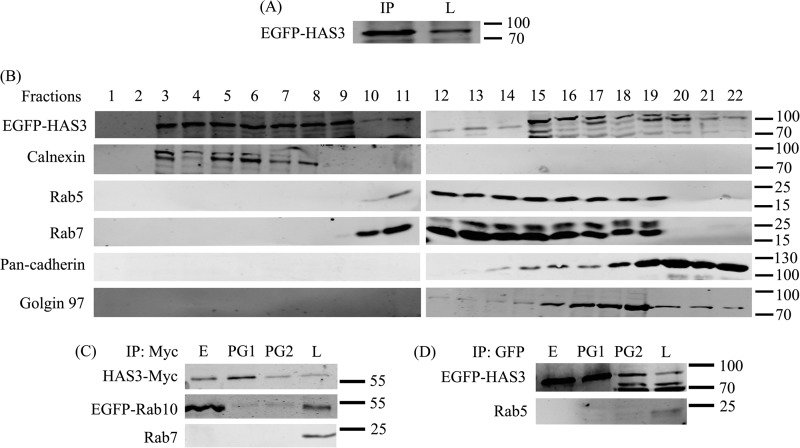
FIGURE 1.
Co-immunoprecipitation of HAS3 and Rab10. A, EGFP-HAS3 was immunoprecipitated from total cell lysate of MDCK cells and identified by anti-GFP antibody in Western blot. IP, immunoprecipitate; L, total cell lysate. B, the distribution of EGFP-HAS3 (∼90 kDa) in the sucrose gradient fractions was analyzed with anti-GFP antibody, and different subcellular organelles in the fractions were identified using the markers calnexin (ER) in fractions 3–8, Rab5 and Rab7 (endosomes) in fractions 10–14, and pan-cadherin (plasma membrane) and Golgin 97 (Golgi) in fractions 15–22. Fractions 10–14 were pooled for endosomes, thus excluding those (15–19) positive also for the indicators of Golgi and plasma membrane. C, HAS3-Myc and EGFP-Rab10 were co-transfected in MCF7 cells. Myc antibody immunoprecipitated EGFP-Rab10 and HAS3-Myc from endosomes, whereas another common endosomal protein (Rab7) did not co-immunoprecipitate with HAS3-Myc. D, EGFP-HAS3 was transiently transfected in MCF7 cells, immunoprecipitated with anti-GFP antibody, and analyzed for the presence of Rab5, another common endosomal protein. Rab5 did not co-immunoprecipitate with EGFP-HAS3. E, endosomes; PG1 and PG2, plasma membrane + Golgi mix, in fractions 10–14 and 15–22, respectively. L, total cell lysate. In some parts of the gradient, the anti-GFP antibody produced several apparently nonspecific bands in addition to that of GFP-HAS3 at ∼90 kDa.