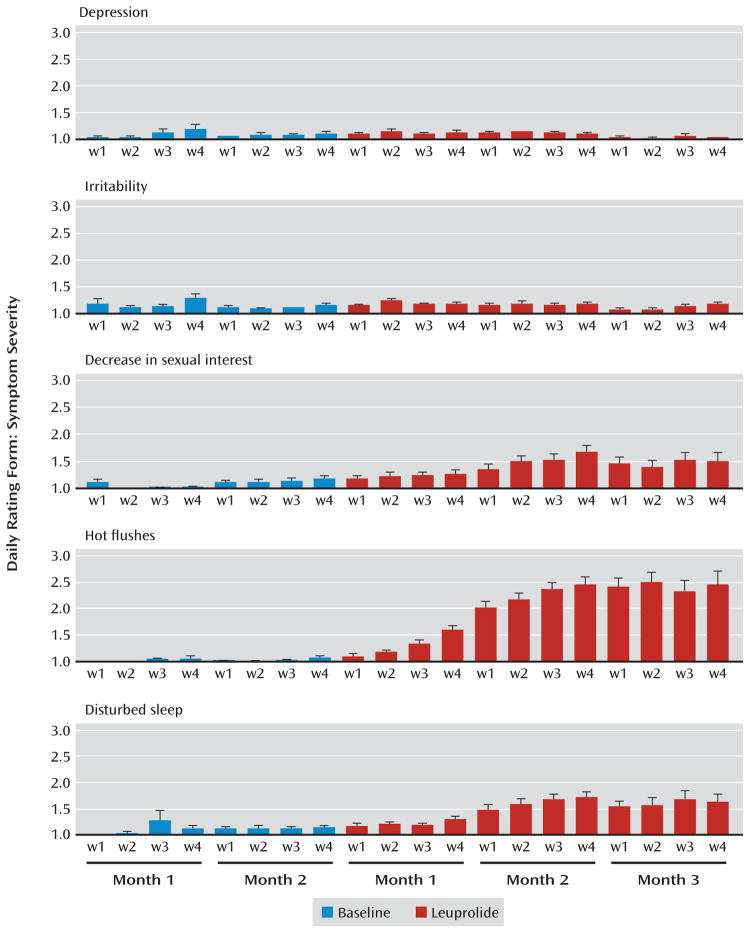
FIGURE 2. Effects of Leuprolide-Induced Ovarian Suppression on Mood and Behaviora.
a Histograms represent the weekly 7-day means of the symptom rating scale scores for each month of baseline and leuprolide treatment (error bars indicate standard error of the mean). Higher scores reflect greater symptom severity. Leuprolide-induced ovarian suppression was accompanied by significantly decreased sexual interest (F=12.9, df=2, 155, p<0.0001), disturbed sleep (F=20.2, df=2, 162, p<0.0001) and increased nighttime hot flushes (F=52.3, df=2, 111, p<0.0001) compared with baseline. In contrast, no significant changes were observed in depression or any other mood or behavioral symptoms measured. We did not observe significant correlations between baseline plasma levels of estradiol (or the changes in plasma levels between week 2 and week 4 of leuprolide treatment [i.e., during flare]) and the severity of any symptom measured (depression, decreased sexual interest, nighttime hot flushes, and disturbed sleep).