Abstract
The aim of this study was to evaluate the efficacy of visual rehabilitation with MP-1 microperimeter biofeedback in advanced optic neural dysfunction due to glaucoma, and to precisely characterize fixation stability and location in affected patients. Ten patients (18 eyes) with advanced glaucoma were submitted to a rehabilitation protocol that consisted of: a 25-item questionnaire (National Eye Institute Visual Functioning Que stionnaire); measurement of visual acuity; a reading speed test; microperimetry with fixation study, retinal sensitivity and the bivariate contour ellipse area (BCEA). The rehabilitation program consisted of 10 training sessions of 10 minutes per eye performed over a period of one week and was repeated at four months, eight months, and one year. Statistical analysis was performed using the Student’s t-test and Spearman correlation; p values less than 0.05 were considered statistically significant. In 13 eyes fixation changed from unstable to relatively unstable while its location changed from predominantly eccentric to predominantly central. In five eyes, fixation changed from relatively unstable to stable with a change of location from poor central fixation to predominantly central fixation. Mean retinal sensitivity changed from 7.43±8.28 dB to 8.33±9.04 dB (p<0.05); the mean best corrected visual acuity was 0.98±0.66 logMAR at the baseline assessment, and 0.75±0.6 logMAR at the end of rehabilitation (p>0.05); reading speed improved from a mean value of 31.4±4.3 words/minute to 55.6±3.2 words/minute at the end of the training (p<0.05). The BCEA changed from 0.94±0.39 deg2 to 0.86±0.46 deg2 (p=0.76). Rehabilitation with MP-1 biofeedback in patients with advanced glaucoma is a useful means of improving these patients’ fixation stability, reading speed and quality of life.
Keywords: bivariate contour ellipse area (BCEA), fixation, glaucoma, microperimetry, optic neural dysfunction, rehabilitation
Introduction
Glaucoma is not a single entity. The term glaucoma refers to a group of ocular disorders with multifactorial etiology, all of which are characterized clinically by the presence of intraocular pressure-associated optic neuropathy. All the forms are potentially progressive and can lead to blindness. The primary site of neurological injury is the optic nerve head (Casson et al., 2012).
It has been estimated that 12.3% of the worldwide population and 21.8% of European adults (including 18% of those over 50 years of age) have been diagnosed with glaucoma (Resnikoff et al., 2004; Bourne, 2006; Prokofyeva and Zrenner 2012).
Overall, glaucoma is responsible for 5.2 million cases of blindness worldwide (15% of global blindness) (Thylefors and Négrel, 1994). The management of patients with advanced glaucoma is complex, partly because the primary risk factor for glaucoma is age. Once the defect approaches the fixation area, visual field assessment may be of little value in monitoring the progression of the disease. Advanced optic neural dysfunction due to glaucoma is considered to be present when there is a loss of vision great enough to produce significant symptoms and functional impairment, which can include difficulty in performing visual tasks and tiring easily on performing such tasks. In view of these difficulties, we decided to evaluate the efficacy of visual rehabilitation with MP-1 microperimeter (NIDEK Technologies Srl, Padua, Italy) biofeedback in patients with advanced glaucoma.
Visual rehabilitation is a therapeutic approach that has been applied to different ocular diseases characterized by visual deterioration and loss of stable central fixation (Vingolo et al., 2009a). Through biofeedback methods, adopted in various branches of medicine, the patient learns in successive stages: i) to appreciate the variations of a bodily function through a system that measures and converts these variations into acoustic and/or luminous signals; ii) to modify these signals and, therefore, the function connected to them; and iii) to automatically control the function through practice, even in the absence of the feedback signal (Contestabile et al., 2002). Biofeedback applied to vision is still being studied, both in its methodological and its physiological aspects. Crossland et al. (2005), in a study of patients with macular disease, showed that the MP-1 microperimeter exploits cerebral plasticity and neurosensory adaptation to the central scotoma to improve these patients’ visual abilities. Indeed, such patients often develop a new preferred retinal locus (PRL), which can be defined as a discrete retinal area that contains more than 20% of the fixation points in a location that is considered unfavorable for reading and usually not the most profitable in terms of retinal sensitivity (Crossland et al., 2005).
This study was conducted to evaluate the efficacy of visual rehabilitation with MP-1 acoustic biofeedback in patients with advanced optic neural dysfunction due to glaucoma; we also aimed, through close monitoring with MP-1 microperimetry, to precisely characterize fixation stability and location in these patients, and to improve their reading speed. We also evaluated the utility of the bivariate contour ellipse area (BCEA) in assessing visual function in advanced glaucoma.
Materials and methods
Between February 2011 and March 2012, we recruited ten patients with advanced open-angle glaucoma (7 males, 3 females; mean age: 65 years, range: 59–71 years) at the Department of Ophthalmology, A. Fiorini Hospital, Sapienza University of Rome, in whom we examined a total of 18 eyes.
Inclusion criteria were: age between 40 and 75 years, a minimum 10-year history of glaucoma, a cup-to-disk ratio ≥ 0.7 or a cup-to-disk ratio asymmetry ≥ 0.2 between adjacent eyes, treatment with hypotonic therapy; all the patients had a middle school diploma. Exclusion criteria were: presence of angle-closure and secondary glaucoma, previous eye surgery (prior to the initial referral), previous corneal diseases, uveitis, vitreo-retinal interface diseases and retinal detachment. The diagnosis of advanced glaucoma was based on a complete eye examination which included: biomicroscopic examination of the anterior and posterior segments, visual acuity, Goldmann applanation tonometry, gonioscopy with three-mirror Goldmann lens, visual field examination by Humphrey 30-2 static perimetry, and spectral domain optical coherence tomography (Heidelberg HRA-2, TMB module, Heidelberg, Germany) with measurement of peripapillary retinal nerve fiber layer thickness.
Advanced glaucoma was defined as the presence of either cup-to-disk ratio ≥ 0.7 or a cup-to-disk ratio asymmetry ≥ 0.2 between adjacent eyes, with one severely affected eye having a best-corrected visual acuity below 20/200. All patients had advanced glaucomatous visual field loss, with a mean deviation worse than -24 dB, only a central or temporal island remaining in the visual field gray scale, and clear evidence of glaucoma in the fellow eye (manifesting as glaucomatous-appearing discs and a glaucomatous visual field).
Institutional review board approval was obtained and written informed consent was obtained from all participants. All procedures adhered to the tenets of the Declaration of Helsinki. This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
All the patients then underwent the same low-vision rehabilitation protocol, which consisted of: a 25-item questionnaire, assessment of distance and near visual acuity, a reading speed test (words/min), a fixation test, microperimetry, and 10 training sessions with microperimeter MP-1 acoustic biofeedback.
The 25-item questionnaire (National Eye Institute Visual Functioning Questionnaire, NEI-VFQ-25) developed under the sponsorship of the United States National Eye Institute (NEI), measures the influence of visual disability and visual symptoms on emotional well-being and social integration, as well as on task-oriented domains dependent on visual functions. The NEI-VFQ-25 comprises 12 subscales: General Health (1 question), General Vision (1 question), Near Vision (3 questions), Distance Vision (3 questions), Driving (2 questions), Peripheral Vision (1 question), Color Vision (1 question), Ocular Pain (2 questions), Role Limitations (2 questions), Dependency (3 questions), Social Function (2 questions), and Mental Health (4 questions). Answers to each question on the NEI-VFQ-25 were converted to a 100-point scale in which 100 was the best possible score and 0 the worst possible score. Each subscale score represents the average of one or more questions, as previously described (Mangione et al., 2001).
Best distance spectacle corrected visual acuity (BCVA) was determined using the Snellen chart, and converted to logarithm of the minimum angle of resolution (logMAR) for statistical analyses. Near visual acuity was determined at 30 cm with appropriate correction. Reading speed for each eye was measured by reading words written in black on a white background (Times New Roman font) at a distance of 30 cm with appropriate correction. Subjects were asked to read aloud, monocularly, as quickly as possible without skipping words. The sentences had no punctuation and contained words that occur frequently in Italian. Microperimetry and fixation test were performed with an MP-1 microperimeter (NIDEK Technologies Srl, Padua, Italy) using the automated program, the 4-2 strategy threshold test, and a 1° single cross as fixation target. However, at the beginning of the study, if a patient was not able to see this target, the size was enlarged to a 2° single cross fixation target. After training, only a 1° single cross target was used for all patients.
Retinal threshold sensitivity was measured in all eyes using the mire of Goldmann III (round shape with a white background) with stimulus intensity ranging from 0 to 20 dB. Stimulus presentation time was 200 ms. According to Fujii et al. (2002), the fixation pattern was graded on the basis of two parameters: i) the location of fixation i.e., the position of fixation with respect to the center of the foveal avascular zone; ii) the stability of fixation i.e., the ability of the eye to maintain a stable fixation in the PRL.
The location of fixation was defined as predominantly central when more than 50% of the fixation points were located within a predetermined limit area of variation of a 2° diameter circle centered in the fovea. Eyes with less than 50% but more than 25% of the preferred fixation points were classified as showing poor central fixation. Eyes with less than 25% of the preferred fixation points were classified as predominantly eccentric fixation.
The grading of fixation stability was based on the variation of the PRL. If more than 75% of the fixation points were located within a predetermined limit area of variation of a 2° diameter circle centered in the baricenter of all fixation points, the fixation defined as stable. If less than 75% of the fixation points were located within a 2° diameter circle, but more than 75% of the fixation points were located within a 4° diameter circle, the fixation was defined as relatively unstable. If less than 75% were located within a 4° diameter circle, the fixation was considered unstable (Fujii et al., 2002). Fixation stability was also quantified by calculating a BCEA encompassing 68% of fixation points based on fixation data collected by the MP-1 microperimeter. For statistical analysis, the BCEA values were converted into their logarithms to better approximate a normal distribution.
The rehabilitation program consisted of 10 training sessions of 10 minutes per eye performed over a period of one a week using the MP-1 acoustic biofeedback examination. The patients were asked to move their eyes according to an audio feedback which advised them whether they were getting closer to the desired final fixation position. All the procedures were followed on a monitor. The training was repeated at four months, eight months and one year.
At the end of the rehabilitation (12 months), the 25-item questionnaire (NEI-VFQ-25), the assessment of distant and near visual acuity, and the reading speed, fixation and microperimetry tests were repeated. Microperimetry was repeated with the follow-up function, which automatically retests the retinal sensitivity in exactly the same locations and under the same conditions as in the previous examination.
Statistical analysis
Statistical analysis was performed using paired Student’s t-test; p values less than 0.05 were considered statistically significant for all tests because of the preliminary nature of this study. We also used a Spearman correlation function to compare retinal sensitivity with visual acuity and reading speed.
Results
We examined 10 patients with advanced glaucoma (7 males and 3 females), for a total of 18 eyes, between February 2011 and March 2012. The mean patient age was 65 years (range: 59–71 years).
All participants completed the rehabilitation program at the end of which, 13 of the 18 eyes showed an improvement in fixation stability from relatively unstable to stable within the 2° diameter circle, while the location of fixation changed from predominantly eccentric to predominantly central. In five eyes fixation stability improved from unstable to relatively unstable within the 2° diameter circle with a change of location from poor central fixation to predominantly central fixation (Fig.s 1 and 2). Fixation stability, quantified by calculating a BCEA encompassing 68% of fixation points based on data collected by the MP-1 microperimeter, changed from 0.94±0.39 deg2 to 0.86±0.46 deg2 but this change between mean BCEA baseline and mean BCEA after rehabilitation was not statistically significant (p=0.76).
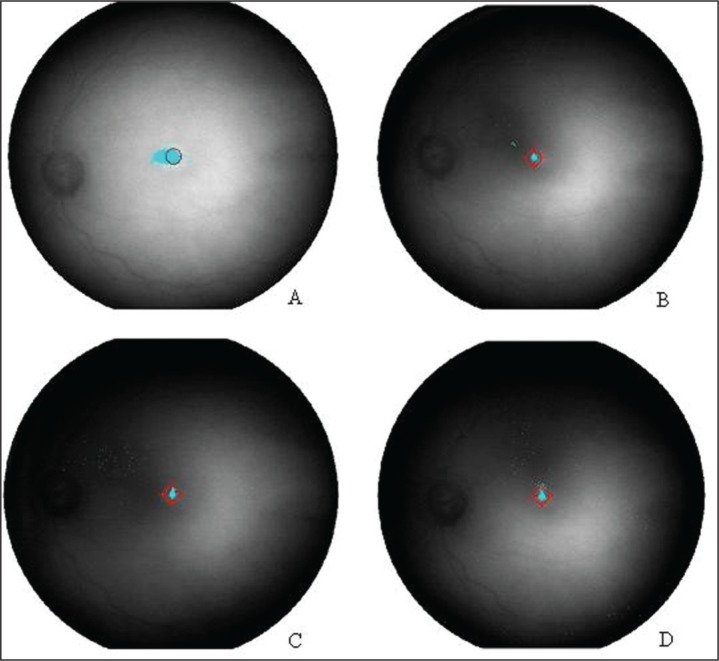
Figure - 1.
MP-1 microperimeter images. The fixation map at baseline (A), after 4 months (B), after 8 months (C) and at the end of the training (D) with MP-1 biofeedback in the left eye of one patient.
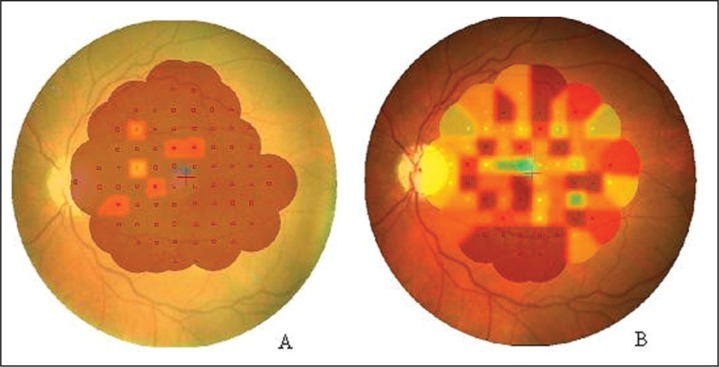
Figure - 2.
Image of the fundus with interpolated colorimetric map obtained with MP-1 microperimeter. (A) left eye of one patient before training, in which fixation is unstable; (B) left eye of the same patient after training, in which fixation is relatively unstable.
Mean retinal sensitivity changed from 7.43±8.28 dB to 8.33±9.04 dB and this result was statistically significant (p=0.022). The mean BCVA was 0.98±0.66 logMAR at the baseline assessment and 0.75±0.60 logMAR at the end of the visual rehabilitation; this result was not statistically significant (p=0.32).
Reading speed improved from a mean value of 31.4±4.3 words/minute at the beginning of the study to 55.6±3.2 words/minute at the end of the training; this result was statistically significant (p=0.031).
The NEI-VFQ-25 scores were found to be statistically increased at the end of the rehabilitation program (p=0.034).
At the end of the training the Spearman coefficient showed a positive correlation of retinal sensitivity with visual acuity and with reading speed (r=0.58).
Discussion
Glaucoma is a leading cause of blindness. At the early stage of the disease, macular function is mostly spared. However, in the advanced stage, the risk of the scotoma involving the fixation area becomes significant. At present, macular function in glaucoma patients is monitored using visual acuity testing and static and kinetic perimetry. Visual acuity is a good indicator of foveal function, but it reveals only one aspect of macular function. Perimetry can show the functioning of individual retinal locations in the macula, but the number and location of the testing points might not be adequate to detect subtle changes around the fixation region and in advanced glaucoma reliable test results are difficult to obtain because of unstable fixation. Advanced glaucoma is considered to be present when there is a loss of vision great enough to produce significant symptoms and functional impairment.
In view of this, we decided to evaluate the efficacy of MP-1 microperimetry as a means of improving fixation behavior and as a rehabilitation tool in patients with advanced glaucoma, with the aim of improving their visual acuity, reading speed and quality of life. Takanori et al. (2009) investigated fixation behavior in 39 eyes with advanced glaucoma using the MP-1 microperimeter and concluded that it is able to illustrate the fixation patterns in glaucomatous eyes and that patterns are well correlated with retinal sensitivity.
Biofeedback techniques applied to vision are still being studied, both in their methodological and physiological aspects. Various authors (Vingolo et al., 2009a; Contestabile et al., 2002; Giorgi et al., 2005; Mezawa et al., 1990) have proposed different visual rehabilitation techniques and instruments using biofeedback strategies ranging from basic systems, such as the Accommotrac Vision Trainer (The Nasa Connection, Seattle, WA) and improved biofeedback integrated system devices, to more complex instruments such as the fundus related MP-1 microperimeter (NIDEK Technologies Srl, Padua, Italy).
The Accommotrac Vision Trainer is a high-speed infrared optometer which records the vergence of light reflected from the retina at a rate of 40 Hz, then converts the signal into an auditory tone which increases in pitch and rate as accommodation decreases. The patient hears the tone and thus receives immediate auditory feedback as to his/her accommodative status. The MP-1 microperimeter biofeedback examination allows the ophthalmologist to train the patient to fixate the target with a new PRL. Patients are asked to move their eyes according to audio feedback which tells them whether they are getting closer to a specific retinal region chosen by the ophthalmologist. The patient’s perception of the sound increases his/her conscious attention, thereby facilitating the lock-in of the visual target and increasing the time the fixation target itself remains on the retina. This mechanism probably facilitates transmission of stimuli between intraretinal neurons as well as between the retina and brain, where the highest level of stimuli processing takes place, thereby supporting a “remapping phenomenon” (Alpeter et al., 2000; Buia and Tiesinga, 2006). Andrade et al. (2001) have shown that patients are usually unaware of their scotoma because, when the retina is damaged by a local lesion (induced scotoma), the cortical neurons driven by stimuli originating in this region do not remain inactive but become selective to stimuli originating in other parts of the retina. This process occurs in two distinct steps, each with its own time scale: i) a fast redistribution of receptive fields (RFs) in the area of the lesion, and ii) a gradual reorganization that leads to the final RF configuration. Although the mechanisms underlying the gradual rearrangement are becoming clearer, the first step remains obscure. Cortical neurons located in the retinotopic position corresponding to the scotoma receive some degree of activity from the unimpaired neurons in the area surrounding the lesion (Andrade et al., 2001). Cortical plasticity allows the brain to adapt to background modifications or to nervous system damage. It also underlies learning and attention processes. As explained by Safran and Landis (1996), “Cortical changes occurring after focal visual differentiation modify visual perception by filling in visual field defects with information from the area surrounding the scotoma”. This modification causes affected subjects to ignore or underestimate their defects. With visual field defects, cortical plasticity also causes distortions in spatial perception. These effects can delay the identification of visual field defects, and hence the initiation of therapy, while also affecting the results of certain visual field testing procedures (Safran and Landis, 1996).
As found by Mezawa et al. (1990), auditory biofeedback can be useful for the treatment of patients affected by congenital nystagmus, who have been found to report a subjective gain and an improvement of foveation time, amplitude, and frequency at the end of the visual training. The effects of auditory biofeedback on visual training have also been exploited in myopia (Rupolo et al., 1997; Angi et al., 1996) to improve visual acuity and psychological distress. Scanning laser ophthalmoscope (SLO) microperimetry is another technique that provides functional results by direct visualization of the macular area. It allows an exact, point-to-point correspondence between fundus image and perimetric results. Instability of fixation during computerized perimetry is a possible misleading factor that can result in inexplicable findings, especially in eyes with decreased visual acuity. The main characteristic of SLO microperimetry is that it allows real-time visualization of the stimuli presented on the retina: this in turn allows accurate monitoring of fixation and correlation of anatomical or pathological features directly with retinal function (Varano and Scassa,1998).
Biofeedback techniques have been performed in the treatment of ametropia (myopia, astigmatism, and presbyopia), nystagmus and amblyopia (Trachtman, 1978; Leung et al., 1996).
Previous studies have demonstrated the efficacy of low-vision rehabilitation by means of MP-1 biofeedback in patients with different macular diseases (vitelliform dystrophy, post-traumatic macular scar, Stargardt disease, myopic macular degeneration, cone dystrophy, age-related macular degeneration), reporting improvements in visual acuity, fixation behavior, retinal sensitivity and reading speed (Vingolo et al., 2009a; Vingolo et al., 2007; Pacella et al., 2012).
Other authors evaluated visual biofeedback training using the Visual Pathfinder (LACE Inc., Rome) system in patients with high myopia and demonstrated improvements in BCVA, amplitude of the main peak of the pattern reversal visual evoked potential (VEP), fixation behavior and retinal sensitivity. All this resulted in improved visual performances, better quality of life, and a positive psychological impact on these patients (Cannata et al., 2009).
Visual Pathfinder rehabilitation was tested in patients with retinitis pigmentosa, and it was shown that a structured stimulus can increase visual acuity and VEP amplitude, probably allowing a rearrangement of information from the residual retinal photoreceptors (Vingolo et al., 2009b).
Acoustic biofeedback and luminous biofeedback (a flickering black and white checkerboard) were compared in patients with age-related macular degeneration randomly divided into two groups (Vingolo et al., 2013). Both groups showed better visual performance after rehabilitation and the luminous flickering biofeedback stimulus was found to be significantly better able to train the patients to modify their PRL compared with the acoustic biofeedback. This suggests that it might be possible, in the damaged retina, to override dead photoreceptor and outer retinal layers and involve residual surviving cells, as well as amplify and integrate retinal and brain cortical plasticity.
Various hypotheses regarding the mechanisms of visual function improvement after visual training techniques can be advanced, such as that of an improvement in ocular motor control and “searching capacity”. Learning to use eccentric fixation could also be a mechanism contributing to improvements (Trachtman, 1994). In particular, it is very important to note that visual function could be improved as a result of the improved ability of patients undergoing training to exploit their visual acuity and other visual abilities to their full potential.
Sabel et al. (2011) proposed the “residual vision activation theory” to explain how visual functions can be reactivated and restored. In the past decade, there have been reports (mostly from Germany) suggesting that vision restoration therapy (VRT), which involves a specific pattern of visual stimulation, directed at the border between the seeing and the blind field, can result in expansion of visual fields in individuals with brain or optic nerve injury (Kasten and Sabel, 1995: Kasten et al., 1998). Romano et al. (2008) demonstrated that VRT improves stimulus detection and results in a shift of the position of the border of the blind field as measured on suprathreshold visual field testing. These results support prior reports and support VRT as a useful rehabilitation intervention for some patients with visual field defects deriving from retrochiasmatic lesions.
The possibility of restoring vision and the extent to which this can be achieved are dependent on the amount of residual tissue and its activation state. Sustained improvements require repetitive stimulation, possibly over days or months.
The results obtained are very likely determined in part by subjective variables such as learning effect, motivation, level of attention, psycho-physical capacities, type of environment and influence of the examiner (Carpineto et al., 2007).
The primary risk factor for glaucoma is age (Pache and Flammer, 2006: Steigerwalt et al., 2012; Nebbioso et al., 2011) and since this disease is characterized by degeneration of the retinal ganglion cells (RGCs) and cupping of the optic nerve head it has been suggested that the visual impairment seen in Alzheimer’s disease may be due to patients having undiagnosed glaucoma. Diseases that result in RGC death or optic nerve fiber degeneration may both be based on a similar biological mechanism (Kirby et al., 2010). Furthermore, recent research has found that damage from glaucoma can extend to the lateral geniculate nucleus and visual cortex (Gupta et al., 2006). This, finding, suggesting that glaucoma could account for some of the visual deficits seen in patients affected by dementia, underlines the importance, nowadays, of developing an integrated approach favoring the connection, within an interdisciplinary setting, of new developments at different levels (D’Angelo, 2012).
In this study we set out to demonstrate that it is possible to apply visual rehabilitation with MP-1 biofeedback also to patients with advanced glaucoma obtaining satisfactory results. In fact, increased fixation stability and retinal sensitivity were found to improve reading speed and visual efficiency. We also evaluated the utility of the BCEA to assess visual function in advanced glaucoma. The BCEA provides a precise continuous value for fixation stability, with smaller values indicating more stable fixation (Crossland et al., 2009).
Our experience with low-vision rehabilitation in patients with advanced glaucoma suggests that it could be possible to improve these subjects’ residual vision and thus restore a better visual performance and a much more positive psychological situation. Studies with larger numbers of patients should be performed to confirm the significance of these data.
References
- Alpeter E, Mackben M, Trauzettel-Klosinski S. The importance of sustained attention for patients with maculopthies. Vision Res. 2000;40:1539–1547. doi: 10.1016/s0042-6989(00)00059-6. [DOI] [PubMed] [Google Scholar]
- Andrade MA, Muro EM, Morán F. Simulation of plasticity in the adult visual cortex. Biol Cybern. 2001;84:445–451. doi: 10.1007/PL00007988. [DOI] [PubMed] [Google Scholar]
- Angi MR, Caucci S, Pilotto E, et al. Changes in myopia, visual acuity, and psychological distress after biofeedback visual training. Optom Vis Sci. 1996;73:35–42. doi: 10.1097/00006324-199601000-00006. [DOI] [PubMed] [Google Scholar]
- Bourne RR. Worldwide glaucoma through the looking glass. Br J Ophthalmol. 2006;90:253–254. doi: 10.1136/bjo.2005.083527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buia C, Tiesinga P. Attentional modulation of firing rate and synchrony in a model cortical network. J Comput Neurosci. 2006;20:247–264. doi: 10.1007/s10827-006-6358-0. [DOI] [PubMed] [Google Scholar]
- Cannata R, Salvatore S, Girolami I, et al. Acoustic biofeedback training in high myopic eyes. ARVO 2009 Annual Meeting; 3–7 May; Fort Lauderdale, Florida. 2009. [Google Scholar]
- Carpineto P, Ciancaglini M, Di Antonio L, et al. Fundus microperimetry patterns of fixation in type 2 diabetic patients with diffuse macular edema. Retina. 2007;27:21–29. doi: 10.1097/01.iae.0000256658.71864.ca. [DOI] [PubMed] [Google Scholar]
- Casson RJ, Chidlow G, Wood JP, et al. Definition of glaucoma: clinical and experimental concepts. Clin Experiment Ophthalmol. 2012;40:341–349. doi: 10.1111/j.1442-9071.2012.02773.x. [DOI] [PubMed] [Google Scholar]
- Contestabile MT, Recupero SM, Palladino D, et al. A new method of biofeedback in the management of low vision. Eye (Lond) 2002;16:472–480. doi: 10.1038/sj.eye.6700046. [DOI] [PubMed] [Google Scholar]
- Crossland MD, Culham LE, Kabanarou SA, et al. Preferred retinal locus development in patients with macular disease. Ophthalmology. 2005;112:1579–1585. doi: 10.1016/j.ophtha.2005.03.027. [DOI] [PubMed] [Google Scholar]
- Crossland MD, Dunbar HM, Rubin GS. Fixation stability measurement using the MP1 microperimeter. Retina. 2009;29:651–656. doi: 10.1097/IAE.0b013e318196bd65. [DOI] [PubMed] [Google Scholar]
- D’Angelo E. New trends in neuroscience: the challenge of functional neurology. Funct Neurol. 2012;27:5. [PMC free article] [PubMed] [Google Scholar]
- Fujii GY, de Juan E, Jr, Sunness J, et al. Patient selection for macular translocation surgery using the scanning laser ophthalmoscope. Ophthalmology. 2002;109:1737–1744. doi: 10.1016/s0161-6420(02)01120-x. [DOI] [PubMed] [Google Scholar]
- Giorgi D, Contestabile MT, Pacella E, et al. An instrument for biofeedback applied to vision. Appl Psychophysiol Biofeedback. 2005;30:389–395. doi: 10.1007/s10484-005-8424-1. [DOI] [PubMed] [Google Scholar]
- Gupta N, Ang LC, Noël de Tilly L, et al. Human glaucoma and neural degeneration in intracranial optic nerve, lateral geniculate nucleus, and visual cortex. Br J Ophthalmol. 2006;90:674–678. doi: 10.1136/bjo.2005.086769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kasten E, Sabel BA. Visual field enlargement after computer training in brain-damaged patients with homonymous deficits: an open pilot trial. Restor Neurol Neurosci. 1995;8:113–127. doi: 10.3233/RNN-1995-8302. [DOI] [PubMed] [Google Scholar]
- Kasten E, Wüst S, Behrens-Baumann W, et al. Computer-based training for the treatment of partial blindness. Nat Med. 1998;4:1083–1087. doi: 10.1038/2079. [DOI] [PubMed] [Google Scholar]
- Kirby E, Bandelow S, Hogervorst E. Visual impairment in Alzheimer’s disease: a critical review. J Alzheimers Dis. 2010;21:15–34. doi: 10.3233/JAD-2010-080785. [DOI] [PubMed] [Google Scholar]
- Leung V, Wick B, Bedell HE. Multifaceted treatment of congenital nystagmus: a report of 6 cases. Optom Vis Sci. 1996;73:114–124. doi: 10.1097/00006324-199602000-00007. [DOI] [PubMed] [Google Scholar]
- Mangione CM, Lee PP, Gutierrez PR, et al. Development of the 25-item National Eye Institute Visual Function Questionnaire. Arch Ophthalmol. 2001;119:1050–1058. doi: 10.1001/archopht.119.7.1050. [DOI] [PubMed] [Google Scholar]
- Mezawa M, Ishikawa S, Ukai K. Changes in waveform of congenital nystagmus associated with biofeedback treatment. Br J Ophthalmol. 1990;74:472–476. doi: 10.1136/bjo.74.8.472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nebbioso M, De Gregorio F, Prencipe L, et al. Psychophysical and electrophysiological testing in ocular hypertension. Optom Vis Sci. 2011;88:E928–939. doi: 10.1097/OPX.0b013e31821c6ca4. [DOI] [PubMed] [Google Scholar]
- Pacella E, Pacella F, Mazzeo F, et al. Effectiveness of vision rehabilitation treatment through MP-1 microperimeter in patients with visual loss due to macular disease. Clin Ter. 2012;163:e423–428. [PubMed] [Google Scholar]
- Pache M, Flammer J. A sick eye in a sick body? Systemic findings in patients with primary open-angle glaucoma. Surv Ophthalmol. 2006;51:179–212. doi: 10.1016/j.survophthal.2006.02.008. [DOI] [PubMed] [Google Scholar]
- Resnikoff S, Pascolini D, Etya’ale D, et al. Global data on visual impairment in the year 2002. Bull World Health Organ. 2004;82:844–851. [PMC free article] [PubMed] [Google Scholar]
- Romano JG, Schulz P, Kenkel S, et al. Visual field changes after a rehabilitation intervention: vision restoration therapy. J Neurol Sci. 2008;273:70–74. doi: 10.1016/j.jns.2008.06.026. [DOI] [PubMed] [Google Scholar]
- Rupolo G, Angi M, Sabbadin E, et al. Treating myopia with acoustic biofeedback: a prospective study on the evolution of visual acuity and psychological distress. Psychosom Med. 1997;59:313–317. doi: 10.1097/00006842-199705000-00014. [DOI] [PubMed] [Google Scholar]
- Sabel BA, Henrich-Noack P, Fedorov A, et al. Vision restoration after brain and retina damage: the “residual vision activation theory”. Prog Brain Res. 2011;192:199–262. doi: 10.1016/B978-0-444-53355-5.00013-0. [DOI] [PubMed] [Google Scholar]
- Safran AB, Landis T. Plasticity in the adult visual cortex: implications for the diagnosis of visual field defects and visual rehabilitation. Curr Opin Ophthalmol. 1996;7:53–64. doi: 10.1097/00055735-199612000-00009. [DOI] [PubMed] [Google Scholar]
- Prokofyeva E, Zrenner E. Epidemiology of major eye disease leading to blindness in Europe: a literature review. Ophthalmic Research. 2012;47:171–188. doi: 10.1159/000329603. [DOI] [PubMed] [Google Scholar]
- Steigerwalt RD, Jr, Vingolo EM, Plateroti P, et al. The effect of latanoprost and influence of changes in body position on patients with glaucoma and ocular hypertension. Eur Rev Med Pharmacol Sci. 2012;16:1723–1728. [PubMed] [Google Scholar]
- Takanori K, Teruyo T, Masanori H, et al. Fixation behavior in advanced stage glaucoma assessed by the microperimeter MP-1. Jpn J Ophthalmol. 2009;53:580–587. doi: 10.1007/s10384-009-0735-y. [DOI] [PubMed] [Google Scholar]
- Thylefors B, Négrel AD. The global impact of glaucoma. Bull World Health Organ. 1994;72:323–326. [PMC free article] [PubMed] [Google Scholar]
- Trachtman JN. Biofeedback of accomodation to reduce myopia: a case report. Am J Optom Physiol Opt. 1978;55:400–406. doi: 10.1097/00006324-197806000-00008. [DOI] [PubMed] [Google Scholar]
- Trachtman JN. Raccolta dei Risultati Ottenuti in Pazienti a Seguito del Trattamento con l’Allenatore Della Vista Accomotrack Vision Trainer-Riduzione Della Miopia. Atti del XVI corso di aggiornamento Apimo; Montecatini Terme, Italy. 1994. pp. 183–195. [Google Scholar]
- Varano M, Scassa C. Scanning laser ophthalmoscope microperimetry. Semin Ophthalmol. 1998;13:203–209. doi: 10.3109/08820539809056054. [DOI] [PubMed] [Google Scholar]
- Vingolo EM, Cavarretta S, Domanico D, et al. Microperimetric biofeedback in AMD patients. Appl Psychophysiol Biofeedback. 2007;32:185–189. doi: 10.1007/s10484-007-9038-6. [DOI] [PubMed] [Google Scholar]
- Vingolo EM, Salvatore S, Cavarretta S. Low-vision rehabilitation by means of MP-1 biofeedback examination in patients with different macular diseases: a pilot study. Appl Psychophysiol Biofeedback. 2009a;34:127–133. doi: 10.1007/s10484-009-9083-4. [DOI] [PubMed] [Google Scholar]
- Vingolo EM, Salvatore S, Grenga PL, et al. Retinal Degeneration: Causes, Diagnosis and Treatment. New York: Nova Publisher; 2009b. pp. 249–261. [Google Scholar]
- Vingolo EM, Salvatore S, Limoli PG. MP-1 Biofeedback: luminous pattern stimulus versus acoustic biofeedback in age related macular degeneration (AMD) Appl Psychophysiol Biofeedback. 2013;38:11–16. doi: 10.1007/s10484-012-9203-4. [DOI] [PubMed] [Google Scholar]