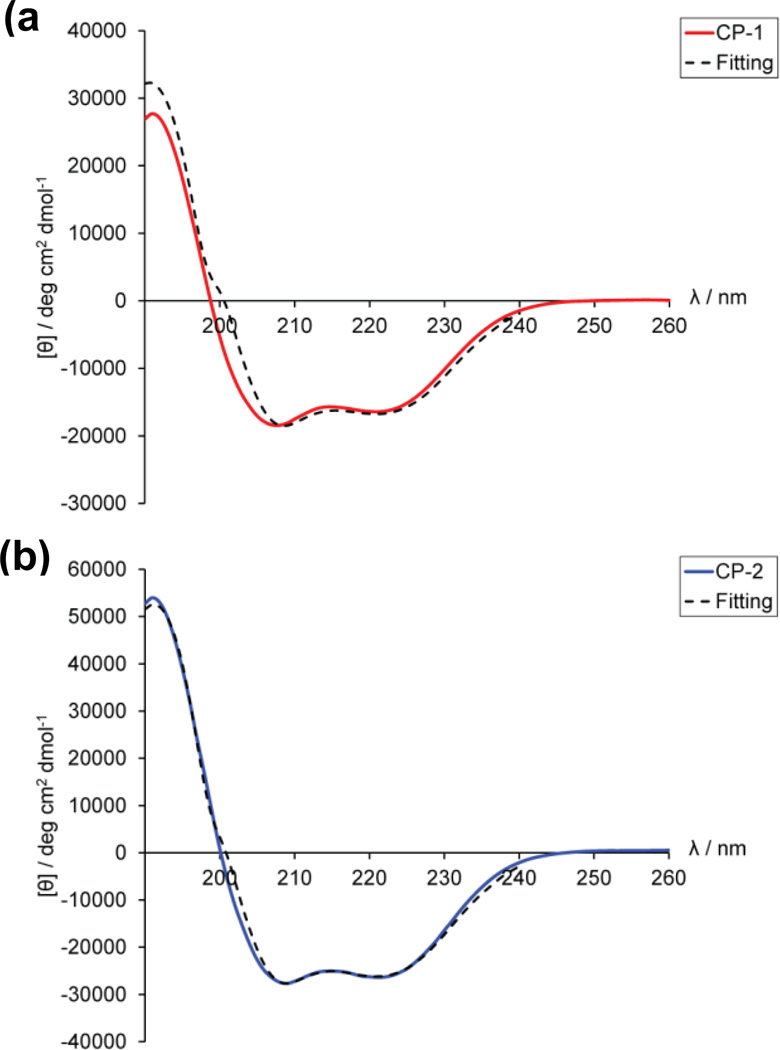
Figure 6.
CD spectra of purified (a) CP-1 and (b) CP-2 in acetate buffer, pH 4.0. Minima at 208 and 222 nm and a [θ]222 : [θ]208 ratio of <1 are representative of single coil α-helix. Concentrations of CP-1 and CP-2 were ∼0.8 and ∼1.2 mg/mL respectively. K2D3 curve-fitting37 for both CP-1 and CP-2 revealed secondary structure compositions that are dominated by α-helices (>50%), with small amounts of β-sheets (∼7%). CD spectroscopy was also performed on mixtures of CP-1 and CP-2 in different molar ratios at pH 4.0. This also produced similar α-helical spectra with a [θ]222 : [θ]208 ratio of <1 which indicates single coil α-helix conformation (supplementary Fig. S3).