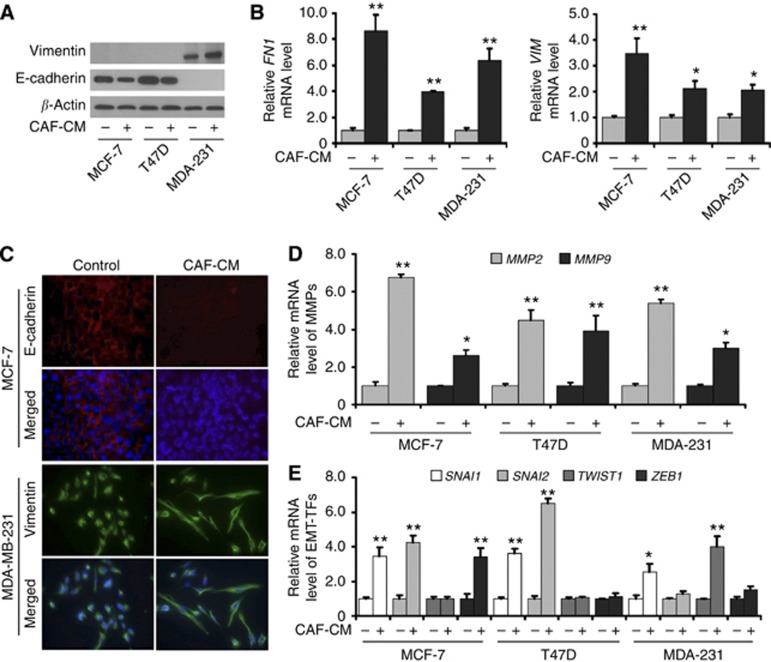
To investigate the changes of EMT phenotype induced by CAF-CM in breast cancer cell lines, we examined the expression of epithelial marker E-cadherin (CDH1), mesenchymal marker vimentin (VIM) and fibronectin 1 (FN1), as well as matrix metalloproteinase 2 (MMP2) and MMP9 in MCF-7, T47D and MDA-MB-231 cells incubated with CAF-CM. Results showed that cells cultured with CAF-CM had decreased expression of epithelial marker E-cadherin in MCF-7 and T47D cells, and increased expression of mesenchymal marker vimentin in MDA-MB-231 cells (Figure 3A and C). The expression levels of mesenchymal marker VIM, FN1, MMP2 and MMP9 were upregulated in all the three cell lines cultured with CAF-CM (Figure 3B and D). These results indicated that factors secreted by CAFs could induce the EMT phenotype in breast cancer cells with different characteristics.
Figure 3.
CAF-CM induces EMT programming and phenotype in different breast cancer cell lines. (A) The expression of epithelial marker E-cadherin and mesenchymal marker vimentin was detected by immunoblotting. Compared with control cells, the cells cultured with CAF-CM had decreased E-cadherin expression in MCF-7 and T47D cells, and increased vimentin expression in MDA-MB-231 cells. (B) The expression of mesenchymal marker VIM and FN1 was detected by reverse transcription (RT)-quantitative PCR (QPCR). Compared with control cells, VIM and FN1 expression levels were upregulated in all the three cell lines cultured with CAF-CM. (C) The expression of epithelial marker E-cadherin and mesenchymal marker vimentin was detected by immunofluorescence staining. Compared with control cells, the cells cultured with CAF-CM had decreased E-cadherin expression in MCF-7 cells and increased vimentin expression in MDA-MB-231 cells cultured with CAF-CM. Nuclei were visualised with DAPI staining (blue). (D) The expression of MMPs was detected by RT-QPCR. MMP2 and MMP9 expression levels were upregulated in all the three cell lines cultured with CAF-CM. (E) The expression of EMT-TFs was detected by RT-QPCR. SNAI1 expression levels were upregulated in all the three cancer cell lines cultured with CAF-CM. SNAI2 expression levels were upregulated in MCF-7 and T47D cells. TWIST1 expression levels were upregulated in MDA-MB-231 cells, and ZEB1 expression levels were upregulated in MCF-7 cells. *P<0.05, **P<0.01.
To investigate whether EMT programming was activated by CAF-secreted factors in these breast cancer cell lines with different characteristics, the expression of EMT-related transcription factors (EMT-TFs) snail homolog 1 (SNAI1), SNAI2, twist basic helix–loop–helix transcription factor 1 (TWIST1) and zinc-finger E-box-binding homeobox 1 (ZEB1) was measured in breast cancer cell lines cultured with CAF-CM. The results showed that SNAI1 expression levels were upregulated in all the three cell lines cultured with CAF-CM, SNAI2 expression levels were upregulated in epithelial MCF-7 and T47D cells but not in mesenchymal-like MDA-MB-231 cells, TWIST1 expression levels were upregulated in mesenchymal-like MDA-MB-231 cells but not in epithelial MCF-7 and T47D cells, and ZEB1 expression levels were upregulated only in MCF-7 cells (Figure 3E). These results indicated that CAF-secreted proteins induced the EMT phenotype through common and/or unique activation of EMT-TFs in breast cancer cell lines with different characteristics.
CAFs activated TGF-β/Smad signalling in breast cancer cells via CAF-secreted TGF-β1
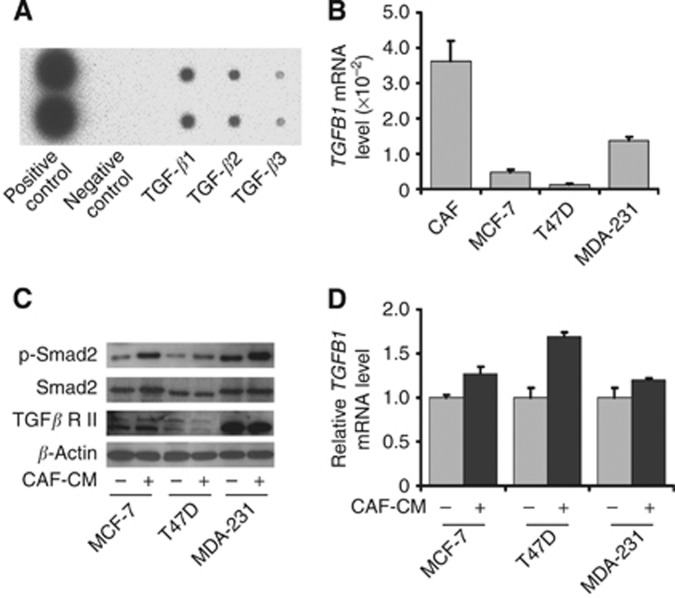
As TGF-β is a prominent EMT-inducer, we examined whether CAFs secreted any TGF isoform into CAF-CM. Our results showed that CAFs secreted significantly more TGF-β1 than TGF-β2 or TGF-β3 (Figure 4A). We then compared the mRNA expression of the TGF-β1-coding gene TGFB1 in CAFs and breast cancer cell lines. Our results showed that TGFB1 expression levels were significantly higher in CAFs than those in the breast cancer cell lines MCF-7, T47D and MDA-MB-231 (Figure 4B). These results indicated that TGF-β1 was mainly secreted by the CAFs in the co-culture of CAFs with breast cancer cells.
Figure 4.
CAFs activated TGF-β/Smad signalling of breast cancer cells through secreted TGF-β1. (A) TGF-β1, TGF-β2 and TGF-β3 secreted by CAFs in CAF-CM were detected by cytokine antibody array. Cancer-associated fibroblasts secreted significantly more TGF-β1 than TGF-β2 and TGF-β3. (B) TGFB1 expression in CAFs and breast cancer cell lines was detected by reverse transcription (RT)-quantitative PCR (QPCR). Cancer-associated fibroblasts expressed significantly higher TGFB1 expression levels than breast cancer cell lines MCF-7, T47D or MDA-MB-231. (C) The expression of TGF-βRII, total Smad2 and phosphorylated Smad2 in breast cancer cell lines cultured with CAF-CM was detected by immunoblotting. Phosphorylated Smad2 expression levels in MCF-7, T47D and MDA-MB-231 cells cultured with CAF-CM were significantly higher than their control cells, whereas total Smad2 and TGF-βRII were not significantly changed. (D) TGFB1 expression in breast cancer cells cultured with CAF-CM was detected by RT-QPCR. TGFB1 expression levels in MCF-7, T47D and MDA-MB-231 cell lines were not significantly affected by culturing in CAF-CM.
To investigate the effect of CAF-CM on activation of TGF-β signalling in breast cancer cell lines, we compared the expression levels of TGF-β receptor type II (TGF-βRII), total Smad2 and phosphorylated Smad2 in breast cancer cells cultured in CAF-CM or control medium. The results showed that CAF-CM significantly increased phosphorylated Smad2 in MCF-7, T47D and MDA-MB-231, whereas total Smad2 was not affected. Although TGF-βRII expression levels were different in the three cell lines, they were not affected by culturing with CAF-CM (Figure 4C). Transforming growth factor-β1 secretion by breast cancer cells was not affected neither (Figure 4D). These results indicate that CAFs activated TGF-β/Smad signalling pathway in breast cancer cells mainly through paracrine TGF-β1.
EMT programming and phenotype were reversed by blocking TGF-β1 in breast cancer cells cultured in CAF-CM
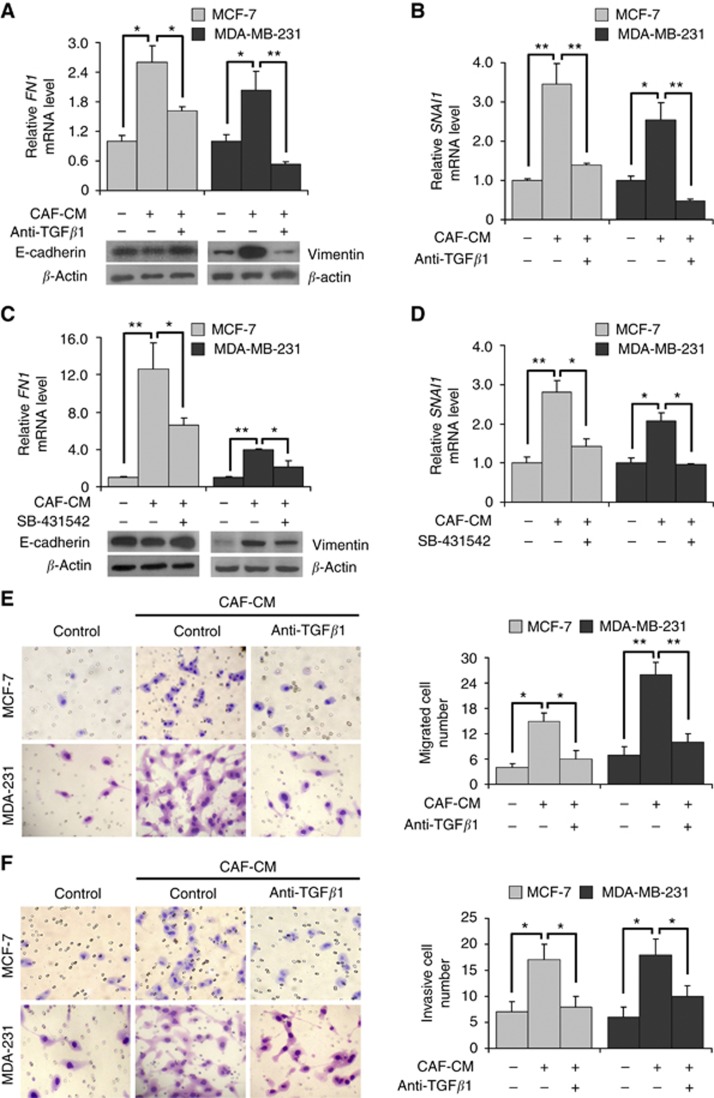
To further test the role of TGF-β1 signalling in EMT phenotype changes of breast cancer cells, TGF-β1 neutralising antibody and TGF-β signalling blocking reagent SB-431542 were added to CAF-CM for culturing epithelial cells MCF-7 and mesenchymal-like cells MDA-MB-231. Then, the expression of epithelial markers, mesenchymal markers and EMT-TFs were examined. After culturing in CAF-CM, the E-cadherin expression in MCF-7 cells was reduced, and vimentin expression in MDA-MB-231 cells was increased. Moreover, the increased vimentin in MDA-MB-231 cells and upregulated FN1 expression in both cell lines stimulated by CAF-CM were reversed by adding TGF-β1-neutralising antibody (Figure 5A). Similarly, the upregulated SNAI1 expression in MCF-7 and MDA-MB-231 cells was also reversed by adding TGF-β1-neutralising antibody (Figure 5B). Consistent with the effects of TGF-β1 neutralising antibody, adding SB-431542 in CAF-CM also reversed the reduced E-cadherin in MCF-7 cells, the increased vimentin in MDA-MB-231 cells, and the upregulated FN1 expression in both cell lines stimulated by CAF-CM (Figure 5C), as well as the upregulated SNAI1 expression in MCF-7 and MDA-MB-231 cells (Figure 5D). Furthermore, the enhanced abilities of migration and invasion of MCF-7 and MDA-MB-231 cells stimulated by CAF-CM were reversed by adding anti-TGF-β1 antibody (Figure 5E and F). These results indicated that TGF-β1 secreted from CAFs was an important regulator for activation of EMT programming and phenotype in breast cancer cells with different characteristics.
Figure 5.
EMT phenotype was reversed by blocking TGF-β1 in CAF-CM cultured breast cancer cells. (A) The expression of epithelial and mesenchymal markers in breast cancer cells was detected by immunoblotting or reverse transcription (RT)-quantitative PCR (QPCR), respectively. After the TGF-β1 signalling pathway was blocked by neutralising antibody, the reduced E-cadherin in MCF-7 cells, increased vimentin in MDA-MB-231 cells and upregulated FN1 expression levels stimulated by CAF-CM in all the three cell lines were reversed. (B) The expression of EMT-TFs was detected by RT-QPCR. After the TGF-β1 signalling pathway was blocked by neutralising antibody, the upregulated SNAI1 expression levels in MCF-7 and MDA-MB-231 cells stimulated by CAF-CM were reversed. (C) The expression of epithelial and mesenchymal markers in breast cancer cells was detected by immunoblotting or RT-QPCR, respectively. After the TGF-β1 signalling pathway was blocked by adding SB-431542 in CAF-CM, the reduced E-cadherin in MCF-7 cells, increased vimentin in MDA-MB-231 cells and upregulated FN1 expression levels in both cancer cells stimulated by CAF-CM were reversed. (D) The expression of EMT-TFs was detected by RT-QPCR. After the TGF-β1 signalling pathway was blocked by adding SB-431542 in CAF-CM, the upregulated SNAI1 expression levels in MCF-7 and MDA-MB-231 cells stimulated by CAF-CM were reversed. (E) Cell migration ability was measured using a Transwell cell migration assay. The migration ability of MCF-7 and MDA-MB-231 cells cultured in CAF-CM were reversed by adding anti-TGF-β1 antibody. (F) Cell invasion ability was measured using a Transwell cell invasion assay. The invasion ability of MCF-7 and MDA-MB-231 cells culturing in CAF-CM were reversed by adding anti-TGF-β1 antibody. *P<0.05, **P<0.01.
Discussion
Isolation of stromal fibroblasts from invasive cancer tissues followed by low-passaged cultures in vitro is the main approach to investigate the biological characteristics of CAFs and their effects on tumour cells (Erez et al, 2010). α-Smooth muscle actin is commonly used as a marker of myofibroblast formation to identify CAFs, and its expression is higher in fibroblasts derived from cancer tissues than in those derived from normal tissues (Orimo et al, 2005; Zhang et al, 2011; Horie et al, 2012). Evidence has shown that stromal fibroblasts isolated from cancer tissues such as breast cancer (Orimo et al, 2005; Lebret et al, 2007; Huang et al, 2010), prostate cancer (Giannoni et al, 2010), colon cancer (Nakagawa et al, 2004), lung cancer (Horie et al, 2012) and ovarian cancer (Zhang et al, 2011) retain the characteristics of CAFs, with high levels of α-SMA expression. As luminal A is the most common subtype of breast cancer, we isolated and cultured stromal fibroblasts from invasive luminal A subtype breast cancer tissues, and confirmed that the fibroblasts expressed high levels of α-SMA and CXCL12/SDF-1, which is a typical characteristic of CAFs. Moreover, CAFs at low passages cultured in vitro retained their original features, which was consistent with the Kalluri's report (Kalluri and Zeisberg, 2006).
There have been reports that CAFs could promote aggressive behaviour of breast cancer cells. Al-Ansari et al (2012) demonstrated that downregulation of p16INK4A transformed normal breast fibroblasts into CAFs, which in turn enhanced migration and invasion of MDA-MB-231 cells by inducing EMT in a CXCL12/SDF-1-dependent manner. Lebret et al (2007) showed that CAFs enhanced migration by promoting EMT of PMC42-LA cells. Gao et al (2010) found that CAFs promoted EMT of MCF-7 and MDA-MB-231 cells. Collectively, these results suggested that CAFs could promote migration and invasion through induction of EMT in various types of breast cancer cells, including luminal cells (MCF-7), basal-like cells (MDA-MB-231) and cells with both luminal epithelial and myoepithelial characteristics (PMC42-LA; Ackland et al, 2001). In this study, we demonstrated that factors secreted by CAFs enhanced the aggressive behaviours of cell–ECM adhesion, migration and invasion in both luminal (MCF-7 and T47D) and basal-like (MDA-MB-231) breast cancer cells by inducing or promoting EMT. This suggests that CAF-induced EMT might be the common mechanism underlying the acquisition of metastatic potential in different breast cancer subtypes.
EMT enables cancer cells to acquire the ability to complete various steps in the metastatic cascade, and this change in phenotype of epithelial cancer cells is induced by paracrine signals from their microenvironment (Scheel et al, 2011). It is known that CAFs can induce or promote EMT in a variety of cancers, however, the underlying molecular mechanisms are still not clear. Transforming growth factor-β is a prominent EMT-inducer (Zavadil et al, 2004; Wendt et al, 2010). Tumour cells can induce and maintain the EMT phenotype through autocrine secretion of TGF-β, and can also transform stromal fibroblasts into CAFs by paracrine TGF-β signalling (Fuyuhiro et al, 2011). Although CAFs can also secrete TGF-β (Rosenthal et al, 2004; Ao et al, 2007; Kojima et al, 2010; Franco et al, 2011), it is little known whether or how CAF-paracrine TGF-β induces EMT in tumour cells. By detection of the CAF-CM collected from CAFs cultured in vitro, we found that TGF-β1 was the major TGF-β family member secreted by CAFs. We also found that TGF-β1 was expressed by CAFs significantly more than by cancer cells, and stimulation of CAF-paracrine factors did not significantly increase expression of TGF-β1 and TGF-βRII in the breast cancer cell lines. Furthermore, by using a pharmacological inhibitor of the TGF-β receptor kinase to inhibit the receptors of tumour cells, and neutralising antibodies to inhibit secreted TGF-β1 by CAFs, we found that TGF-β1, mainly secreted by CAFs, could induce EMT in different subtypes of breast cancers.
Interestingly, we found that CAF-paracrine signalling induced EMT in breast cancer cells with different characteristics are underlying common and/or unique pleiotropically acting EMT-TFs. SNAI1 was activated by CAF-CM in the three breast cancer cell lines: epithelial/luminal A cells MCF-7 and T47D, and mesenchymal-like/basal-like cells MDA-MB-231. SNAI2 and ZEB1 were activated in epithelial/luminal A cells (SNAI2 in both MCF-7 and T47D, ZEB1 only in MCF-7) but not in mesenchymal-like/basal-like cells (MDA-MB-231). TWIST1 was activated in mesenchymal-like/basal-like cells (MDA-MB-231) but not in epithelial cells/luminal A cells (MCF-7 and T47D). This novel observation indicates that different EMT programmes are activated in different subtypes of breast cancer cells specific to their responses to CAF-paracrine signalling. Another interesting finding in our in vitro experiments is that exogenous TGF-β alone did not promote EMT of breast cancer cells (Supplementary Figure 2). Therefore, although TGF-β is a key modulator secreted by CAFs for promoting EMT of breast cancer cells with different biological characteristics, it needs co-factors to facilitate this process, and the exact mechanism remains to be further explored.
In summary, CAFs could stimulate morphological and phenotypic changes in various breast cancer subtype cells to enhance their metastatic potential through EMT induced by paracrine TGF-β1. This might be a common mechanism for metastasis among breast cancer cells with different biological characteristics.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (No. 30872518 and No. 81272357), the Major Program of Applied Basic Research Projects of Tianjin (No. 09JCZDJC19800 and No. 13JCZDJC30100).
The authors declare no conflict of interest.
Footnotes
Supplementary Information accompanies this paper on British Journal of Cancer website (http://www.nature.com/bjc)
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License.
Supplementary Material
References
- Ackland ML, Michalczyk A, Whitehead RH. PMC42, a novel model for the differentiated human breast. Exp Cell Res. 2001;263 (1:14–22. doi: 10.1006/excr.2000.5106. [DOI] [PubMed] [Google Scholar]
- Al-Ansari MM, Hendrayani SF, Shehata AI, Aboussekhra A. p16(INK4A) represses the paracrine tumor-promoting effects of breast stromal fibroblasts. Oncogene. 2012;32 (18:2356–2364. doi: 10.1038/onc.2012.270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ao M, Franco OE, Park D, Raman D, Williams K, Hayward SW. Cross-talk between paracrine-acting cytokine and chemokine pathways promotes malignancy in benign human prostatic epithelium. Cancer Res. 2007;67 (9:4244–4253. doi: 10.1158/0008-5472.CAN-06-3946. [DOI] [PubMed] [Google Scholar]
- Du X, Li XQ, Li L, Xu YY, Feng YM. The detection of ESR1/PGR/ERBB2 mRNA levels by RT-QPCR: a better approach for subtyping breast cancer and predicting prognosis. Breast Cancer Res Treat. 2013;138 (1:59–67. doi: 10.1007/s10549-013-2432-2. [DOI] [PubMed] [Google Scholar]
- Erez N, Truitt M, Olson P, Arron ST, Hanahan D. Cancer-associated fibroblasts are activated in incipient neoplasia to orchestrate tumor-promoting inflammation in an NF-kappaB-dependent manner. Cancer Cell. 2010;17 (2:135–147. doi: 10.1016/j.ccr.2009.12.041. [DOI] [PubMed] [Google Scholar]
- Fan C, Oh DS, Wessels L, Weigelt B, Nuyten DS, Nobel AB, van't Veer LJ, Perou CM. Concordance among gene-expression-based predictors for breast cancer. N Engl J Med. 2006;355 (6:560–569. doi: 10.1056/NEJMoa052933. [DOI] [PubMed] [Google Scholar]
- Franco OE, Jiang M, Strand DW, Peacock J, Fernandez S, Jackson RS, 2nd, Revelo MP, Bhowmick NA, Hayward SW. Altered TGF-beta signaling in a subpopulation of human stromal cells promotes prostatic carcinogenesis. Cancer Res. 2011;71 (4:1272–1281. doi: 10.1158/0008-5472.CAN-10-3142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuyuhiro Y, Yashiro M, Noda S, Kashiwagi S, Matsuoka J, Doi Y, Kato Y, Hasegawa T, Sawada T, Hirakawa K. Upregulation of cancer-associated myofibroblasts by TGF-beta from scirrhous gastric carcinoma cells. Br J Cancer. 2011;105 (7:996–1001. doi: 10.1038/bjc.2011.330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao MQ, Kim BG, Kang S, Choi YP, Park H, Kang KS, Cho NH. Stromal fibroblasts from the interface zone of human breast carcinomas induce an epithelial-mesenchymal transition-like state in breast cancer cells in vitro. J Cell Sci. 2010;123 (Pt 20:3507–3514. doi: 10.1242/jcs.072900. [DOI] [PubMed] [Google Scholar]
- Giannoni E, Bianchini F, Masieri L, Serni S, Torre E, Calorini L, Chiarugi P. Reciprocal activation of prostate cancer cells and cancer-associated fibroblasts stimulates epithelial-mesenchymal transition and cancer stemness. Cancer Res. 2010;70 (17:6945–6956. doi: 10.1158/0008-5472.CAN-10-0785. [DOI] [PubMed] [Google Scholar]
- Horie M, Saito A, Mikami Y, Ohshima M, Morishita Y, Nakajima J, Kohyama T, Nagase T. Characterization of human lung cancer-associated fibroblasts in three-dimensional in vitro co-culture model. Biochem Biophys Res Commun. 2012;423 (1:158–163. doi: 10.1016/j.bbrc.2012.05.104. [DOI] [PubMed] [Google Scholar]
- Huang M, Li Y, Zhang H, Nan F. Breast cancer stromal fibroblasts promote the generation of CD44+CD24- cells through SDF-1/CXCR4 interaction. J Exp Clin Cancer Res. 2010;29:80. doi: 10.1186/1756-9966-29-80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalluri R, Weinberg RA. The basics of epithelial-mesenchymal transition. J Clin Invest. 2009;119 (6:1420–1428. doi: 10.1172/JCI39104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalluri R, Zeisberg M. Fibroblasts in cancer. Nat Rev Cancer. 2006;6 (5:392–401. doi: 10.1038/nrc1877. [DOI] [PubMed] [Google Scholar]
- Kojima Y, Acar A, Eaton EN, Mellody KT, Scheel C, Ben-Porath I, Onder TT, Wang ZC, Richardson AL, Weinberg RA, Orimo A. Autocrine TGF-beta and stromal cell-derived factor-1 (SDF-1) signaling drives the evolution of tumor-promoting mammary stromal myofibroblasts. Proc Natl Acad Sci USA. 2010;107 (46:20009–20014. doi: 10.1073/pnas.1013805107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lebret SC, Newgreen DF, Thompson EW, Ackland ML. Induction of epithelial to mesenchymal transition in PMC42-LA human breast carcinoma cells by carcinoma-associated fibroblast secreted factors. Breast Cancer Res. 2007;9 (1:R19. doi: 10.1186/bcr1656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinez-Outschoorn UE, Trimmer C, Lin Z, Whitaker-Menezes D, Chiavarina B, Zhou J, Wang C, Pavlides S, Martinez-Cantarin MP, Capozza F, Witkiewicz AK, Flomenberg N, Howell A, Pestell RG, Caro J, Lisanti MP, Sotgia F. Autophagy in cancer associated fibroblasts promotes tumor cell survival: Role of hypoxia, HIF1 induction and NFkappaB activation in the tumor stromal microenvironment. Cell Cycle. 2010;9 (17:3515–3533. doi: 10.4161/cc.9.17.12928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakagawa H, Liyanarachchi S, Davuluri RV, Auer H, Martin EW, Jr, de la Chapelle A, Frankel WL. Role of cancer-associated stromal fibroblasts in metastatic colon cancer to the liver and their expression profiles. Oncogene. 2004;23 (44:7366–7377. doi: 10.1038/sj.onc.1208013. [DOI] [PubMed] [Google Scholar]
- Orimo A, Gupta PB, Sgroi DC, Arenzana-Seisdedos F, Delaunay T, Naeem R, Carey VJ, Richardson AL, Weinberg RA. Stromal fibroblasts present in invasive human breast carcinomas promote tumor growth and angiogenesis through elevated SDF-1/CXCL12 secretion. Cell. 2005;121 (3:335–348. doi: 10.1016/j.cell.2005.02.034. [DOI] [PubMed] [Google Scholar]
- Qiao B, Johnson NW, Gao J. Epithelial-mesenchymal transition in oral squamous cell carcinoma triggered by transforming growth factor-beta1 is Snail family-dependent and correlates with matrix metalloproteinase-2 and -9 expressions. Int J Oncol. 2010;37 (3:663–668. doi: 10.3892/ijo_00000715. [DOI] [PubMed] [Google Scholar]
- Rosenthal E, McCrory A, Talbert M, Young G, Murphy-Ullrich J, Gladson C. Elevated expression of TGF-beta1 in head and neck cancer-associated fibroblasts. Mol Carcinog. 2004;40 (2:116–121. doi: 10.1002/mc.20024. [DOI] [PubMed] [Google Scholar]
- Schedin P, Borges V. Breaking down barriers: the importance of the stromal microenvironment in acquiring invasiveness in young women's breast cancer. Breast Cancer Res. 2009;11 (2:102. doi: 10.1186/bcr2235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scheel C, Eaton EN, Li SH, Chaffer CL, Reinhardt F, Kah KJ, Bell G, Guo W, Rubin J, Richardson AL, Weinberg RA. Paracrine and autocrine signals induce and maintain mesenchymal and stem cell states in the breast. Cell. 2011;145 (6:926–940. doi: 10.1016/j.cell.2011.04.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siegel R, Naishadham D, Jemal A. Cancer statistics, 2012. CA Cancer J Clin. 2012;62 (1:10–29. doi: 10.3322/caac.20138. [DOI] [PubMed] [Google Scholar]
- Soon PS, Kim E, Pon CK, Gill AJ, Moore K, Spillane AJ, Benn DE, Baxter RC. Breast cancer-associated fibroblasts induce epithelial-to-mesenchymal transition in breast cancer cells. Endocr Relat Cancer. 2013;20 (1:1–12. doi: 10.1530/ERC-12-0227. [DOI] [PubMed] [Google Scholar]
- Sotgia F, Del Galdo F, Casimiro MC, Bonuccelli G, Mercier I, Whitaker-Menezes D, Daumer KM, Zhou J, Wang C, Katiyar S, Xu H, Bosco E, Quong AA, Aronow B, Witkiewicz AK, Minetti C, Frank PG, Jimenez SA, Knudsen ES, Pestell RG, Lisanti MP. Caveolin-1-/- null mammary stromal fibroblasts share characteristics with human breast cancer-associated fibroblasts. Am J Pathol. 2009;174 (3:746–761. doi: 10.2353/ajpath.2009.080658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tan LD, Xu YY, Yu Y, Li XQ, Chen Y, Feng YM. Serum HER2 level measured by dot blot: a valid and inexpensive assay for monitoring breast cancer progression. PLoS One. 2011;6 (4:e18764. doi: 10.1371/journal.pone.0018764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tejada ML, Yu L, Dong J, Jung K, Meng G, Peale FV, Frantz GD, Hall L, Liang X, Gerber HP, Ferrara N. Tumor-driven paracrine platelet-derived growth factor receptor alpha signaling is a key determinant of stromal cell recruitment in a model of human lung carcinoma. Clin Cancer Res. 2006;12 (9:2676–2688. doi: 10.1158/1078-0432.CCR-05-1770. [DOI] [PubMed] [Google Scholar]
- Wendt MK, Smith JA, Schiemann WP. Transforming growth factor-beta-induced epithelial-mesenchymal transition facilitates epidermal growth factor-dependent breast cancer progression. Oncogene. 2010;29 (49:6485–6498. doi: 10.1038/onc.2010.377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zavadil J, Cermak L, Soto-Nieves N, Bottinger EP. Integration of TGF-beta/Smad and Jagged1/Notch signalling in epithelial-to-mesenchymal transition. EMBO J. 2004;23 (5:1155–1165. doi: 10.1038/sj.emboj.7600069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Y, Tang H, Cai J, Zhang T, Guo J, Feng D, Wang Z. Ovarian cancer-associated fibroblasts contribute to epithelial ovarian carcinoma metastasis by promoting angiogenesis, lymphangiogenesis and tumor cell invasion. Cancer Lett. 2011;303 (1:47–55. doi: 10.1016/j.canlet.2011.01.011. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.