Abstract
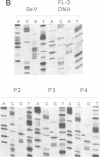
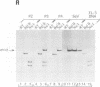
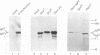
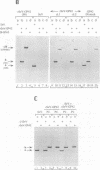
We have recovered infectious Sendai virus (SeV) from full-length cDNA (FL-3) by transfecting this cDNA and pGEM plasmids expressing the nucleocapsid protein (NP), phosphoprotein and large proteins into cells infected with a vaccinia virus which expresses T7 RNA polymerase. These cells were then injected into chicken eggs, in which SeV grows to very high titers. FL-3 was marked with a BglII site in the leader region and an NsiI site (ATGCAT) in the 5' nontranslated region of the NP gene, creating a new, out-of-frame, 5' proximal AUG. All the virus stocks generated eventually removed this impediment to NP expression, by either point mutation or recombination between FL-3 and pGEM-NP. The recovery system was found to be highly recombinogenic. Even in the absence of selective pressure, one in 20 of the recombinant SeV generated had exchanged the NP gene of FL-3 with that of pGEM-NP. When a fifth plasmid containing a new genomic 3' end without the presumably deleterious BglII site was included as another target for recombination, the new genomic 3' end was found in the recombinant SeV in 12 out of 12 recoveries. Using this approach, a novel copy-back nondefective virus was generated which interferes with wild-type virus replication.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ball L. A. Fidelity of homologous recombination in vaccinia virus DNA. Virology. 1995 Jun 1;209(2):688–691. doi: 10.1006/viro.1995.1305. [DOI] [PubMed] [Google Scholar]
- Baudin F., Bach C., Cusack S., Ruigrok R. W. Structure of influenza virus RNP. I. Influenza virus nucleoprotein melts secondary structure in panhandle RNA and exposes the bases to the solvent. EMBO J. 1994 Jul 1;13(13):3158–3165. doi: 10.1002/j.1460-2075.1994.tb06614.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calain P., Curran J., Kolakofsky D., Roux L. Molecular cloning of natural paramyxovirus copy-back defective interfering RNAs and their expression from DNA. Virology. 1992 Nov;191(1):62–71. doi: 10.1016/0042-6822(92)90166-m. [DOI] [PubMed] [Google Scholar]
- Calain P., Roux L. Functional characterisation of the genomic and antigenomic promoters of Sendai virus. Virology. 1995 Sep 10;212(1):163–173. doi: 10.1006/viro.1995.1464. [DOI] [PubMed] [Google Scholar]
- Calain P., Roux L. The rule of six, a basic feature for efficient replication of Sendai virus defective interfering RNA. J Virol. 1993 Aug;67(8):4822–4830. doi: 10.1128/jvi.67.8.4822-4830.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Curran J., Boeck R., Kolakofsky D. The Sendai virus P gene expresses both an essential protein and an inhibitor of RNA synthesis by shuffling modules via mRNA editing. EMBO J. 1991 Oct;10(10):3079–3085. doi: 10.1002/j.1460-2075.1991.tb07860.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Enami M., Palese P. High-efficiency formation of influenza virus transfectants. J Virol. 1991 May;65(5):2711–2713. doi: 10.1128/jvi.65.5.2711-2713.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engelhorn M., Stricker R., Roux L. Molecular cloning and characterization of a Sendai virus internal deletion defective RNA. J Gen Virol. 1993 Jan;74(Pt 1):137–141. doi: 10.1099/0022-1317-74-1-137. [DOI] [PubMed] [Google Scholar]
- Evans D. H., Stuart D., McFadden G. High levels of genetic recombination among cotransfected plasmid DNAs in poxvirus-infected mammalian cells. J Virol. 1988 Feb;62(2):367–375. doi: 10.1128/jvi.62.2.367-375.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuerst T. R., Niles E. G., Studier F. W., Moss B. Eukaryotic transient-expression system based on recombinant vaccinia virus that synthesizes bacteriophage T7 RNA polymerase. Proc Natl Acad Sci U S A. 1986 Nov;83(21):8122–8126. doi: 10.1073/pnas.83.21.8122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heggeness M. H., Scheid A., Choppin P. W. the relationship of conformational changes in the Sendai virus nucleocapsid to proteolytic cleavage of the NP polypeptide. Virology. 1981 Oct 30;114(2):555–562. doi: 10.1016/0042-6822(81)90235-x. [DOI] [PubMed] [Google Scholar]
- Hidaka Y., Kanda T., Iwasaki K., Nomoto A., Shioda T., Shibuta H. Nucleotide sequence of a Sendai virus genome region covering the entire M gene and the 3' proximal 1013 nucleotides of the F gene. Nucleic Acids Res. 1984 Nov 12;12(21):7965–7973. doi: 10.1093/nar/12.21.7965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holland J. J., De La Torre J. C., Steinhauer D. A. RNA virus populations as quasispecies. Curr Top Microbiol Immunol. 1992;176:1–20. doi: 10.1007/978-3-642-77011-1_1. [DOI] [PubMed] [Google Scholar]
- Jacques J. P., Hausmann S., Kolakofsky D. Paramyxovirus mRNA editing leads to G deletions as well as insertions. EMBO J. 1994 Nov 15;13(22):5496–5503. doi: 10.1002/j.1460-2075.1994.tb06884.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jordan J. A., Youngner J. S. Dominance of temperature-sensitive phenotypes. II. Vesicular stomatitis virus mutants from a persistent infection interfere with shut-off of host protein synthesis by wild-type virus. Virology. 1987 Jun;158(2):407–413. doi: 10.1016/0042-6822(87)90212-1. [DOI] [PubMed] [Google Scholar]
- Lawson N. D., Stillman E. A., Whitt M. A., Rose J. K. Recombinant vesicular stomatitis viruses from DNA. Proc Natl Acad Sci U S A. 1995 May 9;92(10):4477–4481. doi: 10.1073/pnas.92.10.4477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luytjes W., Krystal M., Enami M., Parvin J. D., Palese P. Amplification, expression, and packaging of foreign gene by influenza virus. Cell. 1989 Dec 22;59(6):1107–1113. doi: 10.1016/0092-8674(89)90766-6. [DOI] [PubMed] [Google Scholar]
- Lynch S., Kolakofsky D. Ends of the RNA within Sendai virus defective interfering nucleocapsids are not free. J Virol. 1978 Nov;28(2):584–589. doi: 10.1128/jvi.28.2.584-589.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parks R. J., Evans D. H. Effect of marker distance and orientation on recombinant formation in poxvirus-infected cells. J Virol. 1991 Mar;65(3):1263–1272. doi: 10.1128/jvi.65.3.1263-1272.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parks R. J., Evans D. H. Enhanced recombination associated with the presence of insertion and deletion mutations in poxvirus-infected cells. Virology. 1991 Sep;184(1):299–309. doi: 10.1016/0042-6822(91)90846-4. [DOI] [PubMed] [Google Scholar]
- Pattnaik A. K., Wertz G. W. Replication and amplification of defective interfering particle RNAs of vesicular stomatitis virus in cells expressing viral proteins from vectors containing cloned cDNAs. J Virol. 1990 Jun;64(6):2948–2957. doi: 10.1128/jvi.64.6.2948-2957.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perrotta A. T., Been M. D. The self-cleaving domain from the genomic RNA of hepatitis delta virus: sequence requirements and the effects of denaturant. Nucleic Acids Res. 1990 Dec 11;18(23):6821–6827. doi: 10.1093/nar/18.23.6821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Racaniello V. R., Baltimore D. Cloned poliovirus complementary DNA is infectious in mammalian cells. Science. 1981 Nov 20;214(4523):916–919. doi: 10.1126/science.6272391. [DOI] [PubMed] [Google Scholar]
- Radecke F., Spielhofer P., Schneider H., Kaelin K., Huber M., Dötsch C., Christiansen G., Billeter M. A. Rescue of measles viruses from cloned DNA. EMBO J. 1995 Dec 1;14(23):5773–5784. doi: 10.1002/j.1460-2075.1995.tb00266.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rose J. K., Buonocore L., Whitt M. A. A new cationic liposome reagent mediating nearly quantitative transfection of animal cells. Biotechniques. 1991 Apr;10(4):520–525. [PubMed] [Google Scholar]
- Schnell M. J., Mebatsion T., Conzelmann K. K. Infectious rabies viruses from cloned cDNA. EMBO J. 1994 Sep 15;13(18):4195–4203. doi: 10.1002/j.1460-2075.1994.tb06739.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shioda T., Hidaka Y., Kanda T., Shibuta H., Nomoto A., Iwasaki K. Sequence of 3,687 nucleotides from the 3' end of Sendai virus genome RNA and the predicted amino acid sequences of viral NP, P and C proteins. Nucleic Acids Res. 1983 Nov 11;11(21):7317–7330. doi: 10.1093/nar/11.21.7317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shioda T., Iwasaki K., Shibuta H. Determination of the complete nucleotide sequence of the Sendai virus genome RNA and the predicted amino acid sequences of the F, HN and L proteins. Nucleic Acids Res. 1986 Feb 25;14(4):1545–1563. doi: 10.1093/nar/14.4.1545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Youngner J. S., Frielle D. W., Whitaker-Dowling P. Dominance of temperature-sensitive phenotypes. I. Studies of the mechanism of inhibition of the growth of wild-type vesicular stomatitis virus. Virology. 1986 Nov;155(1):225–235. doi: 10.1016/0042-6822(86)90182-0. [DOI] [PubMed] [Google Scholar]
- Zhang W., Evans D. H. DNA strand exchange catalyzed by proteins from vaccinia virus-infected cells. J Virol. 1993 Jan;67(1):204–212. doi: 10.1128/jvi.67.1.204-212.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]