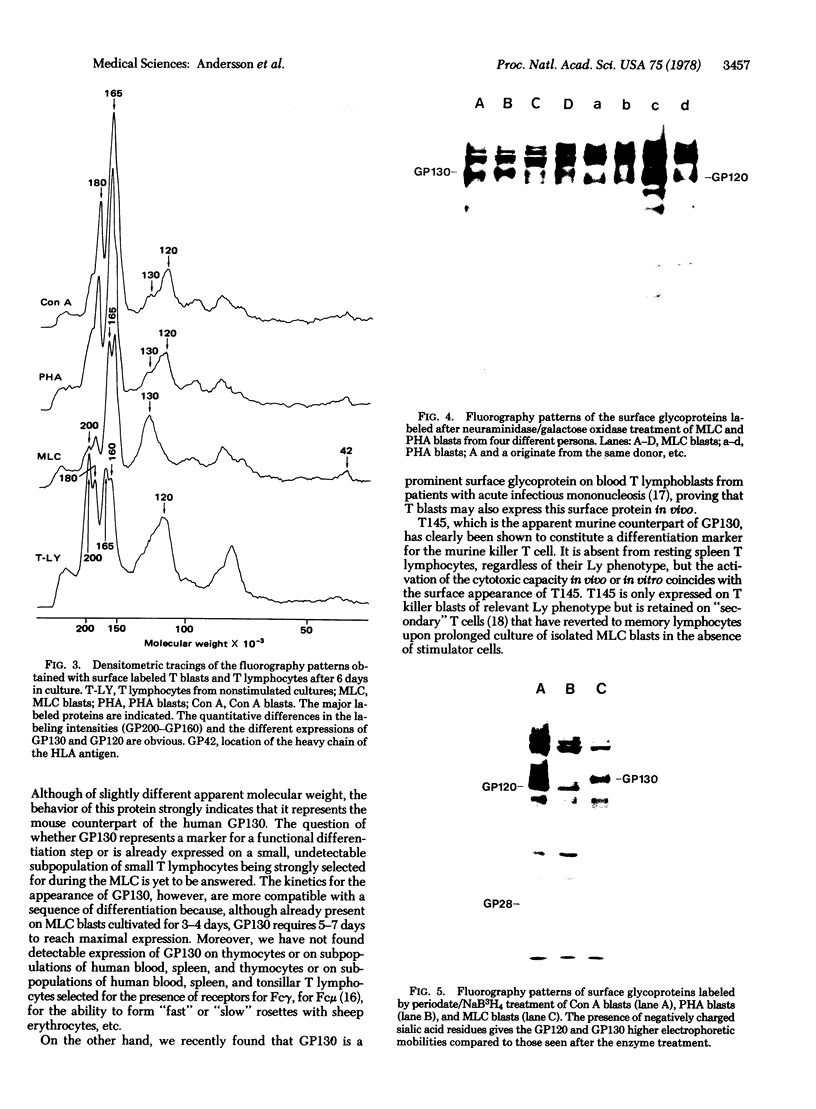
Abstract
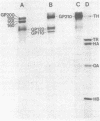
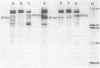
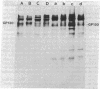
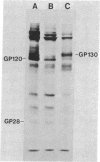
We have analyzed the surface glycoproteins of resting and in vitro activated human T lymphocytes by the galactose oxidase/NaB3H4 and the periodate/NaB3H4 labeling techniques. The labeled glycoproteins were separated by polyacrylamide slab gel electrophoresis and visualized by fluorography. A "new" glycoprotein with an apparent molecular weight of 130,000 (GP130) was strongly labeled on alloantigen-activated T blasts but only weakly or not at all on mitogen-stimulated T blasts and resting T lymphocytes. These results demonstrate that human T cells, as earlier found in the mouse system, express different surface molecules in relation to the particular mode of activation and stage of differentiation.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Andersson L. C., Gahmberg C. G. Blood lymphoblasts in infectious mononucleosis express the surface glycoprotein pattern of killer T cells. Clin Immunol Immunopathol. 1978 May;10(1):41–46. doi: 10.1016/0090-1229(78)90007-7. [DOI] [PubMed] [Google Scholar]
- Andersson L. C., Gahmberg C. G., Nilsson K., Wigzell H. Surface glycoprotein patterns of normal and malignant human lymphoid cells. I. T cells T blasts and leukemic T cell lines. Int J Cancer. 1977 Nov 15;20(5):702–707. doi: 10.1002/ijc.2910200509. [DOI] [PubMed] [Google Scholar]
- Andersson L. C., Häyry P. Clonal isolation of alloantigen-reactive T-cells and characterization of their memory functions. Transplant Rev. 1975;25:121–162. doi: 10.1111/j.1600-065x.1975.tb00728.x. [DOI] [PubMed] [Google Scholar]
- Andersson L. C., Wasastjerna C., Gahmberg C. G. Different surface glycoprotein patterns on human T-, B- and leukemic-lymphocytes. Int J Cancer. 1976 Jan 15;17(1):40–46. doi: 10.1002/ijc.2910170107. [DOI] [PubMed] [Google Scholar]
- Bonner W. M., Laskey R. A. A film detection method for tritium-labelled proteins and nucleic acids in polyacrylamide gels. Eur J Biochem. 1974 Jul 1;46(1):83–88. doi: 10.1111/j.1432-1033.1974.tb03599.x. [DOI] [PubMed] [Google Scholar]
- Cantor H., Boyse E. A. Functional subclasses of T-lymphocytes bearing different Ly antigens. I. The generation of functionally distinct T-cell subclasses is a differentiative process independent of antigen. J Exp Med. 1975 Jun 1;141(6):1376–1389. doi: 10.1084/jem.141.6.1376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gahmberg C. G., Andersson L. C. Selective radioactive labeling of cell surface sialoglycoproteins by periodate-tritiated borohydride. J Biol Chem. 1977 Aug 25;252(16):5888–5894. [PubMed] [Google Scholar]
- Gahmberg C. G., Hakomori S. I. External labeling of cell surface galactose and galactosamine in glycolipid and glycoprotein of human erythrocytes. J Biol Chem. 1973 Jun 25;248(12):4311–4317. [PubMed] [Google Scholar]
- Gahmberg C. G., Häyry P., Andersson L. C. Characterization of surface glycoproteins of mouse lymphoid cells. J Cell Biol. 1976 Mar;68(3):642–653. doi: 10.1083/jcb.68.3.642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jandinski J., Cantor H., Tadakuma T., Peavy D. L., Pierce C. W. Separation of helper T cells from suppressor T cells expressing different Ly components. I. Polyclonal activation: suppressor and helper activities are inherent properties of distinct T-cell subclasses. J Exp Med. 1976 Jun 1;143(6):1382–1390. doi: 10.1084/jem.143.6.1382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kimura A. K., Wigzell H. Cell surface glycoproteins of murine cytotoxic T lymphocytes. I. T 145, a new cell surface glycoprotein selectively expressed on Ly 1-2+ cytotoxic T lymphocytes. J Exp Med. 1978 May 1;147(5):1418–1434. doi: 10.1084/jem.147.5.1418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller R. G., Phillips R. A. Separation of cells by velocity sedimentation. J Cell Physiol. 1969 Jun;73(3):191–201. doi: 10.1002/jcp.1040730305. [DOI] [PubMed] [Google Scholar]
- Moretta L., Webb S. R., Grossi C. E., Lydyard P. M., Cooper M. D. Functional analysis of two human T-cell subpopulations: help and suppression of B-cell responses by T cells bearing receptors for IgM or IgG. J Exp Med. 1977 Jul 1;146(1):184–200. doi: 10.1084/jem.146.1.184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nilsson K., Andersson L. C., Gahmberg C. G., Wigzell H. Surface glycoprotein patterns of normal and malignant human lymphoid cells. II. B cells, B blasts and Epstein-Barr virus (EBV)-positive and -negative B lymphoid cell lines. Int J Cancer. 1977 Nov 15;20(5):708–716. doi: 10.1002/ijc.2910200510. [DOI] [PubMed] [Google Scholar]
- Paul W. E., Benacerraf B. Functional specificity of thymus- dependent lymphocytes. Science. 1977 Mar 25;195(4284):1293–1300. doi: 10.1126/science.320663. [DOI] [PubMed] [Google Scholar]
- Svedmyr E., Jondal M. Cytotoxic effector cells specific for B Cell lines transformed by Epstein-Barr virus are present in patients with infectious mononucleosis. Proc Natl Acad Sci U S A. 1975 Apr;72(4):1622–1626. doi: 10.1073/pnas.72.4.1622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wigzell H., Sundqvist K. G., Yoshida T. O. Separation of cells according to surface antigens by the use of antibody-coated columns. Fractionation of cells carrying immunoglobulins and blood group antigen. Scand J Immunol. 1972;1(1):75–87. doi: 10.1111/j.1365-3083.1972.tb03737.x. [DOI] [PubMed] [Google Scholar]