Abstract

Photochemical reactions employing TiO2 and carboxylic acids under dry anaerobic conditions led to several types of C–C bond-forming processes with electron-deficient alkenes. The efficiency of alkylation varied appreciably with substituents in the carboxylic acids. The reactions of aryloxyacetic acids with maleimides resulted in a cascade process in which a pyrrolochromene derivative accompanied the alkylated succinimide. The selectivity for one or other of these products could be tuned to some extent by employing the photoredox catalyst under different conditions. Aryloxyacetic acids adapted for intramolecular ring closures by inclusion of 2-alkenyl, 2-aryl, or 2-oximinyl functionality reacted rather poorly. Profiles of reactant consumption and product formation for these systems were obtained by an in situ NMR monitoring technique. An array of different catalyst forms were tested for efficiency and ease of use. The proposed mechanism, involving hole capture at the TiO2 surface by the carboxylates followed by CO2 loss, was supported by EPR spectroscopic evidence of the intermediates. Deuterium labeling indicated that the titania likely donates protons from surface hydroxyl groups as well as supplying electrons and holes, thus acting as both a catalyst and a reaction partner.
Introduction
Carboxylic acids are pervasive in nature and are produced industrially on a large scale. Therefore, methods of employing them in organic preparations have received a lot of attention. Hard UV (λ ≤ 250 nm) irradiations generate carbon-centered radicals but also induce much degradation.1,2 Conventionally, decarboxylative alkylations have relied on preparations of unappealing precursors such as peroxides,3 Barton esters,4 or Hunsdieker salts.5 Recently attention shifted toward discovering catalytic systems capable of utilizing them in target-oriented syntheses. Decarboxylative copper-catalyzed reactions of carboxylic acids with carbonyls6 and with aldimines,7 and of amino acids with alkynes8 have been reported. Photoredox methods have also been used for this purpose9 but can suffer from difficulties in removing the catalyst. Interest in heterogeneous photoredox methods for carboxylates dates back to the 1970s and the photo-Kolbé reaction.10
Semiconductor photoredox catalysis (SCPC) offers tangible advantages for organic preparations. The chosen semiconductor can be nontoxic and can easily be removed by filtration, and when visible or soft UVA light is used, the procedure can be convenient and benign. Most applications of TiO2 SCPC have utilized aerobic conditions, resulting in selective oxidations of organic substrates11 or more complete substrate mineralizations.12 Under anhydrous, anaerobic conditions and with certain substrates, TiO2 SCPC can lead to the generation of specific radicals adapted for molecular assembly applications. To date, however, only a modest number of such processes have been identified and developed.13 Notably, SCPC additions of enol ethers to various acceptors have been investigated,14 and successful additions of tertiary amines to electron-deficient alkenes, some bearing chiral auxiliaries, have been described.15 We recently discovered that under dry, anaerobic conditions, TiO2 mediation with carboxylic acid precursors could result in carbon–carbon bond-forming processes. Alkylations and annulations were achieved when photolyses of certain acid precursors were carried out in the presence of suitable electron-deficient alkenes.16 This paper reports advances in crucial aspects of this process in the following areas: (1) structural features in the carboxylic acids that are necessary and sufficient; (2) the range of functionality in the alkene acceptors that can be tolerated; (3) how the reactions respond to modifications to the catalyst form and the catalyst support; and (4) the mechanism followed by the reaction.
Results and Discussion
In our preliminary study,16 we discovered an experimental protocol with reasonably wide applicability utilizing Degussa (Evonik) P25 as a 1–5 mg mL–1 dispersion in dried acetonitrile. This dispersion, with added carboxylic acid and substrate, in an oven-dried Pyrex tube was purged with argon and then irradiated by two face-to-face sunlamp arrays (UVA) at ambient temperature. After photolysis, the catalyst was removed by filtration and the products were isolated by conventional organic techniques.
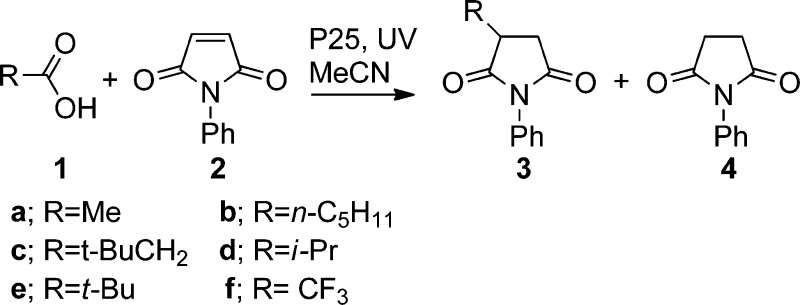
1. Alkylations of Maleimide with Aliphatic Carboxylic Acids
During initial investigations, N-phenylmaleimide (2) was found to be an ideal radical acceptor in this system, as it is symmetrical and photostable, its photodimerization is negligible (under our experimental conditions), and its electron-withdrawing character favors addition by weakly nucleophilic radicals. This alkene was therefore chosen to test the applicabliity of our process with simple aliphatic carboxylic acids 1. Standard unoptimized conditions were adopted so a clear comparison of behavior from acid to acid could be obtained (Table 1). In all instances, 0.1 mmol of 1 and 0.2 mmol of 2 were photolyzed in a suspension of the P25 catalyst (12 mg, 0.15 mmol) in CH3CN (12 mL) for 18 h. The reactions did indeed lead to the formation of adducts 3 incorporating the R moiety of the carboxylic acid and an additional H atom, together with significant amounts of succinimide 4 (Scheme 1).
Table 1. TiO2-Promoted Reactions of N-Phenylmaleimide with Aliphatic Carboxylic Acids.
| yield (%)a |
|||
|---|---|---|---|
| entry | R in 1 | adduct 3 | 4 |
| 1 | Me | 23 | 41 |
| 2 | Me(CH2)4 | 4 | 2 |
| 3 | t-BuCH2 | 18 | 40 |
| 4 | i-Pr | 25 | 25 |
| 5 | t-Bu | 38 | 31 |
| 6 | CF3 | ∼1 | trace |
NMR yields.
Scheme 1. P25-Promoted Alkylations of N-Phenylmaleimide with Simple Carboxylic Acids.
For acids that decarboxylated to give methyl (entry 1) or primary radicals (entries 2 and 3), the yields were low; however, increased yields were obtained with secondary and tertiary radicals (entries 4 and 5). Negligible adduct was detected for the destabilized, σ-type CF3 radical (entry 6).
The efficiency of the process evidently increased as the stabilization energy of the released radical increased (see the Supporting Information for a plot of radical stabilization energy vs yield). The observation of a significant amount of 4 in each case suggested that alkene 2 plays the role of an electron sink, consuming electrons in two sequential reduction–protonations.
2. Additions of Stabilized Radicals to N-Phenylmaleimide
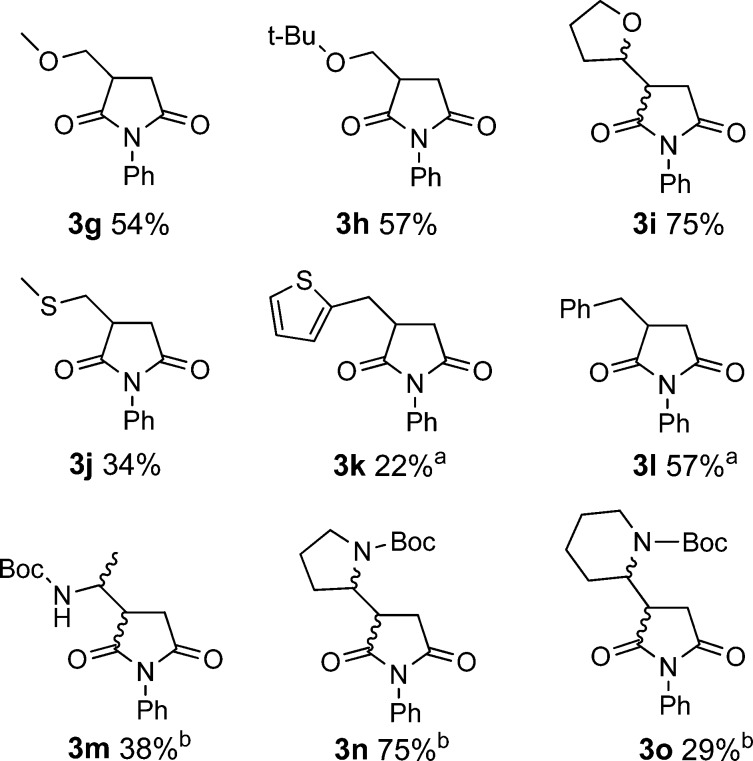
We next examined a set of addition reactions with N-phenylmaleimide involving more diverse carboxylic acids (Scheme 2). Primary alkyl radicals stabilized by α-alkoxy groups proved to be a good deal more reactive than the simple alkyl radicals. Methoxy- and tert-butoxyacetic acids gave the corresponding adducts (3g and 3h) in reasonable yields of 54% and 57%, respectively, while 2-tetrahydrofuroic acid furnished a 1:1 mixture of the two diastereomers of 3i in a very pleasing yield of 75%. However, methylthioacetic acid returned only a disappointing 34% yield of the desired product 3j. Moderate and poor yields were obtained with benzyl (57%) and 2-thienylmethyl acids (22%), but these adducts (3k and 3l) were accompanied by the dimers bibenzyl (18%) and 1,2-bis(thiophen-2-yl)ethane (27%), respectively.
Scheme 2. P25-Promoted Alkylations of N-Phenylmaleimide with Diverse Carboxylic Acids.
Accompanied by dimers (see the text).
Obtained as a 1:1 mixture of two diastereoisomers.
Decarboxylative additions also worked with Boc-protected α-amino acids, and the bottom row in Scheme 2 displays the adducts and the useful yields obtained. While Boc-proline was transformed into adduct 3n in a fine yield of 75%, Boc-alanine and the unnatural α-amino acid Boc-piperidinecarboxylic acid gave much more modest yields of 38% and 29%, respectively. As expected, these adducts were 1:1 mixtures of stereoisomers. No retention of chirality was observed when optically pure amino acids were employed because the released radicals have planar, or close to planar, configurations at their reactive centers. Surprisingly, vinylacetic, ethynylacetic, and cyanoacetic acids failed to react, even though the released radicals would be strongly stabilized.
3. Comparison of Catalyst Efficiencies
To compare the efficiencies of different forms of the titania catalyst, the reaction of phenoxyacetic acid with acrylamide, which was clean and efficient (82% adduct isolated)16 was chosen. For each photolysis, suboptimum conditions of phenoxyacetic acid (0.1 mmol) and acrylamide (0.2 mmol) with irradiation for 17 h in anhydrous CH3CN (12 mL) were employed. Where applicable, TiO2 (12 mg) was employed in a 1 mg mL–1 dispersion (see the Supporting Information for full details). With P25 itself (Table 2, entry 1), a 47% yield of the 4-phenoxybutanamide adduct was recorded. With Millenium PC500 titania, having about 6 times the surface area,17 the yield dropped slightly (entry 2). Reactions were also carried out in Schlenk tubes coated with a fine internal layer of TiO2 by a sol–gel process (see the Supporting Information for details). These proved to be very efficient and led to the best yield (entry 3). TiO2 Photospheres consisting of hollow Pyrex beads coated externally with rutile titania18 delivered lower conversion and adduct yields (entry 4). This was attributed to difficulties in dispersing the Photospheres satisfactorily throughout the reaction flask. Because of their buoyancy, they tended to accumulate at the top, and this was compounded by the fact that the high rate of magnetic stirring used in attempts to overcome this tended to result in their becoming damaged. An isolated yield of 68% was achieved upon scale-up of the reaction in the coated tube (entry 3). Incorporation of 4-tert-butyl and 4-trifluoromethyl substituents in the aryl ring of the acid component furnished the corresponding adducts in similarly pleasing yields of 68% and 66%, respectively. Although the sol–gel-coated tubes gave the best yields and conversions, the coatings tended to detach, so they could only be used about three times. We therefore concluded that for ease of handling with conventional organic techniques, scale-up, and efficient product formation, the orthodox P25 catalyst was the best compromise.
Table 2. Comparison of TiO2 Catalysts for the Reaction of Phenoxyacetic Acid with Acrylamide.
| entry | catalyst | yield of adduct (%)a |
|---|---|---|
| 1 | P25 | 47 (82b) |
| 2 | PC500c | 39 |
| 3 | sol–gel tubed | 73 (68e) |
| 4 | Photospheresf | 9 |
NMR yields.
Isolated yield from 5:1 acid/acrylamide irradiated for 62 h.
Millenium PC500 (anatase, surface area ∼300 m2 g–1, particle size 5–10 nm).
Schlenk tubes coated internally with P25 by a sol–gel process.
Yield of the isolated product.
Hollow glass spheres (mean diameter 45 μm, density 0.22 g/mL) coated with TiO2, from Microsphere Technology.
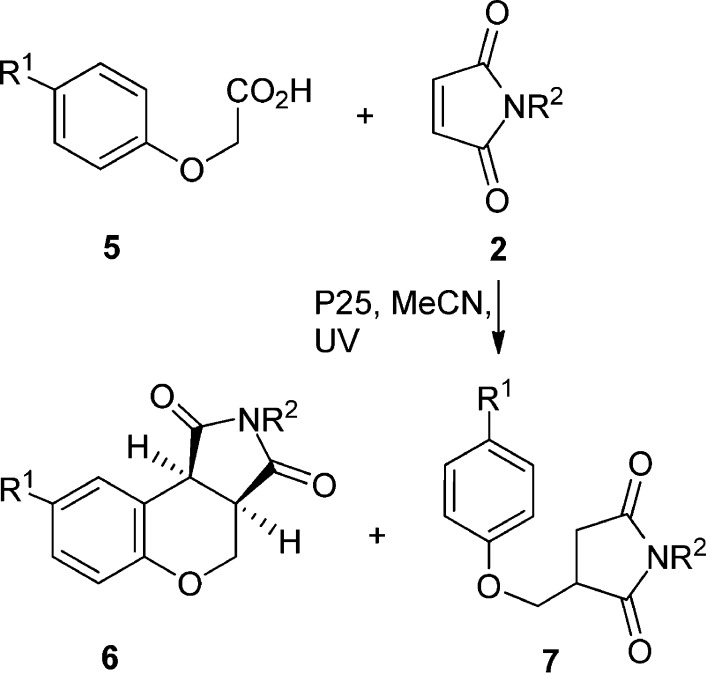
4. Addition–Cyclizations with Aryloxyacetic Acids
The reaction of aryloxyacetic acids (5) with N-substituted maleimides (2) led to N-substituted-3,4-dihydrochromeno[3,4]pyrrole-1,3-dione derivatives 6, each accompanied by a significant amount of the expected adduct 7 (Scheme 3). Tricyclics 6 presumably resulted from cyclization of the initial adduct radical onto the aryl ring followed by rearomatization. Good to excellent overall yields were obtained under all conditions (Table 3). A moderate amount of succinimide (11–43% yield) was also formed in each case.
Scheme 3. Titania-Promoted Reactions of Aryloxyacetic Acids with N-Substituted Maleimides.
Table 3. Titania-Promoted Reactions of Aryloxyacetic Acids with Maleimides.
| entry | R1 R2 (a–f) | conditionsa | yield (%)b | 6:7 ratio |
|---|---|---|---|---|
| 1 | H Me (a) | P25, 20 h | 75 | 2.38:1.0 |
| 2 | H Me (a) | P25 dd, 45 h | 75c | 10:1 |
| 3 | H Me (a) | coated tube, 22 h | 75c | 0.25:1.0 |
| 4 | H Ph (b) | P25, 11 h | 79 | 1.0:1.0 |
| 5 | H Ph (b) | coated tube, 22 h | 85c | 0.18:1.0 |
| 6 | t-Bu Me (c) | P25, 22 h | 57d | 4.20:1.0 |
| 7 | t-Bu Me (c) | P25 dd, 42 h | 56c | >10:1.0e |
| 8 | t-Bu Ph (d) | P25, 18 h | 74 | 1.64:1.0 |
| 9 | t-Bu Ph (d) | coated tube, 19 h | 51c | 0.82:1.0 |
| 10 | CF3 Me (e) | P25, 16 h | 61 | 1.10:1.0 |
| 11 | CF3 Me (e) | P25 dd, 42 h | 62c | 1.70:1.0 |
| 12 | CF3 Ph (f) | P25, 16 h | 63 | 0.57:1.0 |
| 13 | CF3 Ph (f) | coated tube, 17 h | 93c | 0.38:1.0 |
Dispersion of 1 mg mL–1 except for dd = dense dispersion of 5 mg mL–1.
mol % isolated products, except as noted.
NMR yield.
15% 6 was obtained in the absence of P25.
Only 6 was observed.
Table 3 shows that the process worked well with both electron-releasing and electron-withdrawing substituents in the aromatic ring and for methyl- and phenyl-substituted maleimides. The reactions with just P25 generally gave a slight excess of the chromenopyrrole 6 (entries 1, 4, 6, 8, and 10), except in the case of the CF3 substituent (entry 12), for which an excess of adduct 7 was observed. The selectivity for chromenopyrroles 6 was improved by using a more dense dispersion of P25 (entries 2, 7, and 11). On the other hand, photolyses in the sol–gel TiO2-coated tubes (entries 3, 5, 9, and 13) led to a reversal in selectivity with predominant formation of adduct 7. The possibility to tune the selectivity of reactions in this manner is an advantage of the technique. The 1H NMR spectra of all 6 had 3JH–H coupling constants between the two protons at the junction of the pyran and dihydropyrrole rings within the range of 9.2–9.3 Hz. This indicated a cis arrangement,19 in agreement with the conclusion that the reaction selectively formed the cis isomer in every case. Very few syntheses of dihydrochromeno[3,4]pyrrole-1,3-diones 6 have been reported,20 but compounds with the corresponding reduced ring system, chromenopyrrolidines, are well-known biologically active species.21
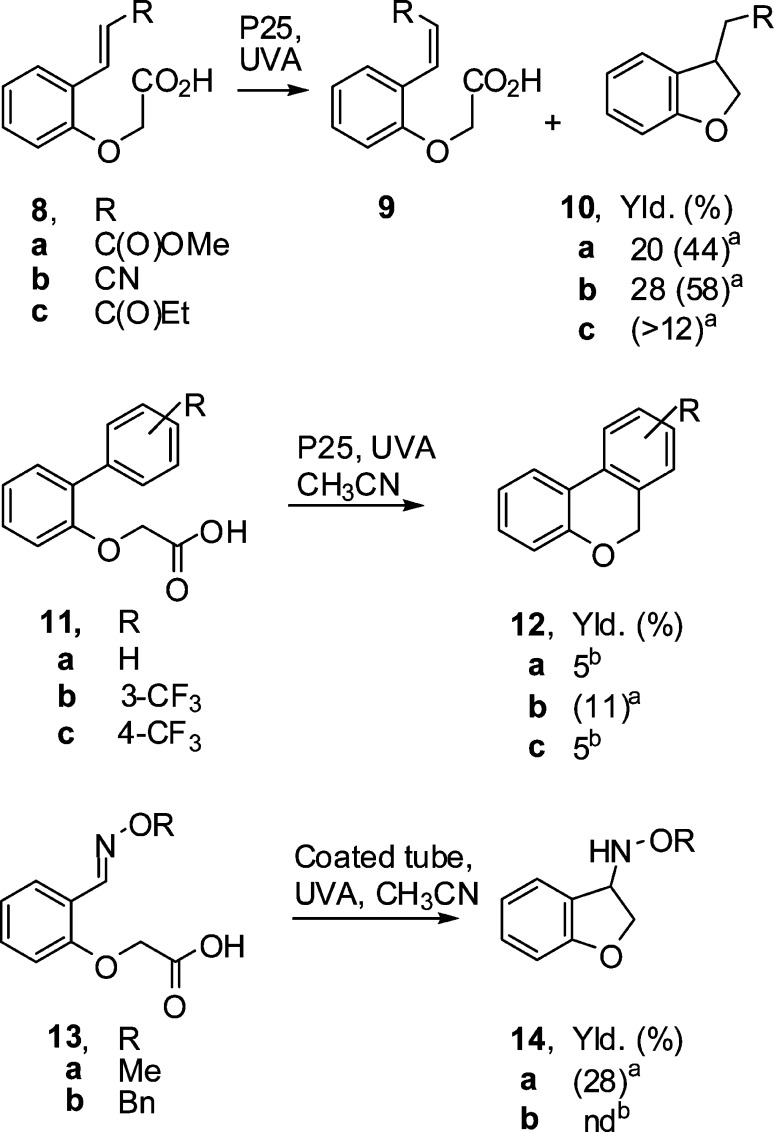
5. Intramolecular Ring Closures with Acceptor-Functionalized Aryloxyacetic Acids
Three types of acid framework with different acceptor groups were prepared to test the applicability of P25-mediated ring closures. 2-(2-Vinylphenoxy)acetic acids having alkene acceptors with ester (8a), nitrile (8b), and ketone (8c) substituents (Scheme 4) were obtained by treatment of 2-formylphenoxyacetic acid with the appropriate phosphorane (see the Experimental Section). 2-(Biphenyl-2-yloxy)acetic acids 11a–c having aromatic acceptors were prepared by coupling of the appropriate 1,1′-biphenyl-2-ol with methyl bromoacetate and subsequent hydrolysis of the ester with LiOH in MeOH/H2O. Acids 13a and 13b having oxime ether acceptor groups were prepared by condensation of 2-formylphenoxyacetic acid with the appropriate alkoxylamine hydrochloride. Reactions with these acids were carried out in two ways. The first used P25 under standard conditions as described above. The second used 1 mL aliquots of acid solutions (10 mM) in CD3CN (5 mL) in sol–gel-coated NMR tubes. The tubes were purged with argon and then placed in an overhead stirrer (turned on its side), which was then spun at 250 rpm. The tube was held at a slight angle with respect to horizontal in the “chuck” of the overhead stirrer so that, when rotated, it described a small (ca. 2 cm) circle at its non-chuck-held end so as to agitate the reaction solution. Irradiation was performed using a photoreactor with twelve 8 W black light blue (BLB) UV lamps (λmax = 365 nm) (see the Supporting Information for a graphic). Periodically the tube was removed for NMR analysis. This was repeated until all of the starting material had been consumed or the product concentration plateaued. For acids 8a–c with alkene acceptors, control photolyses in the absence of TiO2 showed that E/Z isomerization was significant (52–79%). In conventional P25-mediated reactions, 8a–c all gave rather complex product mixtures containing unreacted alkene as a mixture of E and Z isomers (8/9) as well as the 5-exo ring-closed dihydrobenzofurans 10. None of the 4-substituted chroman derivatives from 6-endo cyclizations were detected. Disappointing yields of 10a and 10b were isolated. The acids with aromatic acceptors 11a–c all reacted very poorly under both sets of conditions. NMR and GC–MS analyses showed complex product mixtures containing possible trace amounts of the cyclized 6H-benzo[c]chromenes 12a–c. The 2,3-dihydrobenzofuran core of 10 is found in many natural products and pharmaceutically relevant molecules and has generated much interest.22
Scheme 4. Substrates and Products for Intramolecular Reactions.
Yields in brackets are maximum yields from NMR monitoring of sol–gel tube reactions.
Yield estimated by 1H NMR analysis (nd = not determined).
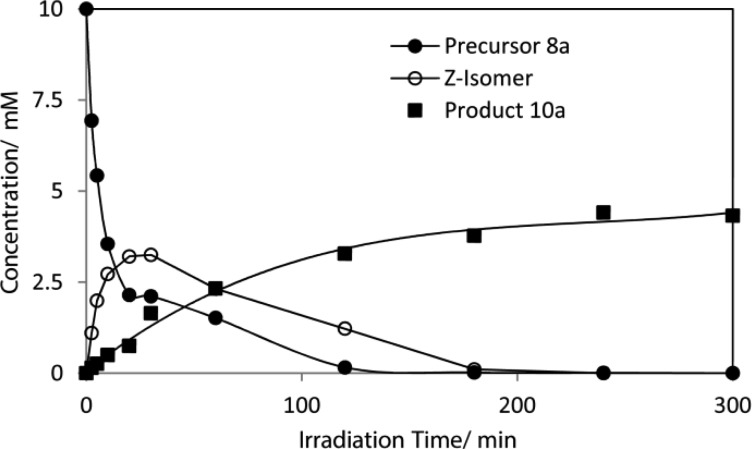
The reaction profile from irradiation of acid 8a in a sol–gel-coated tube obtained by NMR monitoring (Figure 1) shows the sharp decrease in 8a (the E isomer), which was all consumed in <150 min. The Z isomer 9a initially increased, passed through a maximum at about 25 min, and then steadily decreased as it too was converted to cyclic product 10a. The latter steadily increased, reaching a plateau by about 200 min. Analogous reaction profiles were obtained for the sol–gel tube reactions of 8a–c, 11b, and 13a. No cyclized product was identified in the sol–gel tube reactions of 11a and 11b. A significant yield (28%) of the cyclized product N-(2,3-dihydrobenzofuranyl)-O-methylhydroxylamine (14a) was found in the sol–gel tube reaction of oxime ether-functionalized acid 13a (Scheme 4). However, attempts to isolate and fully characterize this alkoxyamine were not successful.
Figure 1.
Reaction profile for acid 8a obtained by NMR monitoring.
6. Mechanism of the Reaction
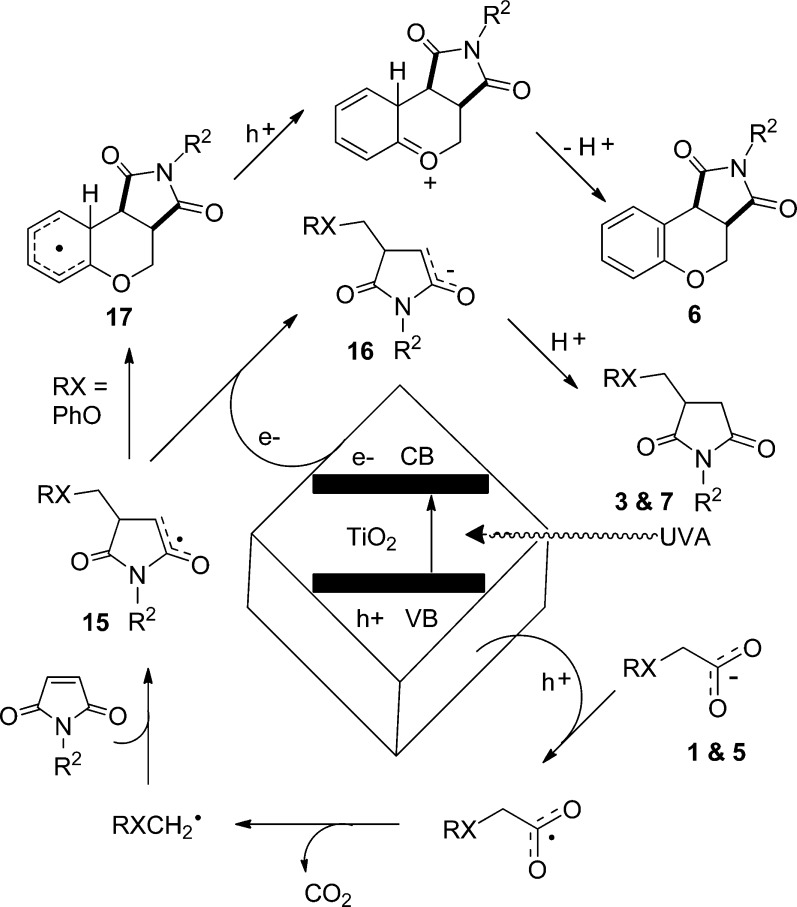
Hole capture at the TiO2 surface by a carboxylate creates the corresponding acyloxyl radical. Decarboxylation of this species furnishes the corresponding alkyl radical. Radicals of this type were directly observed by CW X-band EPR spectroscopy of frozen suspensions of P25 and t-BuCO2H in CH3CN.16 Transient t-Bu• radicals and PhOCH2• radicals were observed during UV irradiation of the acids t-BuCO2H and PhOCH2CO2H, respectively, with PC-50023 in fluid PhH at 300 K.16 In similar EPR experiments with vinylacetic acid and 2,2,2-triphenylacetic acid, we were also able to detect and characterize allyl and triphenylmethyl radicals, respectively (see the Supporting Information). The isotropic character of the solution EPR spectra of all the RXCH2• radicals established that in the main they were freely tumbling and not attached to the TiO2 surface (Scheme 5).
Scheme 5. Proposed Mechanism for TiO2-Promoted Reactions of Carboxylic Acids and Maleimides.
Literature precedents imply that weakly nucleophilic RXCH2• radicals should add rapidly to the electron-deficient double bonds of maleimides (and similar acceptors).24 Most likely the resulting adduct radicals 15 will be converted to enolates 16 by electron transfer from the TiO2 particles. Ready protonation25 will afford adducts 3, 7, etc. (Scheme 5). For ArOCH2• radicals containing aromatic rings, addition produces electrophilic imidoalkyl radicals 15, for which a competition exists between reduction to adducts 7 or homolytic closure (6-endo-trig) onto the aryl ring. As radicals 15 are weakly electrophilic in character, this annulation should be favored by increasing the electron density in the ring and, conversely, disfavored by decreasing it.26 In agreement with this, the proportion of 6 increased when an electron-releasing tert-butyl group was introduced to the ring, whereas it decreased when an electron-withdrawing trifluoromethyl group was introduced. The resonance-stabilized cyclohexadienyl-type radicals 17 will rearomatize to yield the functionalized chromenes 6. This aromatization could result from hole capture from TiO2 by the radicals 17 and subsequent proton loss as shown. Alternatively, electron transfer to maleimide might take place, as suggested in related work by Hoffmann.15b,15c Protonation of the resulting maleimide radical anions, followed by further electron capture and protonation steps, would explain the significant yields of succinimides obtained in our reactions. However, as greater amounts of succinimides than cyclized products 6 were generally formed, it is thought that direct reduction of the maleimide acceptor by the conduction band of the P25 also initiates this process.
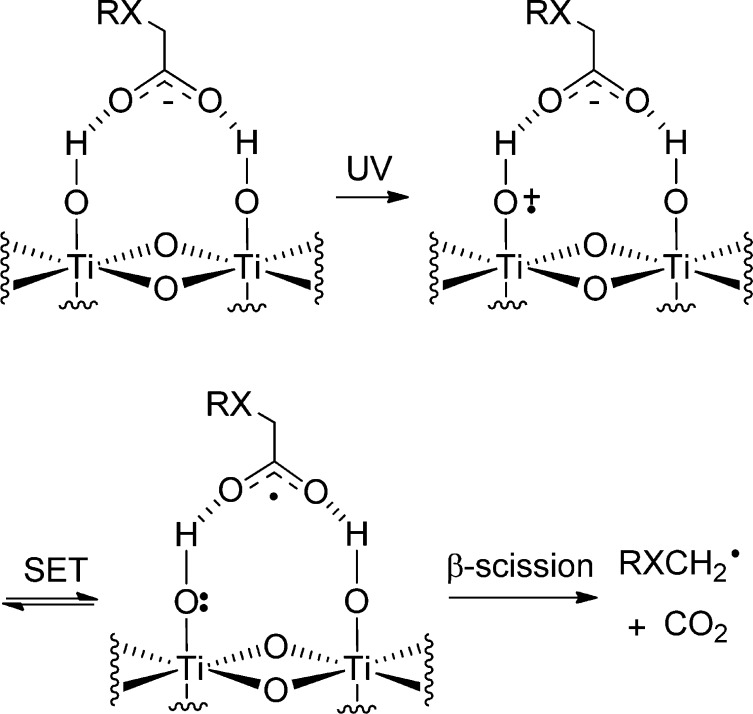
From the results described in Schemes 1 and 2 and Table 1, it can be deduced that the stability of the initial radicals is an important factor in this system. Carboxylic acids dissociate on the TiO2 surface to give the corresponding surface-bound carboxylate and a proton.27 Photolysis with UVA and resultant photoexcitation generates an electron–hole pair, which can migrate through the bulk of the semiconductor to the surface. The TiO2 surface is extensively hydroxylated,28 and it is thought that these species act as surface trapping sites for the valence-band holes.29 Electron transfer from the π system of the carboxylate to a hole trap site furnishes the surface-bound RXCH2–CO2• radical. We propose that a competition exists between β-scission of this surface-bound species to yield the desired free RXCH2• radical and back transfer of the electron, either from the trap site or from trapped conduction-band electrons, to the carboxylate. For acid precursors that generate stabilized RXCH2• radicals, such as those depicted in Scheme 2, the decarboxylation step is more favorable, thus explaining the improved yields and conversions recorded. Conversely, for the simple aliphatic acids of Table 1, the radical stabilization energy from the RX groups is minimal, so back transfer of an electron to the TiO2 will take precedence over the loss of CO2. This is illustrated in Scheme 6.
Scheme 6. SET Photoxidation Events Taking Place between Carboxylic Acids RXCH2CO2H and the TiO2 Surface.
It should be noted that while we believe radical stability to be a key aspect in this system, it is clear that there are other factors at play as well and that these take precedence in deciding the reaction outcome in a few instances. For example, vinylacetic acid would be expected to generate the allyl radical very readily in the presence of photoexcited TiO2. While we did observe the allyl radical in our EPR experiments, no products at all were observed in the preparative photolysis involving vinylacetic acid and 2. In this case, other as yet unknown factors at the TiO2 surface must come into play.
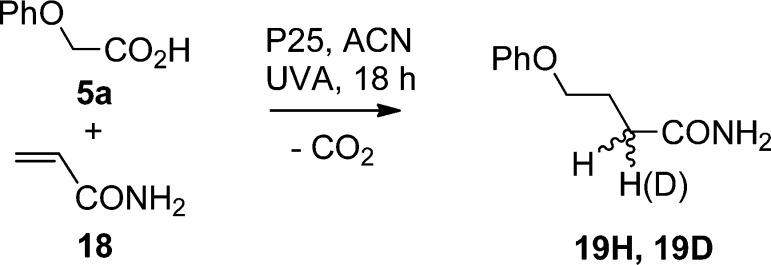
A crucial feature of the process is that in the formation of adducts 3 and 7 a hydrogen atom is gained from some source within the dispersion. This is not the proton lost from the aromatic ring because reduced adducts were formed equally well from acids lacking aromatic rings. Several deuterium labeling experiments were carried out to try to identify the source. The reaction of phenoxyacetic acid with acrylamide was again chosen as a test-bed process (Scheme 7) because it was clean and practically quantitative.
Scheme 7. Deuterium Labeling Test Reactions.
When this reaction was carried out in d3-CH3CN solvent, the isolated adduct was screened for D incorporation by 1H and 2H NMR spectroscopy and GC–MS. However, no deuterium could be detected in the 4-phenoxybutanamide (19H). Reaction with d1-phenoxyacetic acid also yielded adduct with no detectable D incorporation. Thus, unless the acidic D atom of 5a rapidly and completely exchanges, the source of the additional H atom is neither the acid nor the solvent. The most likely source therefore is the P25 semiconductor. Obviously it is counterintuitive that a metal oxide would supply protons. However, it is well-established that H2O molecules and HO groups are attached to the surface of the TiO2 particles.28a Drying P25 in a vacuum at 150 °C removes a significant amount of the surface H2O while leaving the chemically attached HO groups. When P25 dried in this way was used, the yield of 18 was scarcely reduced, suggesting that the OH groups were likely the proton donors. Attempts were made to deuterate the H2O and OH groups on the P25 surface by refluxing in degassed D2O in a glovebox. However, it was found that back exchange occurred immediately upon exposure to air and rapidly with any moisture traces in solvents or on surfaces. Only partly deuterated P25 (ca. 80% as judged by IR) could be obtained. Definitive experiments were difficult to achieve because of this. When the partly deuterated P25D was photolyzed with the undeuterated acid, the phenoxybutanamide product contained no deuterium. It seemed possible, however, that P25 surface OD groups could have rapidly exchanged with protons from the COOH groups of 5a. Experiments with partly deuterated P25D and 5a-COOD were also carried out, but deuterated 19D was again not found. In view of the partially deuterated nature of the P25D and the ease with which it reverts to the protiated form, making handling difficult, our tentative conclusion is still that the P25 is the source of the H atoms.
Conclusions
We have found that photochemical reactions employing P25 and carboxylic acids under dry anaerobic conditions generate C-centered radicals that can be deployed in several types of C–C bond-forming processes. Simple aliphatic carboxylic acids alkylated N-phenylmaleimide, albeit rather inefficiently. Acids that yielded alkoxyalkyl, alkylthioalkyl, and benzyl radicals alkylated electron-deficient alkenes in moderate to good yields. Boc-protected amino acids furnished aminoalkyl radicals that alkylated N-phenylmaleimide in useful yields. The reactions of aryloxyacetic acids with maleimides resulted in a cascade process in which a pyrrolochromene derivative accompanied the alkylated succinimide. Selectivity for one or other of these products could be tuned to some extent by employing the TiO2 catalyst under different conditions. Aryloxyacetic acids adapted for intramolecular ring closures by the inclusion of 2-alkenyl, 2-aryl, or 2-oximinyl functionality reacted rather poorly, especially the 2-aryl acceptor types.
The mechanism (Scheme 5) is supported by EPR spectroscopic evidence of the intermediates. An intriguing aspect of the process is the identity of the proton donor. Experiments with deuterium-labeled solvent and reactants implied that this was the hydroxyl groups on the surface of the P25. It appears that the titania supplies electrons and holes upon photostimulation but also donates protons from surface hydroxyl groups, thus acting as both a catalyst and a reaction partner.
These photoredox reactions are cleanly, safely, and cheaply carried out in the laboratory, and the heterogeneous catalyst is simply filtered off during workup. Thus, with the exception mentioned above, they constitute a useful synthetic protocol.
Experimental Section
General Procedure for TiO2 Photoredox Experiments
To a suspension of semiconductor (1–5 mg mL–1) in MeCN (freshly distilled over CaH2) in an oven-dried Pyrex tube were added known amounts of the desired carboxylic acid and alkene. The resulting suspension was degassed by bubbling with argon for 20 min. The mixture was then irradiated with eight 29 cm 15 W Philips Cleo tubes (λ = 350 nm) for the desired reaction time at ambient temperature. Following irradiation, the semiconductor powder was removed by filtration through a Celite pad. The solvent was removed under reduced pressure, and the reaction mixture was purified by chromatography. Yields were determined from the amounts of isolated products and/or from the 1H NMR spectra by reference to CH2Br2 as an internal standard. Additional general procedures are described in the Supporting Information.
NMR Tube Irradiations
Irradiations were carried out using titania sol–gel-coated NMR tubes. A photoreactor consisting of two lots of six 8 W BLB UV lamps (emission λmax = 365 nm), each arranged in a semicylinder with an aluminum reflector, was used to irradiate the contents of the NMR tube. The two photoreactor semicylinders were brought together to surround the NMR tube (see the Supporting Information for a graphic). For a typical reaction, a stock solution comprising acid (10 mM) in d3-CH3CN was prepared. This solution was purged with argon for 5 min, and then 1 mL was pipetted into an argon-flushed sol–gel-coated NMR tube. The cap was quickly replaced on the tube and sealed with Parafilm. The loaded NMR tube was placed in an overhead stirrer (turned on its side), which was spun at 250 rpm. The tube was held at a slight angle with respect to horizontal in the chuck of the overhead stirrer so that, when rotated, it described a small (ca. 2 cm) circle at its non-chuck-held end so as to agitate the reaction solution. Once the NMR tube was in place, the irradiation was started, and the tube was periodically removed and analyzed by NMR.
Deuterium Labeling Experiments
These experiments were carried out in a similar fashion to the photoredox experiments outlined above. Following photolysis, the reaction mixtures were scrutinized for deuterium incorporation by 1H and 2H NMR spectroscopy and GC–MS. Further details of these experiments are provided in the Supporting Information.
3-Methyl-1-phenylpyrrolidine-2,5-dione (3a)
Acetic acid (6 mg, 0.1 mmol), N-phenylmaleimide (34.7 mg, 0.2 mmol), and TiO2 (12 mg, 0.15 mmol) were reacted in accordance with the general procedure to yield 3a (24% by NMR) as a white solid. Mp: 94–96 °C. 1H NMR (400 MHz, CDCl3, 296 K): δ 1.44 (d, J = 7.2 Hz, 3H), 2.47–2.54 (m, 1H), 2.97–3.14 (m, 2H), 7.27–7.30 (m, 2H), 7.36–7.42 (m, 1H), 7.44–7.50 (m, 2H). 13C NMR (100 MHz, CDCl3, 297 K): δ 16.9, 34.9, 36.7, 126.4, 128.6, 129.2, 132.0, 175.5, 179.6. LR-ESIMS: m/z 190 [M + H]+. HR-ESIMS: m/z 190.0861 (calcd for C11H12NO2, 190.0863). Also observed was N-phenylsuccinimide (41% by NMR).
3-Pentyl-1-phenylpyrrolidine-2,5-dione (3b)
n-Hexanoic acid (11.6 mg, 0.1 mmol), N-phenylmaleimide (34.7 mg, 0.2 mmol), and TiO2 (12 mg, 0.15 mmol) were reacted in accordance with the general procedure. Following irradiation, a yield of 4% (0.04 mmol) was determined for 3b. Yields were determined by assigning peaks “a” and “b” by analogy with the previously characterized 3-ethyl adduct.301H NMR (500 MHz, CDCl3, 295 K): δ 2.60 (dd, J = 3.3, 17.0 Hz, 1H), 2.97–3.07 (m, 1H). Also observed were N-phenylsuccinimide (4% by NMR) and unreacted n-hexanoic acid (65% conversion by NMR).
3-Neopentyl-1-phenylpyrrolidine-2,5-dione (3c)
tert-Butylacetic acid (11.6 mg, 0.1 mmol), N-phenylmaleimide (34.7 mg, 0.2 mmol), and TiO2 (12 mg, 0.15 mmol) were reacted in accordance with the general procedure to yield 3c (18% by NMR) as a white solid. Mp: sample decomposed at >250 °C. 1H NMR (400 MHz, CDCl3, 296 K): δ 0.94 (s, 9H), 1.36 (dd, J = 10.7, 14.0 Hz, 1H), 2.12 (dd, J = 2.0, 13.9 Hz, 1H), 2.54 (dd, J = 5.3, 18.1 Hz, 1H), 3.03 (dd, J = 9.1, 18.1 Hz, 1H), 2.84–2.92 (m, 1H), 7.19–7.22 (m, 2H), 7.29–7.34 (m, 1H), 7.37–7.42 (m, 2H). 13C NMR (100 MHz, CDCl3, 297 K): δ 29.6, 29.7, 31.0, 37.6, 45.9, 126.4, 128.6, 129.2, 132.1, 175.8, 179.8. LR-ESIMS: m/z 246 [M + H]+. HR-ESIMS: m/z 246.1486 (calcd for C15H20NO2, 246.1489). Also observed was N-phenylsuccinimide (40% by NMR).
3-Isopropyl-1-phenylpyrrolidine-2,5-dione (3d)
Isobutyric acid (8.1 mg, 0.1 mmol), N-phenylmaleimide (34.7 mg, 0.2 mmol), and TiO2 (12 mg, 0.15 mmol) were reacted in accordance with the general procedure to yield 3d (25% by NMR) as a white solid. Mp: 90–92 °C. 1H NMR (400 MHz, CDCl3, 297 K): δ 0.99 (d, J = 6.8 Hz, 3H), 1.07 (d, J = 6.9 Hz, 3H), 2.32–2.40 (m, 1H), 2.56 (dd, J = 4.4, 18.3 Hz, 1H), 2.79 (dd, J = 9.4, 18.3 Hz, 1H), 2.87–2.92 (m, 1H), 7.18–7.21 (m, 2H), 7.29–7.34 (m, 1H), 7.37–7.43 (m, 2H). 13C NMR (100 MHz, CDCl3, 300 K): δ 17.3, 20.0, 29.2, 30.6, 45.8, 126.5, 128.6, 129.2, 131.9, 175.9, 178.4. LR-ESIMS: m/z 218 [M + H]+. HR-ESIMS: m/z 218.1178 (calcd for C13H16NO2, 218.1176). Also observed was N-phenylsuccinimide (25% by NMR).
3-tert-Butyl-1-phenylpyrrolidine-2,5-dione (3e)
Pivalic acid (10.2 mg, 0.1 mmol), N-phenylmaleimide (34.7 mg, 0.2 mmol), and TiO2 (12 mg, 0.15 mmol) were reacted in accordance with the general procedure to yield 3e (38% by NMR) as a white solid. Mp: 101–103 °C. 1H NMR (400 MHz, CDCl3, 300 K): δ 1.12 (s, 9H), 2.67–2.80 (m, 2H), 2.89 (dd, J = 8.8, 17.6 Hz, 1H), 7.24–7.26 (m, 2H), 7.36–7.41 (m, 1H), 7.45–7.49 (m, 2H). 13C NMR (100 MHz, CDCl3, 300 K): δ 27.2, 32.0, 33.8, 49.9, 126.6, 128.6, 129.2, 132.0, 175.6, 177.4. LR-ESIMS: m/z 232 [M + H]+. HR-ESIMS: m/z 232.1326 (calcd for C14H18NO2, 232.1332). Also observed was N-phenylsuccinimide (31% by NMR).
3-(Trifluoromethyl)-1-phenylpyrrolidine-2,5-dione (3f)
Trifluoroacetic acid (11.4 mg, 0.1 mmol), N-phenylmaleimide (34.7 mg, 0.2 mmol), and TiO2 (12 mg, 0.15 mmol) were reacted in accordance with the general procedure to yield 3f (∼1% by NMR). 1H NMR (400 MHz, CDCl3, 295 K): δ 2.86 (dd, J = 5.5, 18.6 Hz, 1H), 2.99 (dd, J = 9.5, 18.6 Hz, 1H), 3.61 (m, 1H), 7.10 (m, 2H), 7.39 (m, 3H). 19F NMR (375 MHz, CDCl3, 296 K): δ −69.3 (s, 3F). Data are consistent with literature values.31 Also observed was N-phenylsuccinimide (trace).
3-(Methoxymethyl)-1-phenylpyrrolidine-2,5-dione (3g)
Methoxyacetic acid (54.4 mg, 0.63 mmol), N-phenylmaleimide (433.6 mg, 2.50 mmol), and TiO2 (75 mg, 0.94 mmol) were reacted in accordance with the general procedure. Following irradiation for 18 h, the reaction mixture was purified by column chromatography on silica gel (eluent: 30% EtOAc in petroleum ether 40–60) to yield 3g as a white powder (74.4 mg, 54%). Mp: 109–111 °C. 1H NMR (400 MHz, CDCl3, 299 K): δ 2.86–2.99 (m, 2H), 3.08–3.15 (m, 1H), 3.38 (s, 3H), 3.36 (dd, J = 3.3, 9.1 Hz, 1H), 3.91 (dd, J = 3.9, 9.1 Hz, 1H), 7.26–7.30 (m, 2H), 7.36–7.41 (m, 1H), 7.44–7.49 (m, 2H). 13C NMR (75 MHz, CDCl3, 298 K): δ 31.9, 41.2, 59.3, 70.8, 126.6, 128.6, 129.1, 132.1, 175.8, 177.3. Data are consistent with literature values.16
3-(tert-Butoxymethyl)-1-phenylpyrrolidine-2,5-dione (3h)
tert-Butoxyacetic acid (87.3 mg, 0.66 mmol), N-phenylmaleimide (462.3 mg, 2.67 mmol), and TiO2 (80 mg, 1.0 mmol) were reacted in accordance with the general procedure. Following irradiation for 17 h, 1H NMR analysis revealed that N-phenylmaleimide had been consumed entirely while a significant quantity of tert-butoxyacetic acid remained. A further 462.3 mg of N-phenylmaleimide was hence added, and the reaction was photolyzed for another 18 h. The reaction mixture was then purified by column chromatography on silica gel (eluent: 25% EtOAc in pentanes) to yield 3h as a yellow oil (97.8 mg, 57%). 1H NMR (500 MHz, CDCl3, 295 K): δ 1.18 (s, 9H), 2.85–2.89 (m, 2H), 3.07–3.11 (m, 1H), 3.59 (dd, J = 2.9, 8.5 Hz, 1H), 3.91 (dd, J = 3.5, 8.5 Hz, 1H), 7.27 (d, J = 7.4 Hz, 2H), 7.39 (t, J = 7.4 Hz, 1H), 7.48 (t, J = 7.4 Hz, 2H). 13C NMR (75 MHz, CDCl3, 297 K): δ 27.4, 32.3, 41.3, 60.7, 73.3, 126.6, 128.6, 129.2, 134.2, 176.3, 178.0. LR-ESIMS: m/z 262 [M + H]+. HR-ESIMS: m/z 262.1440 (calcd for C15H20NO3, 262.1438).
1-Phenyl-3-(tetrahydrofuran-2-yl)-pyrrolidine-2,5-dione (3i)
2-Tetrahydrofuroic acid (76.6 mg, 0.66 mmol), N-phenylmaleimide (462.3 mg, 2.67 mmol), and TiO2 (80 mg, 1.00 mmol) were reacted in accordance with the general procedure. Following irradiation for 18 h, the reaction mixture was purified by column chromatography on silica gel (eluent: Et2O) to yield a 1:1 mixture of the two diastereoisomers of 3i as a yellow oil (121.8 mg, 75%). Diastereomer 1: 1H NMR (400 MHz, CDCl3, 295 K): δ 1.92–2.02 (m, 2H), 2.02–2.12 (m, 2H), 2.79–2.84 (m, 1H), 2.99 (dd, J = 9.3, 18.3 Hz, 1H), 3.26–3.30 (m, 1H), 3.76–3.84 (m, 2H), 4.23–4.27 (m, 1H), 7.26–7.32 (m, 2H), 7.37–7.42 (m, 1H), 7.45–7.51 (m, 2H). 13C NMR (100 MHz, CDCl3, 297 K): δ 25.9, 27.9, 32.7, 43.4, 68.8, 79.1, 126.6, 128.6, 129.1, 132.0, 175.7, 175.9. Diastereomer 2: 1H NMR (500 MHz, CDCl3, 295 K): δ 1.63–1.71 (m, 1H), 1.92–2.02 (m, 2H), 2.10–2.19 (m, 1H), 2.79–2.84 (m, 2H), 3.08–3.12 (m, 1H), 3.86–3.94 (m, 2H), 4.39–4.44 (m, 1H), 7.26–7.32 (m, 2H), 7.37–7.42 (m, 1H), 7.45–7.51 (m, 2H). 13C NMR (100 MHz, CDCl3, 297 K): δ 25.9, 29.7, 29.8, 44.4, 68.8, 77.3, 126.6, 128.6, 129.1, 132.0, 176.5, 177.4. GC–MS: for tR = 15.75 min, m/z (%) 245 (38), 217 (10), 202 (58), 175 (100), 147 (10), 119 (28), 111 (11), 71 (98); for tR = 16.20 min, m/z (%) 245 (51), 217 (11), 202 (33), 175 (81), 147 (6), 119 (22), 111 (12), 71 (100). LR-ESIMS: m/z 246 [M + H]+. HR-ESIMS: m/z 246.1127 (calcd for C14H16NO3, 246.1125).
3-(Methylthiomethyl)-1-phenylpyrrolidine-2,5-dione (3j)
Methylthioacetic acid (70.8 mg, 0.67 mmol), N-phenylmaleimide (462.3 mg, 2.67 mmol), and TiO2 (80 mg, 1.00 mmol) were reacted in accordance with the general procedure. Following irradiation for 42 h, the reaction mixture was purified by column chromatography on silica gel (eluent: gradient of 20–40% EtOAc in petroleum ether 40–60) to yield 3j as a white powder (53.6 mg, 34%). Mp: 90–94 °C. 1H NMR (500 MHz, CDCl3, 299 K): δ 2.14 (s, 3H), 2.84 (dd, 1H, J = 5.0, 18.4 Hz), 2.92 (dd, 1H, J = 7.4, 13.5 Hz), 3.01 (dd, 1H, J = 9.2, 18.4 Hz), 3.04 (dd, 1H, J = 4.1, 13.5 Hz), 3.21–3.28 (m, 1H), 7.22–7.27 (m, 2H), 7.35–7.39 (m, 1H), 7.42–7.47 (m, 2H). 13C NMR (75 MHz, CDCl3, 297 K): δ 16.5, 33.7, 35.5, 40.1, 126.5, 128.7, 129.2, 131.9, 175.2, 177.5. LR-ESIMS: m/z 236 [M + H]+. HR-ESIMS: m/z 236.0741 (calcd for C12H14NO2S, 236.0740).
1-Phenyl-3-(thiophen-2-ylmethyl)pyrrolidine-2,5-dione (3k)
2-Thiopheneacetic acid (93.9 mg, 0.66 mmol), N-phenylmaleimide (462.3 mg, 2.67 mmol), and TiO2 (80 mg, 1.00 mmol) were reacted in accordance with the general procedure. Following irradiation for 17 h, the reaction mixture was purified by column chromatography on silica gel (eluent: 20% EtOAc in pentanes) to yield 3k as a yellow oil (45.9 mg, 26%). 1H NMR (500 MHz, CDCl3, 295 K): δ 2.72 (dd, J = 4.8, 18.4 Hz, 1H), 2.98 (dd, J = 9.2, 18.4 Hz, 1H), 3.30–3.35 (m, 1H), 3.37–3.45 (m, 2H), 6.89 (m, 1H), 6.98 (m, 1H), 7.20 (m, 2H), 7.39 (m, 1H), 7.46 (m, 2H). 13C NMR (75 MHz, CDCl3, 297 K) 30.7, 33.4, 41.3, 125.0, 126.5, 127.0, 127.3, 128.7, 129.2, 131.8, 138.2, 175.2, 177.8, 177.5. LR-ESIMS: m/z 272 [M + H]+. HR-ESIMS: m/z 272.0743 (calcd for C15H14NO2S, 272.0740). 1,2-Bis(thiophen-2-yl)ethane was also isolated as a clear oil (14.1 mg, 22%). 1H NMR (500 MHz, CDCl3, 295 K): δ 3.20 (s, 4H), 6.80 (d, J = 3.4 Hz, 2H), 6.91–6.94 (m, 2H), 7.14 (d, J = 5.2 Hz, 2H). Data are consistent with literature values.32
3-Benzyl-1-phenylpyrrolidine-2,5-dione (3l)
Phenylacetic acid (89.9 mg, 0.66 mmol), N-phenylmaleimide (462.3 mg, 2.67 mmol), and TiO2 (80 mg, 1.00 mmol) were reacted in accordance with the general procedure. Following irradiation for 22 h, the reaction mixture was purified by dry flash chromatography on silica gel (eluent: gradient of 10–20% EtOAc in CH2Cl2) to yield 3l as a white powder (99.3 mg, 57%). 1H NMR (500 MHz, CDCl3, 295 K): δ 2.65 (dd, J = 4.7, 18.5 Hz, 1H), 2.88 (dd, J = 9.2, 18.5 Hz, 1H), 3.08 (dd, J = 8.0, 13.8 Hz, 1H), 3.25 (dd, J = 4.4, 13.8 Hz, 1H), 3.30–3.35 (m, 1H), 7.17 (m, 2H), 7.22 (m, 2H), 7.29 (m, 1H), 7.34 (m, 2H), 7.39 (m, 1H), 7.46 (m, 2H). 13C NMR (75 MHz, CDCl3, 297 K): δ 33.5, 36.7, 41.4, 77.2, 126.6, 127.4, 128.8, 129.0, 129.3, 129.4, 132.0, 136.8, 175.4, 178.4. Data are consistent with literature values.33 Bibenzyl (10.9 mg, 18%) was observed by NMR prior to purification of the reaction mixture. 1H NMR (500 MHz, CDCl3, 298 K): δ 2.95 (s, 4H), 7.21–7.24 (m, 6H), 7.30–7.33 (m, 4H). Data are consistent with literature values.34
tert-Butyl (1-(2,5-Dioxo-1-phenylpyrrolidin-3-yl)ethyl)carbamate (3m)
N-Boc-l-alanine (126.2 mg, 0.67 mmol), N-phenylmaleimide (462.3 mg, 2.67 mmol), and TiO2 (80 mg, 1.00 mmol) were reacted in accordance with the general procedure. Following irradiation for 18 h, the mixture was purified by column chromatography on silica gel (eluent: gradient of 20–40% EtOAc in petroleum ether 40–60) to yield two diastereomers of 3m. Diastereomer 1 was isolated as a white powder (36.7 mg, 18%). Mp: 155–157 °C. 1H NMR (400 MHz, CDCl3, 296 K): δ 1.29 (d, J = 6.9 Hz, 3H), 1.38 (s, 9H), 2.78 (dd, J = 4.8, 18.6 Hz, 1H), 2.89 (dd, J = 9.1, 18.5 Hz, 1H), 3.15–3.20 (m, 1H), 4.08–4.18 (m, 1H), 4.53 (bs, 1H), 7.18–7.22 (m, 2H), 7.30–7.35 (m, 1H), 7.38–7.43 (m, 2H). 13C NMR (75 MHz, CDCl3, 297 K): δ 18.7, 28.3, 32.2, 45.3, 47.7, 80.2, 126.5, 128.7, 129.2, 131.8, 155.5, 175.3, 176.6. LR-ESIMS: m/z 341 [M + Na]+. HR-ESIMS: m/z 341.1467 (calcd for C17H22O4N2Na, 341.1472). Diastereomer 2 (20% wrt CH2Br2 standard) was obtained as a 1:1 mixture with N-phenylsuccinimide. 1H NMR (500 MHz, CDCl3, 297 K): δ 1.25 (d, J = 6.8 Hz, 3H), 1.44 (s, 9H), 2.63 (dd, J = 4.5, 18.6 Hz, 1H), 3.00 (dd, J = 9.6, 18.5 Hz, 1H), 3.17–3.23 (m, 1H), 4.13–4.17 (m, 1H), 5.20 (bs, 1H), 7.27–7.30 (m, 2H), 7.39–7.42 (m, 1H), 7.46–7.50 (m, 2H). 13C NMR (75 MHz, CDCl3, 297 K): δ 17.1, 28.5, 31.9, 45.1, 47.1, 80.0, 126.6, 128.8, 129.3, 131.8, 155.2, 175.2, 177.5. LR-ESIMS: m/z 341 [M + Na]+. HR-ESIMS: m/z 341.1475 (calcd for C17H22O4N2Na, 341.1472).
tert-Butyl 2′,5′-Dioxo-1′-phenyl-[2,3′-bipyrrolidine]-1-carboxylate (3n)
N-Boc-l-proline (144.3 mg, 0.67 mmol), N-phenylmaleimide (462.3 mg, 2.67 mmol), and TiO2 (80 mg, 1.00 mmol) were reacted in accordance with the general procedure. Following irradiation for 17 h, the crude mixture was purified by dry flash chromatography on silica gel (eluent: gradient of 20–60% EtOAc in petroleum ether 40–60) to yield an inseparable mixture of two diastereomers of 3n (171 mg, 75%, 1:1 ratio) that also contained 7% N-phenylsuccinimide. 1H NMR (500 MHz, CDCl3, 326 K): diastereomer 1 δ 1.44 (s, 9H), 1.94–1.99 (m, 2H), 2.18–2.24 (m, 2H), 2.73–2.75 (m, 1H), 2.83 (dd, J = 3.8, 9.4 Hz, 1H), 3.53 (dd, J = 7.3, 11.1 Hz, 1H), 3.79–3.85 (m, 2H), 4.34–4.39 (m, 2H), 7.27–7.47 (m, 5H); diastereomer 2 δ 1.49 (s, 9H), 2.14–2.17 (m, 2H), 1.66–1.71 (m, 2H), 2.69–2.71 (m, 1H), 2.86 (dd, J = 4.3, 9.7 Hz, 1H), 3.30 (dd, J = 3.4, 7.8 Hz, 1H), 3.95–3.99 (m, 2H), 4.31–4.35 (m, 2H), 7.27–7.47 (m, 5H). 13C NMR (75 MHz, CDCl3, 300 K): δ 23.3–24.0, 27.9, 28.4, 28.5, 29.7–30.1, 31.8, 43.7–44.1, 47.2–47.8, 57.1–57.8, 79.9–80.5, 126.5–126.8, 128.4–129.2, 131.2–132.0, 155.0–156.0, 174.1–177.3. GC–MS: for tR = 19.81 min, m/z (%) 344 (2), 288 (19), 271 (17), 243 (48), 175 (16), 114 (50), 70 (100) 57 (41); for tR = 20.09 min, m/z (%) 344 (2), 288 (19), 271 (17), 243 (48), 175 (16), 114 (50), 70 (100) 57 (41). LR-ESIMS: m/z 367 [M + Na]+. HR-ESIMS: m/z 367.1632 (calcd for C19H24N2O4Na, 367.1628).
tert-Butyl 2-(2,5-Dioxo-1-phenylpyrrolidin-3-yl)piperidine-1-carboxylate (3o)
N-Boc-d/l-2-piperidinecarboxylic acid (151.4 mg, 0.67 mmol), N-phenylmaleimide (462.3 mg, 2.67 mmol), and TiO2 (80 mg, 1.00 mmol) were reacted in accordance with the general procedure. Following irradiation for 18 h, the crude mixture was purified by column chromatography on silica gel (eluent: 30% EtOAc in pentanes) to yield two diastereomers of 3o. Diastereomer 1 was isolated as an off-white solid (32.7 mg, 14%). Mp: 107–110 °C. 1H NMR (500 MHz, CDCl3, 295 K): δ 1.39 (s, 9H), 1.46–1.59 (bm, 2H), 1.67–1.79 (bm, 4H), 2.52 (dd, J = 3.0, 18.2 Hz, 1H), 2.98 (dd, J = 8.9, 18.2 Hz, 1H), 3.04 (t, J = 13.2 Hz, 1H), 3.26–3.30 (m, 1H), 4.06 (d, J = 13.2 Hz, 1H), 4.34–4.37 (m, 1H), 7.32–7.36 (m, 3H), 7.41–7.45 (m, 2H). 13C NMR (75 MHz, CDCl3, 297 K): δ 18.9, 24.8, 27.0, 28.4, 34.2, 38.1, 40.2, 53.3, 79.9, 126.7, 128.4, 128.9, 132.1, 155.5, 174.9, 176.1. LR-ESIMS: m/z 381 [M + Na]+. HR-ESIMS: m/z 381.1787 (calcd for C20H26N2O4Na, 381.1785). Diastereomer 2 (15% wrt CH2Br2 standard) was obtained as a 1:2 mixture with N-phenylsuccinimide. 1H NMR (500 MHz, CDCl3, 295 K): δ 1.42 (s, 9H), 1.53 (m, 1H), 1.68–1.72 (m, 1H), 1.90–1.96 (m, 1H), 2.21–2.23 (m, 1H), 2.40–2.45 (m, 1H), 2.54–2.58 (m, 1H), 2.95 (dd, J = 4.4, 19.0 Hz, 1H), 3.05 (dd, J = 9.6, 17.6 Hz, 1H), 3.26 (dd, J = 9.1, 19.0 Hz, 1H), 3.35 (dd, J = 6.3, 17.6 Hz, 1H), 3.37–3.42 (m, 1H), 3.94–3.98 (m, 1H), 7.38–7.42 (m, 3H), 7.44–7.49 (m, 2H). 13C NMR (125 MHz, CDCl3, 295 K): δ 19.6, 24.4, 24.6, 28.3, 33.4, 37.8, 39.5, 46.1, 82.5, 126.4, 128.5, 129.1, 131.4, 152.6, 175.2, 175.5. LR-ESIMS: m/z 381 [M + Na]+. HR-ESIMS: m/z 381.1787 (calcd for C20H26N2O4Na, 381.1785).
Attempted Preparation of 2-(2,5-Dioxo-1-phenylpyrrolidin-3-yl)acetonitrile
Cyanoacetic acid (56.2 mg, 0.67 mmol), N-phenylmaleimide (462.3 mg, 2.67 mmol), and TiO2 (80 mg, 1.00 mmol) were reacted in accordance with the general procedure. Following irradiation for 18 h, the reaction mixture was scrutinized by 1H NMR and GC–MS, but none of the desired product was obtained.
N-Methyl-3-(phenoxymethyl)pyrrolidine-2,5-dione (7a) and N-Methyl-3a,4-dihydrochromeno[3,4-c]pyrrole-1,3(2H,9bH)-dione (6a)
Phenoxyacetic acid (100 mg, 0.65 mmol), N-methylmaleimide (145 mg, 1.3 mmol), and TiO2 (75 mg, 0.98 mmol) were reacted in accordance with the general procedure. Following irradiation for 20 h, the crude mixture was purified by column chromatography on silica gel (eluent: 20% EtOAc in petroleum ether 40–60) to yield 7a as a colorless oil (32.6 mg, 23%). 1H NMR (400 MHz, CDCl3, 297 K): δ 2.88–2.90 (m, 2H), 3.04 (s, 3H), 3.17–3.23 (m, 1H), 4.17 (dd, J = 3.4, 9.2 Hz, 1H), 4.40 (dd, J = 4.4, 9.2 Hz, 1H), 6.87 (d, J = 8.8 Hz, 2H), 6.98 (t, J = 7.4 Hz, 1H), 7.28 (t, J = 7.4 Hz, 2H). 13C NMR (75 MHz, CDCl3, 297 K): δ 25.4, 32.1, 41.1, 66.4, 115.1, 122.0, 130.0, 158.5, 177.0 178.0. 6a was also obtained as a white powder (77.8 mg, 55%). 1H NMR (400 MHz, CDCl3, 296 K): δ 3.00 (s, 3H), 3.36–3.40 (m, 1H), 4.01 (dd, J = 4.2, 11.4 Hz, 1H), 4.07 (d, J = 9.2 Hz, 1H), 4.62 (dd, J = 2.9, 11.4 Hz, 1H), 6.89 (d, J = 8.2 Hz, 1H), 7.05 (t, J = 7.5 Hz, 1H), 7.21 (t, J = 7.7 Hz, 1H), 7.58 (d, J = 7.4 Hz, 1H). 13C NMR (75 MHz, CDCl3, 297 K): δ 27.8, 40.1, 42.7, 64.3, 118.1, 118.3, 123.2, 129.4, 130.5, 155.7, 176.6, 177.2. Data are consistent with literature values.16
3-(Phenoxymethyl)-1-phenylpyrrolidine-2,5-dione (7b) and N-Phenyl-3a,4-dihydrochromeno[3,4-c]pyrrole-1,3(2H,9bH)-dione (6b)
Phenoxyacetic acid (100 mg, 0.65 mmol), N-phenylmaleimide (230 mg, 1.3 mmol), and TiO2 (75 mg, 0.98 mmol) were reacted in accordance with the general procedure. Following irradiation for 11 h, the crude mixture was purified by column chromatography on silica gel (eluent: 20% EtOAc in petroleum ether 40–60) to yield 7b as a yellow oil (71.5 mg, 39%). 1H NMR (400 MHz, CDCl3, 297 K): δ 3.01–3.07 (m, 2H), 3.33–3.39 (m, 1H), 4.28 (dd, J = 3.2, 9.0 Hz, 1H), 4.55 (dd, J = 4.1, 9.1 Hz, 1H), 6.91 (d, J = 8.8 Hz, 2H), 7.00 (t, J = 7.3 Hz, 1H), 7.28–7.33 (m, 2H), 7.41 (t, J = 7.4 Hz, 1H), 7.48 (d, J = 7.2, 2H). 13C NMR (75 MHz, CDCl3, 297 K): δ 32.4, 41.1, 66.9, 115.1, 122.1, 127.0, 129.2, 129.7, 130.0, 132.4, 158.5, 175.9, 177.1. 6b was also obtained as a white powder (72.8 mg, 40%). 1H NMR (400 MHz, CDCl3, 297 K): δ 3.54–3.58 (m, 1H), 4.11, (dd, J = 4.4, 11.4 Hz, 1H), 4.23 (d, J = 9.3 Hz, 1H), 4.7 (dd, J = 3.3, 11.4 Hz, 1H), 6.94 (d, J = 8.2 Hz, 1H), 7.07 (t, J = 7.5 Hz, 1H), 7.22–7.27 (m, 2H), 7.37 (t, J = 7.4 Hz, 2H), 7.44 (t, J = 7.1 Hz, 1H), 7.63 (d, J = 7.4 Hz, 1H). 13C NMR (75 MHz, CDCl3, 297 K): δ 40.2, 42.6, 64.4, 77.1, 77.5, 77.9, 117.7, 118.3, 123.3, 126.7, 129.2, 129.5, 129.6, 130.7, 132.1 155.7 175.5, 176.1. Data are consistent with literature values.16
3-((4-(tert-Butyl)phenoxy)methyl)-1-methylpyrrolidine-2,5-dione (7c) and 8-(tert-Butyl)-2-methyl-3a,4-dihydrochromeno[3,4-c]pyrrole-1,3(2H,9bH)-dione (6c)
4-(tert-Butyl)phenoxyacetic acid (130.3 mg, 0.62 mmol), N-methylmaleimide (275.6 mg, 2.48 mmol), and TiO2 (75 mg, 0.98 mmol) were reacted in accordance with the general procedure. Following irradiation for 22 h, the reaction mixture was purified by column chromatography on silica gel (eluent: 20% EtOAc in petroleum ether 40–60) to yield 7c as a clear oil (18.4 mg, 11%). 1H NMR (400 MHz, CDCl3, 296 K): δ 1.29 (s, 9H), 2.84–2.89 (m, 2H), 3.03 (s, 3H), 3.21–3.16 (m, 1H), 4.16 (dd, J = 3.4, 9.2 Hz, 1H), 4.39 (dd, J = 4.4, 9.2 Hz, 1H), 6.81 (d, J = 8.8 Hz, 2H), 7.29 (d, J = 8.8 Hz, 2H). 13C NMR (100 MHz, CDCl3, 296 K): δ 25.0, 29.7, 31.5, 34.1, 40.7, 66.1, 114.2, 126.4, 144.4, 155.8, 176.6, 177.6. LR-ESIMS: m/z 276 [M + H]+. HR-ESIMS: m/z 276.1599 (calcd for C16H22NO3, 276.1594). 6c was also obtained as a white powder (77.2 mg, 46%). Mp: 134–137 °C. 1H NMR (400 MHz, CDCl3, 296 K): δ 1.32 (s, 9H), 2.98 (s, 3H), 3.33–3.37 (m, 1H), 3.98 (dd, J = 4.1, 11.3 Hz, 1H), 4.05 (d, J = 9.2 Hz, 1H), 4.59 (dd, J = 3.0, 11.3 Hz, 1H), 6.82 (d, J = 8.6 Hz, 1H), 7.23 (dd, J = 2.4, 8.6 Hz, 1H), 7.58 (d, J = 2.4 Hz, 1H). 13C NMR (75 MHz, CDCl3, 296 K): δ 25.8, 31.9, 34.8, 40.3, 42.7, 64.3, 117.1, 117.6, 126.5, 127.3, 146.0, 153.3, 176.7, 177.3. LR-ESIMS: m/z 274 [M + H]+. HR-ESIMS: m/z 274.1442 (calcd for C16H20NO3, 274.1438).
3-((4-(tert-Butyl)phenoxy)methyl)-1-phenylpyrrolidine-2,5-dione (7d) and 8-(tert-Butyl)-2-phenyl-3a,4-dihydrochromeno[3,4-c]pyrrole-1,3(2H,9bH)-dione (6d)
4-(tert-Butyl)phenoxyacetic acid (70 mg, 0.33 mmol), N-phenylmaleimide (117 mg, 0.66 mmol), and TiO2 (40 mg, 0.5 mmol) were reacted in accordance with the general procedure. Following irradiation for 19 h, the crude mixture was purified by column chromatography on silica gel (eluent: 20% EtOAc in petroleum ether 40–60) to yield 7d as a clear oil (31.2 mg, 28%). 1H NMR (300 MHz, CDCl3, 295 K): δ 1.30 (s, 9H), 2.98–3.13 (m, 2H), 3.30–3.37 (m, 1H), 4.23 (dd, J = 3.2, 9.1 Hz, 1H), 4.53 (dd, J = 3.9, 9.1 Hz, 1H), 6.85 (d, J = 8.9 Hz, 2H), 7.27–7.33 (m, 3H), 7.40–7.51 (m, 4H). 13C NMR (75 MHz, CDCl3, 297 K): δ 29.7, 31.5, 34.2, 40.8, 66.6, 114.2, 126.4, 126.6, 128.7, 129.2, 132.0, 144.6, 155.8, 175.6, 176.8. LR-ESIMS: m/z 338 [M + H]+. HR-ESIMS: m/z 338.1755 (calcd for C21H24NO3, 338.1751). 6d was also obtained as a clear oil (50.3 mg, 46%). 1H NMR (300 MHz, CDCl3, 296 K): δ 1.32 (s, 9H), 3.51–3.56 (m, 1H), 4.10 (dd, J = 4.3, 11.4 Hz, 1H), 4.23 (d, J = 7.3 Hz, 1H), 4.66 (dd, J = 3.3, 11.5 Hz, 1H), 6.87 (d, J = 8.1 Hz, 1H), 7.24–7.29 (m, 2H), 7.33–7.48 (m, 4H). 13C NMR (75 MHz, CDCl3, 297 K): δ 31.5, 34.4, 40.0, 42.3, 64.1, 116.4, 117.3, 126.3, 127.0, 128.7, 129.1, 131.8, 134.2, 145.7, 153.0, 175.2, 175.7. LR-ESIMS: m/z 336 [M + H]+. HR-ESIMS: m/z 336.1598 (calcd for C21H22NO3, 336.1600).
1-Methyl-3-((4-(trifluoromethyl)phenoxy)methyl)pyrrolidine-2,5-dione (7e) and 2-Methyl-8-(trifluoromethyl)-3a,4-dihydrochromeno[3,4-c]pyrrole-1,3(2H,9bH)-dione (6e)
4-(Trifluoromethyl)phenoxyacetic acid (143 mg, 0.65 mmol), N-methylmaleimide (145 mg, 1.3 mmol), and TiO2 (75 mg, 0.98 mmol) were reacted in accordance with the general procedure. Following irradiation for 16 h, the reaction mixture was purified by two rounds of column chromatography on silica gel (eluent: 20% EtOAc in petroleum ether 40–60 and subsequently 10% EtOAc in CH2Cl2) to yield 7e as a clear oil (54.7 mg, 29%). 1H NMR (400 MHz, CDCl3, 300 K): δ 2.82–2.96 (m, 2H), 3.05 (s, 3H), 3.20–3.26 (m, 1H), 4.22 (dd, J = 3.4, 9.2 Hz, 1H), 4.46 (dd, J = 4.3, 9.2 Hz, 1H), 6.94 (d, J = 8.5 Hz, 2H), 7.54 (d, J = 8.5 Hz, 2H). 13C NMR (75 MHz, CDCl3, 300 K): δ 25.1, 31.6, 40.5, 66.1, 114.6, 123.9 (q, 2JC–F = 32.9 Hz, 1C), 124.4 (q, 3JC–F = 271.2 Hz, 2C), 127.0 (q, 1JC–F = 3.7 Hz, 1C), 160.4, 176.2, 177.1. 19F NMR (376 MHz, CDCl3, 300 K): δ −62.1. LR-ESIMS: m/z 288 [M + H]+. HR-ESIMS: m/z 288.0838 (calcd for C13H13NO3F3, 288.0842). 6e was also obtained as a clear oil (60.1 mg, 32%). 1H NMR (400 MHz, CDCl3, 298 K): δ 3.01 (s, 3H), 3.40–3.44 (m, 1H), 4.04–4.10 (m, 2H), 4.64 (dd, J = 3.2, 11.5 Hz, 1H), 6.98 (d, J = 8.6 Hz, 1H), 7.46 (d, J = 8.5 Hz, 1H), 7.87 (s, 1H). 13C NMR (75 MHz, CDCl3, 298 K): δ 25.5, 39.4, 41.8, 64.0, 117.9, 118.4, 123.9 (q, 1JC–F = 271.7 Hz, 1C), 125.1 (q, 2JC–F = 33.1 Hz, 1C), 127.2 (q, 3JC–F = 3.5 Hz, 1C), 127.6 (q, 3JC–F = 3.8 Hz, 1C), 157.7, 175.3, 176.1. 19F NMR (376 MHz, CDCl3, 298 K): δ −62.3. LR-ESIMS: m/z 286 [M + H]+. HR-ESIMS: m/z 286.0687 (calcd for C13H11NO3F3, 286.0686).
1-Phenyl-3-((4-(trifluoromethyl)phenoxy)methyl)pyrrolidine-2,5-dione (7f) and 8-(Trifluoromethyl)-2-phenyl-3a,4-dihydrochromeno[3,4-c]pyrrole-1,3(2H,9bH)-dione (6f)
4-(Trifluoromethyl)phenoxyacetic acid (143 mg, 0.65 mmol), N-phenylmaleimide (230 mg, 1.3 mmol), and TiO2 (75 mg, 0.98 mmol) were reacted in accordance with the general procedure. Following irradiation for 16 h, the reaction mixture was purified by column chromatography on silica gel (eluent: 25% EtOAc in petroleum ether 40–60) to yield 7f as a colorless solid (52.9 mg, 40%). Mp: 121–124 °C. 1H NMR (400 MHz, CDCl3, 297 K): δ 2.98–3.13 (m, 2H), 3.35–3.40 (m, 1H), 4.28 (dd, J = 3.1, 9.1 Hz, 1H), 4.57 (dd, J = 4.0, 9.1 Hz, 1H), 6.97 (d, J = 8.5 Hz, 2H), 7.29 (d, J = 7.2 Hz, 2H), 7.41 (t, J = 7.3 Hz, 1H), 7.49 (t, J = 7.2 Hz, 2H), 7.56 (d, J = 8.6 Hz, 2H). 13C NMR (75 MHz, CDCl3, 297 K): δ 31.9, 40.6, 66.6, 114.7, 124.0 (q, 2JC–F = 32.8 Hz, 1C), 124.3 (q, 1JC–F = 271.1 Hz, 1C), 126.5, 127.1 (q, 3JC–F = 3.7 Hz, 2C), 128.8, 129.3, 131.9, 160.4, 175.2, 176.3. 19F NMR (376 MHz, CDCl3, 297 K): δ −62.1. LR-ESIMS: m/z 367 [M + NH4]+. HR-ESIMS: m/z 367.1269 (calcd for C18H18N2O3F3, 367.1275). 6f was also obtained as a yellow oil (90 mg, 23%). 1H NMR (400 MHz, CDCl3, 297 K): δ 3.57–3.61 (m, 1H), 4.15 (dd, J = 4.2, 11.4 Hz, 1H), 4.25 (d, J = 9.3 Hz, 1H), 4.71 (dd, J = 3.4, 11.4 Hz, 1H), 7.04 (d, J = 8.5 Hz, 1H), 7.24–7.27 (m, 2H), 7.36–7.51 (m, 4H), 7.93 (s, 1H). 13C NMR (75 MHz, CDCl3, 297 K): δ 39.5, 41.7, 64.1, 117.7, 118.5, 123.9 (q, 1JC–F = 271.8 Hz, 1C), 125.2 (q, 2JC–F = 33.0 Hz, 1C), 126.2, 126.4 (q, 3JC–F = 3.5 Hz, 1C), 127.7 (q, 3JC–F = 3.7 Hz, 1C), 128.9, 129.2, 131.5, 157.8, 174.3, 175.0. 19F NMR (376 MHz, CDCl3, 297 K): δ −62.3. LR-ESIMS: m/z 365 [M + NH4]+. HR-ESIMS: m/z 365.1114 (calcd for C18H16N2O3F3, 365.1108).
(E)-2-(2-(3-Methoxy-3-oxoprop-1-en-1-yl)phenoxy)acetic Acid (8a)
A THF (60 mL) solution of methyl bromoacetate (0.62 mL, 6.54 mmol, 1.0 equiv) and triphenylphosphine (1.715 g, 6.54 mmol, 1.0 equiv) was heated at reflux for 4 h until a white precipitate formed. The suspension was allowed to cool to room temperature before filtration; the precipitate was washed with Et2O (3 × 50 mL), and the precipitate was then redissolved in CH2Cl2 (30 mL) and H2O (20 mL). Then 2 M NaOH (10 mL) was added, and the biphasic mixture was stirred overnight. The reaction mixture was extracted with CH2Cl2 (3 × 100 mL), and the combined organic layers were washed with brine, dried (MgSO4), and concentrated under reduced pressure to yield the phosphorane as a colorless powder (1.923 g, 88%). 1H NMR (400 MHz, CDCl3, 295 K): δ 2.90 and 3.51 (br, 3H), 7.43–7.48 (m, 6H), 7.52–7.57 (m, 3H), 7.62–7.70 (m, 6H). 13C NMR (100 MHz, CDCl3, 298 K): δ 49.9, 128.5 (d, J = 12.0 Hz), 128.8 (d, J = 12.2 Hz), 131.9 (d, J = 2.7 Hz), 132.1 (d, J = 9.8 Hz), 132.9, 133.0 (d, J = 10.2 Hz). 31P NMR (161 MHz, CDCl3, 295 K): δ 17.73. LR-ESIMS: m/z 335 [M + H]+. HR-ESIMS: m/z 335.1200 (calcd for C21H20O2P, 335.1195). A CHCl3 (200 mL) solution of phosphorane (1.893 g, 5.66 mmol, 1.1 equiv) and 2-formylphenoxyacetic acid (0.927 g, 5.15 mmol, 1.0 equiv) was heated at reflux for 18 h. The solvent was removed under reduced pressure, and the crude residue was purified by column chromatography (eluent: gradient of petroleum ether to 1:1 EtOAc/petroleum ether) to yield 8a as a colorless powder (1.037 g, 85%). Mp: 96 °C. 1H NMR (300 MHz, CDCl3, 295 K): δ 3.80 (s, 3H), 4.77 (s, 2H), 6.61 (d, J = 16.2 Hz, 1H), 6.81 (d, J = 8.3 Hz, 1H), 7.03 (t, J = 7.6 Hz, 1H), 7.33 (td, J = 1.7, 7.5 Hz, 1H), 7.54 (dd, J = 1.6, 7.7 Hz, 1H), 8.04 (d, J = 16.2 Hz, 1H), 8.89–9.30 (br, 1H). 13C NMR (75 MHz, CDCl3, 298 K): δ 52.2, 65.4, 112.5, 119.5, 122.5, 124.4, 129.8, 131.8, 140.4, 156.7, 168.6, 173.8. LR-ESIMS: m/z 235 [M – H]−. HR-ESIMS: m/z 235.0610 (calcd for C12H11O5, 235.0612).
(E)-2-(2-(2-Cyanovinyl)phenoxy)acetic Acid (8b)
Prepared in the same manner as 8a. Bromoacetonitrile (0.88 mL, 12.5 mmol) and PPh3 (3.28 g, 12.5 mL) were reacted to give the salt (4.580 g, 96%). The phosphorane was prepared as before to give a colorless powder (3.422 g, 91%). 1H NMR (300 MHz, CDCl3, 297 K): δ 7.45–7.70 (m, 15H). 13C NMR (121 MHz, CDCl3, 297 K): δ 128.9 (d, J = 12.1 Hz), 129.5 (d, J = 12.4 Hz), 132.3 (d, J = 2.6 Hz), 132.5 (d, J = 9.9 Hz), 133.0 (d, J = 2.1 Hz), 133.2 (d, J = 10.1 Hz). 31P NMR (121 MHz, CDCl3, 295 K): δ 23.3. 8b was prepared from the phosphorane (3.346 g, 11.11 mmol) and 2-formylphenoxyacetic acid (1.819 g, 10.10 mmol) to give a colorless powder (1.477 g, 72%). Mp: 118 C°. 1H NMR (400 MHz, CDCl3, 297 K): δ 4.67 (s, 3H), 6.31 (d, J = 16.8 Hz, 1H), 6.83 (d, J = 1.1 Hz, 1H), 7.01 (t, J = 7.6 Hz, 1H), 7.33–7.39 (m, 2H), 7.62 (d, J = 16.8 Hz, 1H). 13C NMR (100 MHz, CDCl3, 297 K): δ 65.1, 98.0, 112.2, 119.1, 121.9, 123.0, 129.8, 132.2, 146.5, 156.5, 170.5. LR-ESIMS: m/z 202 [M – H]−. HR-ESIMS: m/z 202.0511 (calcd for C11H8O3N1, 202.0510).
(E)-2-(2-(3-Oxopent-1-en-1-yl)phenoxy)acetic Acid (8c)
Prepared in the same manner as 8a. 1-Bromo-2-butanone (1.0 g, 6.62 mmol) and PPh3 (1.737 g, 6.62 mmol) were reacted to give the salt (2.613 g, 96%). The phosphorane was prepared as before (2.051 g, 93%). 1H NMR (500 MHz, CDCl3, 295 K): δ 1.18 (t, J = 7.6 Hz, 3H), 2.33 (qd, J = 1.3, 7.6 Hz, 2H), 7.43–7.47 (m, 6H), 7.52–7.56 (m, 3H), 7.62–7.67 (m, 6H). 8c was prepared from the phosphorane (1.712 g, 5.15 mmol) and 2-formylphenoxyacetic acid (0.843 g, 4.70 mmol) to give a colorless powder (0.829 g, 75%). Mp: 125 °C. 1H NMR (400 MHz, CDCl3, 295 K): δ 1.17 (t, J = 7.3 Hz, 3H), 2.73 (q, J = 7.3 Hz, 2H), 4.78 (s, 2H), 6.82 (d, J = 6.82 Hz, 1H), 6.90 (d, J = 16.4 Hz, 1H), 7.04 (t, J = 7.1 Hz, 1H), 7.35 (td, J = 1.7, 7.4 Hz, 1H), 7.58 (dd, J = 1.7, 7.7 Hz, 1H), 7.96 (d, J = 16.4 Hz, 1H). 13C NMR (100 MHz, CDCl3, 298 K): δ 8.3, 33.5, 65.4, 112.2, 121.8, 124.0, 127.2, 128.9, 131.5, 137.8, 156.7, 170.6, 202.5. LR-ESIMS: m/z 233 [M – H]−. HR-ESIMS: m/z 233.0820 (calcd for C13H13O4, 233.0819).
2-([1,1′-Biphenyl]-2-yloxy)acetic Acid (11a)
2-Bromophenol (6.4 mL, 60.5 mmol), tert-butyldimethylsilyl chloride (10.0 g, 66.5 mmol), and imidazole (8.23 g, 121.0 mmol) were dissolved in the minimum volume of DMF, and the mixture was allowed to stir at room temperature overnight. The DMF was removed under reduced pressure, and the resultant residue was taken up in EtOAc and washed with 1 M HCl, sat. NaHCO3, and sat. LiCl before being dried over MgSO4. The solvent was then removed under reduced pressure to yield (2-bromophenoxy)(tert-butyl)dimethylsilane (17.29 g, 99%). 1H NMR (400 MHz, CDCl3, 296 K): δ 0.25 (s, 6H), 1.05 (s, 9H), 6.80–6.84 (m, 1H), 6.87 (dd, J = 1.5, 8.1 Hz, 1H), 7.14–7.19 (m, 1H), 7.51 (dd, J = 1.7, 7.9 Hz, 1H). 13C NMR (100 MHz, CDCl3, 297 K): δ −4.2, 18.4, 25.8, 115.4, 120.3, 122.4, 128.2, 133.4, 152.6. LR-ESIMS: m/z 287 [M + H]+. HR-ESIMS: m/z 287.0461 (calcd for C12H20OBrSi, 287.0461). The aforementioned silane (0.54 g, 1.9 mmol) was coupled with phenylboronic acid (0.27 g, 2.2 mmol) by refluxing overnight in toluene (20 mL) and ethanol (5 mL) with Pd[P(Ph)3]4 (0.2 g, 0.2 mmol) and K2CO3 (0.9 g, 6.5 mmol). The solvent was then removed under reduced pressure, and the reaction mixture was taken up in H2O before being extracted with EtOAc. The combined extracts were dried over MgSO4 before being concentrated under reduced pressure. The resultant mixture was subjected to column chromatography on silica gel (eluent: 25% CH2Cl2 in petroleum ether 40–60) to yield [1,1′-biphenyl]-2-yloxy)(tert-butyl)dimethylsilane (0.39 g, 73%). 1H NMR (400 MHz, CDCl3, 296 K): δ −0.05 (s, 6H), 0.83 (s, 9H), 6.92 (dd, J = 1.2, 8.0 Hz, 1H), 7.04 (td, J = 1.2, 7.5 Hz, 1H), 7.21–7.24 (m, 1H), 7.30–7.33 (m, 2H), 7.36–7.40 (m, 2H), 7.50 (dd, J = 1.5, 8.5 Hz, 1H). 13C NMR (100 MHz, CDCl3, 297 K): δ −4.6, 18.1, 25.6, 120.4, 121.6, 126.7, 127.8, 128.3, 129.8, 130.9, 133.6, 139.2, 152.6. The aforementioned biphenylsilane (0.36 g, 1.3 mmol) was treated overnight with acetyl chloride (0.3 mL, 0.4 mmol) in methanol (15 mL). Following removal of the solvent under reduced pressure, the reaction mixture was subjected to column chromatography on silica gel (eluent: CH2Cl2) to yield [1,1′-biphenyl]-2-ol (0.19 g, 86%). 1H NMR (400 MHz, CDCl3, 296 K): δ 5.13 (bs, 1H), 6.88–6.92 (m, 2H), 7.14–7.19 (m, 2H), 7.28–7.32 (m, 1H), 7.36–7.42 (m, 4H). The aforementioned biphenyl alcohol (0.19 g, 1.11 mmol), methyl bromoacetate (0.16 mL, 0.26 mmol), and K2CO3 (0.76 g, 5.5 mmol) were refluxed overnight in THF (20 mL). The solvent was then removed under reduced pressure, and the resultant residue was taken up in CH2Cl2 before being washed with H2O and dried over MgSO4. The solvent was removed under reduced pressure, and the resultant mixture was subjected to column chromatography on silica gel (eluent: 25% CH2Cl2 in petroleum ether 40–60) to afford methyl 2-([1,1′-biphenyl]-2-yloxy)acetate (0.25 g, 94%). 1H NMR (300 MHz, CDCl3, 294 K): δ 3.82 (s, 3H), 4.66 (s, 2H), 6.94 (d, J = 8.3 Hz, 1H), 7.15 (td, J = 1.0, 7.5 Hz, 1H), 7.33–7.51 (m, 5H), 7.69 (dd, J = 1.4, 8.4 Hz, 1H). 13C NMR (75 MHz, CDCl3, 295 K): δ 52.2, 65.8, 112.8, 122.2, 127.1, 128.1, 128.6, 129.7, 131.3, 131.4, 138.1, 154.8, 169.6. LR-ESIMS: m/z 243 [M + H]+. HR-ESIMS: m/z 243.1020 (calcd for C15H15O3, 243.1016). The aforementioned ester (0.19 g, 0.8 mmol) was hydrolyzed with LiOH (0.13 g, 3.2 mmol) in a 3:1 mixture of MeOH and H2O. Following overnight stirring at room temperature, the reaction mixture was concentrated to ca. 10% of its original volume, diluted with sat. (NH3)2SO4 (30 mL), adjusted to pH 3 with conc. H2SO4, and extracted with EtOAc (3 × 30 mL). The combined extracts were dried over MgSO4 and concentrated under reduced pressure, and the resultant residue was chromatographed on silica gel (eluent: CH2Cl2) to yield 11a (0.13 g, 73%). Mp: 84 °C. 1H NMR (300 MHz, CDCl3, 294 K): δ 4.63 (s, 2H), 6.92 (dd, J = 1.1, 8.2 Hz, 1H), 7.13 (td, J = 1.1, 7.5 Hz, 1H), 7.29–7.46 (m, 5H), 7.57 (dd, J = 1.5, 8.4 Hz, 2H), 8.80–10.10 (br, 1H). 13C NMR (75 MHz, CDCl3, 295 K): δ 65.9, 113.4, 123.1, 127.7, 128.7, 129.1, 129.9, 131.8, 131.9, 138.3, 154.6, 173.7. LR-ESIMS: m/z 227 [M – H]−. HR-ESIMS: m/z 227.0707 (calcd for C14H11O3, 227.0714).
2-((3′-(Trifluoromethyl)-[1,1′-biphenyl]-2-yl)oxy)acetic Acid (11b)
Prepared in the same manner as 11a. (2-Bromophenoxy)(tert-butyl)dimethylsilane (0.95 g, 3.3 mmol), 4-(trifluoromethyl)phenylboronic acid (0.75 g, 4.0 mmol), Pd[P(Ph)3]4 (0.38 g, 0.33 mmol), and K2CO3 (1.2 g, 9.9 mmol) were reacted and then columned on silica gel (eluent: 25% CH2Cl2 in petroleum ether 40–60) to afford 3′-(trifluoromethyl)-[1,1′-biphenyl]-2-ol (0.13 g, 14%). 1H NMR (400 MHz, CDCl3, 296 K): δ 4.98–5.10 (br, 1H), 6.96 (d, J = 8.5 Hz, 1H), 7.03 (td, J = 1.1, 7.5 Hz, 1H), 7.26–7.31 (m, 2H), 7.57–7.65 (m, 2H), 7.71 (d, J = 7.5 Hz, 1H), 7.78 (s, 1H). 19F NMR (376 MHz, CDCl3, 296 K): δ −63.0. 13C NMR (100 MHz, CDCl3, 297 K): δ 116.2, 121.3, 126.9, 129.3, 129.7, 130.5, 132.5, 138.3, 152.4. LR-ESIMS: m/z 237 [M – H]−. HR-ESIMS: m/z 237.0526 (calcd for C13H8OF3, 237.0533). The aforementioned biphenyl alcohol (0.13 g, 0.55 mmol) was alkylated as before with methyl bromoacetate (0.1 mL, 1.1 mmol) and K2CO3 (0.38 g, 2.7 mmol) and columned on silica gel (eluent: 50% CH2Cl2 in petroleum ether 40–60) to afford methyl 2-((3′-(trifluoromethyl)-[1,1′-biphenyl]-2-yl)oxy)acetate (0.14 g, 84%). 1H NMR (400 MHz, CDCl3, 296 K): δ 3.79 (s, 3H), 4.65 (s, 2H), 6.89 (d, J = 7.7 Hz, 1H), 7.12 (td, J = 1.0, 7.5 Hz), 7.32–7.39 (m, 2H), 7.54 (t, J = 7.7 Hz, 1H), 7.60 (d, J = 7.8 Hz, 1H), 7.81 (d, J = 7.6 Hz, 1H), 7.92 (s, 1H). 19F NMR (376 MHz, CDCl3, 296 K): δ −62.9. 13C NMR (100 MHz, CDCl3, 297 K): δ 52.3, 65.5, 112.3, 122.3, 123.8 (q, 3JCF = 3.7 Hz), 124.4 (q, 1JCF = 272.2 Hz), 126.5 (q, 3JCF = 3.7 Hz), 128.5, 129.4, 129.7, 130.4 (q, 2JCF = 32.1 Hz), 131.2, 133.0, 138.8, 154.6, 169.2. LR-ESIMS: m/z 328 [M + NH4]+. HR-ESIMS: m/z 328.1162 (calcd for C16H17NO3F3, 328.1155). The aforementioned ester (0.14 g, 0.5 mmol) was hydrolyzed with LiOH (0.08 g, 1.8 mmol) to afford 11b (0.13g, 95%). 1H NMR (400 MHz, CDCl3, 296 K): δ 4.68 (s, 2H), 6.91 (d, J = 8.2 Hz, 1H), 7.14 (td, J = 1.0, 7.5 Hz, 1H), 7.34–7.39 (m, 2H), 7.54 (t, J = 7.7 Hz, 1H), 7.60 (d, J = 7.8 Hz, 1H), 7.76 (d, J = 7.6 Hz, 1H), 7.87 (s, 1H). 19F NMR (376 MHz, CDCl3, 296 K): δ −63.0. 13C NMR (100 MHz, CDCl3, 297 K): δ 65.0, 112.4, 122.6, 123.9 (q, 3JCF = 3.3 Hz), 124.2 (q, 1JCF = 272.4 Hz), 126.4 (q, 3JCF = 3.6 Hz), 128.5, 129.4, 129.7, 130.6 (q, 2JCF = 32.5 Hz), 131.2, 132.8, 138.6, 154.1, 173.0. LR-ESIMS: m/z 314 [M + NH4]+. HR-ESIMS: m/z 314.1001 (calcd for C15H15NO3F3, 314.0999).
2-((4′-(Trifluoromethyl)-[1,1′-biphenyl]-2-yl)oxy)acetic Acid (11c)
Prepared in the same manner as 11a. (2-Bromophenoxy)(tert-butyl)dimethylsilane (0.63 g, 2.2 mmol), 4-(trifluoromethyl)phenylboronic acid (0.5 g, 2.6 mmol), Pd[P(Ph)3]4 (0.22 g, 0.22 mmol), and K2CO3 (0.9 g, 6.5 mmol) were reacted and then columned on silica gel (eluent: 25% CH2Cl2 in petroleum ether 40–60) to afford 4′-(trifluoromethyl)-[1,1′-biphenyl]-2-ol (0.43 g, 82%). Mp: 99 °C. 1H NMR (400 MHz, CDCl3, 296 K): δ 4.99 (s, 1H), 6.97 (dd, J = 0.5, 8.1 Hz, 1H), 7.04 (td, J = 1.1, 7.5 Hz, 1H), 7.26–7.32 (m, 2H), 7.64 (d, J = 8.6 Hz, 2H), 7.74 (d, J = 8.6 Hz, 2H). 19F NMR (376 MHz, CDCl3, 296 K): δ −63.05. 13C NMR (100 MHz, CDCl3, 297 K): δ 116.2, 121.3, 124.1 (q, 1JCF = 272.0 Hz), 125.9 (q, 3JCF = 3.7 Hz), 126.9, 129.4 (q, 2JCF = 32.4 Hz), 129.6, 129.8, 130.4, 141.1, 152.3. LR-ESIMS: m/z 237 [M – H]−. HR-ESIMS: m/z 237.0539 (calcd for C13H8OF3, 237.0533). The aforementioned biphenyl alcohol (0.4 g, 1.66 mmol) was alkylated as before with methyl bromoacetate (0.24 mL, 2.5 mmol) and K2CO3 (1.15 g, 8.3 mmol) and columned on silica gel (eluent: 25% CH2Cl2 in petroleum ether 40–60) to afford methyl 2-((4′-(trifluoromethyl)-[1,1′-biphenyl]-2-yl)oxy)acetate (0.529 g, 99%). 1H NMR (400 MHz, CDCl3, 296 K): δ 3.78 (s, 3H), 4.64 (s, 2H), 6.88 (d, J = 8.2 Hz, 1H), 7.10 (td, J = 1.0, 7.5 Hz, 1H), 7.32–7.36 (m, 2H), 7.67 (d, J = 8.1 Hz, 2H), 7.73 (d, J = 8.1 Hz, 2H). 19F NMR (376 MHz, CDCl3, 296 K): δ −62.9. 13C NMR (100 MHz, CDCl3, 297 K): δ 52.2, 65.5, 112.3, 122.2, 124.4 (q, 1JCF = 271.6 Hz), 124.9 (q, 3JCF = 3.7 Hz), 129.1 (q, 2JCF = 32.4 Hz), 129.4, 129.8, 129.9, 131.2, 141.7, 154.6, 169.2. LR-ESIMS: m/z 328 [M + NH4]+. HR-ESIMS: m/z 328.1158 (calcd for C16H17NO3F3, 328.1155). The aforementioned ester (0.49 g, 1.6 mmol) was hydrolyzed with LiOH (0.26 g, 6.3 mmol) to afford 11c (0.44g, 95%). Mp: 126 °C. 1H NMR (400 MHz, CDCl3, 296 K): δ 4.68 (s, 2H), 6.91 (dd, J = 1.1, 8.9 Hz, 1H), 7.13 (td, J = 1.0, 7.6 Hz, 1H), 7.33–7.39 (m, 2H), 7.66–7.72 (m, 4H). 19F NMR (376 MHz, CDCl3, 296 K): δ −62.9. 13C NMR (100 MHz, CDCl3, 297 K): δ 65.5, 112.9, 123.0, 125.0 (q, 1JCF = 273.5 Hz), 125.4 (q, 3JCF = 3.9 Hz), 129.7 (q, 2JCF = 31.2 Hz), 129.9, 130.3 (×2), 131.7, 142.0, 154.6, 173.2. LR-ESIMS: m/z 295 [M – H]−. HR-ESIMS: m/z 295.0592 (calcd for C15H10O3F3, 295.0588).
2-(2-((Methoxyimino)methyl)phenoxy)acetic Acid (13a)
A suspension of 2-formylphenoxyacetic acid (0.515 g, 2.86 mmol, 1.0 equiv), methoxylamine hydrochloride (0.478 g, 5.72 mmol, 2.0 equiv), and sodium acetate (0.469 g, 5.72 mmol, 2.0 equiv) in ethanol (30 mL) was heated at reflux for 18 h. The solvent was removed under reduced pressure, and the crude residue was redissolved in H2O (100 mL) and extracted into CH2Cl2 (3 × 100 mL). The combined organic layers were dried (MgSO4), concentrated under reduced pressure, and purified by column chromatography (5% EtOAc/CH2Cl2) to yield 13a as a colorless solid (0.390 g, 65%). 1H NMR (300 MHz, CDCl3, 294 K): δ 4.01 (s, 3H), 4.72 (s, 2H), 6.91 (d, J = 8.2 Hz, 1H), 7.12 (m, 1H), 7.34 (d, J = 7.6 Hz, 1H), 7.40 (m, 1H), 8.15 (s, 1H). 13C NMR (75 MHz, CDCl3, 295 K): δ 61.9, 66.6, 114.2, 120.6, 123.0, 131.7, 132.4, 148.8, 155.4, 169.5. LR-ESIMS: m/z 208 [M – H]−. HR-ESIMS: m/z 208.0614 (calcd for C10H10NO4, 208.0615).
2-(2-(((Benzyloxy)imino)methyl)phenoxy)acetic Acid (13b)
A suspension of 2-formylphenoxyacetic acid (1.00 g, 5.55 mmol, 1.0 equiv), benzoxylamine hydrochloride (1.773 g, 11.11 mmol, 2.0 equiv), and sodium acetate (0.911 g, 11.11 mmol, 2.0 equiv) in ethanol (60 mL) was heated at reflux for 18 h. The solvent was removed under reduced pressure, and the crude residue was redissolved in H2O (100 mL) and extracted into CH2Cl2 (3 × 100 mL). The combined organic layers were dried (MgSO4), concentrated under reduced pressure, and purified by column chromatography (5% EtOAc/CH2Cl2) to yield 13b as a colorless solid (0.422 g, 26%). 1H NMR (300 MHz, CDCl3, 294 K): δ 4.70 (s, 2H), 5.25 (s, 2H), 6.90 (d, J = 8.2 Hz, 1H), 7.11 (t, J = 6.6 Hz, 1H), 7.32–7.44 (m, 7H), 8.26 (s, 1H). 13C NMR (75 MHz, CDCl3, 295 K): δ 66.5, 76.2, 114.0, 120.6, 122.9, 128.2, 128.5, 128.5, 131.7, 132.2, 136.6, 149.3, 155.3, 169.5. LR-ESIMS: m/z 284 [M – H]−. HR-ESIMS: m/z 284.0923 (calcd for C16H14NO4, 284.0928).
d1-Phenoxyacetic Acid (5aD)
Phenoxyacetic acid (304 mg, 2.0 mmol) and SOCl2 (0.74 mL, 10.0 mmol) were refluxed in CHCl3 for 72 h. The volatiles were removed under reduced pressure to yield phenoxyacetyl chloride. 1H NMR (400 MHz, CDCl3, 296 K): δ 4.95 (s, 2H), 6.93 (m, 2H), 7.09 (m, 1H), 7.35 (m, 2H). 13C NMR (75 MHz, CDCl3, 297 K): δ 64.9, 77.2, 114.8, 122.3, 129.9, 157.5, 174.2. This compound was immediately treated with D2O (0.9 mL, 5.0 mmol) in anhydrous CH3CN (10 mL), and the mixture was allowed to stir overnight under an argon atmosphere. The solvent was removed under reduced pressure, and the resultant residue was recrystallized from anhydrous Et2O to yield 5aD as a white crystalline solid (141 mg, 46%). 1H NMR (500 MHz, CDCl3, 295 K): δ 4.70 (s, 2H), 6.93 (m, 2H), 7.03 (m, 1H), 7.32 (m, 2H). 2H NMR (76 MHz, CHCl3, 315 K): δ 10.13 (bs, 1D). 13C NMR (75 MHz, CDCl3, 297 K): δ 64.9, 114.8, 122.4, 129.9, 157.4, 172.8. LR-ESIMS: m/z 153 [M]+. HR-ESIMS: m/z 153.0543 (calcd for C8H8O3D, 153.0513).
4-Phenoxybutanamide (19H)
Phenoxyacetic acid (100 mg, 0.66 mmol) and acrylamide (93.9 mg, 1.32 mmol) in MeCN (45 mL) were reacted in accordance with the general procedure in a TiO2 sol–gel-coated tube. Following irradiation for 24 h, the reaction mixture was purified by dry flash chromatography on silica gel (eluent: gradient of 2–10% MeOH in CH2Cl2) to yield 19H as an off-white solid (79.8 mg, 68%). 1H NMR (300 MHz, CDCl3, 294 K): δ 2.13 (m, 2H), 2.45 (t, J = 7.2 Hz, 2H), 4.02 (t, J = 6.0 Hz, 2H), 5.63 (bs, 1H), 5.82 (bs, 1H), 6.90 (d, J = 8.8 Hz, 2H), 6.95 (t, J = 7.4 Hz, 1H), 7.29 (t, J = 7.4 Hz, 2H). 13C NMR (75 MHz, CDCl3, 295 K): δ 25.4, 32.6, 67.1, 114.9, 121.2, 129.9, 159.2, 175.4. Data are consistent with literature values.16
4-(tert-Butyl)phenoxybutanamide
4-(tert-Butyl)phenoxyacetic acid (137.5 mg, 0.66 mmol) and acrylamide (93.9 mg, 1.32 mmol) in MeCN (45 mL) were reacted in accordance with the general procedure in a TiO2 sol–gel-coated tube. Following irradiation for 30 h, the reaction mixture was purified by dry flash chromatography on silica gel (eluent: gradient of 2–7.5% MeOH in CH2Cl2) to yield the title compound as an off-white solid (105.4 mg, 68%) containing ∼5% 4-(tert-butyl)phenol. Mp: 85–88 °C. 1H NMR (400 MHz, CD3OD, 297 K): δ 1.30 (s, 9H), 2.04–2.11 (m, 2H), 2.43 (t, J = 7.3 Hz, 2H), 4.00 (t, J = 6.3 Hz, 2H), 6.85 (d, J = 8.9 Hz, 2H), 7.30 (dd, J = 3.2, 11.5 Hz, 1H). 13C NMR (75 MHz, CDCl3, 297 K): δ 27.0, 32.4, 33.4, 35.3, 68.5, 115.5, 127.6, 144.8, 158.5, 179.0. LR-ESIMS: m/z 236 [M + H]+. HR-ESIMS: m/z 236.1647 (calcd for C14H22NO2, 236.1645).
4-(Trifluoromethyl)phenoxybutanamide
4-(Trifluoromethyl)phenoxyacetic acid (145.3 mg, 0.66 mmol) and acrylamide (93.9 mg, 1.32 mmol) in MeCN (45 mL) were reacted in accordance with the general procedure in a TiO2 sol–gel-coated tube. Following irradiation for 24 h, the reaction mixture was purified by dry flash chromatography on silica gel (eluent: 5% MeOH in CH2Cl2) to yield the title compound as an off-white solid (108.4 mg, 66%). Mp: 93–94 °C. 1H NMR (500 MHz, d6-acetone, 295 K): δ 2.06–2.11 (m, 2H), 2.41 (t, J = 7.2 Hz, 2H), 4.14 (t, J = 5.9 Hz, 2H), 6.30 (bs, 1H), 6.86 (bs, 1H), 7.14 (d, J = 8.7 Hz, 2H), 7.63 (d, J = 8.7 Hz, 2H). 13C NMR (75 MHz, d6-acetone, 297 K): δ 29.9, 36.3, 72.7, 120.0, 127.2 (q, 2JC–F = 33.1 Hz, 1C), 130.0 (q, 1JC–F = 271.1 Hz, 1C), 132.0 (q, 3JC–F = 3.6 Hz, 2C), 167.2, 178.9. 19F NMR (470 MHz, d6-acetone, 295 K): δ −61.8. LR-ESIMS: m/z 248 [M + H]+. HR-ESIMS: m/z 248.0896 (calcd for C11H13NO2F3, 248.0896).
Photocatalyzed Reaction of (E)-2-(2-(3-oxopent-1-en-1-yl)phenoxy)acetic Acid (8c)
A suspension of 8c (0.050 g, 0.21 mmol, 1.0 equiv) and TiO2 (P25, 0.025 g, 0.32 mmol, 1.5 equiv) in MeCN (20 mL) was reacted in accordance with the general procedure for 9 h. Following removal of the solvent under reduced pressure, the mixture was subjected to analysis by 1H NMR and GC–MS. The 1H NMR spectrum showed a complex array of components. GC–MS indicated four major components, and one of these (tR = 10.7 min) had the correct mass and fragmentation pattern for the cyclized product 10c: m/z (%): 190 (40), 161 (36), 146 (18), 133 (23), 119 (38), 118 (100), 91 (41). 1H NMR (400 MHz, CD3CN, 298 K): δ 0.98 (t, J = 7.3 Hz, 3H), 2.47 (q, J = 7.3 Hz, 2H), 3.26–3.29 (m, 1H), 4.37 (dd, J = 2.3, 8.4 Hz, 1H), 4.94 (dd, J = 5.2, 9.4 Hz, 1H), 7.03–7.17 (m, 2H,), 7.50–7.54 (m, 2H). Attempts to isolate and thoroughly characterize 10c were not successful. Photoisomerization of 8c to its Z isomer was observed during the course of this experiment. 1H NMR (400 MHz, CD3CN, 298 K): δ 7.38 (d, J = 7.3 Hz, 1H), 7.30 (t, J = 7.3 Hz, 1H), 7.02 (d, J = 12.0 Hz, 1H), 6.95 (t, J = 7.6 Hz, 1H), 6.88 (d, J = 8.3 Hz, 1H), 6.28 (d, J = 12.5 Hz, 1H), 4.69 (s, 2H), 2.47 (q, J = 7.3 Hz, 2H), 0.97 (t, J = 7.3 Hz, 3H).
Photocatalyzed Reaction of 2-([1,1′-Biphenyl]-2-yloxy)acetic Acid (11a)
A suspension of 11a (0.052 g, 0.23 mmol, 1.0 equiv) and TiO2 (P25, 0.027 g, 0.34 mmol, 1.5 equiv) in MeCN (20 mL) was reacted in accordance with the general procedure for 18 h. Following removal of the solvent under reduced pressure, 1H NMR analysis of the reaction mixture showed mainly unreacted 11a together with a small amount of the cyclized product 12a (0.01 mmol, 5% wrt CH2Br2). 1H NMR (400 MHz, CDCl3, 296 K): δ 5.05 (s, 2H), 6.90–6.95 (m, 2H), 7.00–7.10 (m, obscured by SM), 7.60–7.70 (m, 3H). Attempts to isolate and thoroughly characterize 12a were unsuccessful.
Methyl 2-(2,3-Dihydrobenzofuran-3-yl)acetate (10a)
A suspension of 8a (0.050 g, 0.21 mmol, 1.0 equiv) and TiO2 (P25, 0.025 g, 0.32 mmol, 1.5 equiv) in MeCN (20 mL) was reacted according to the general procedure for 9 h to yield 10a as a colorless powder (20% by NMR). 1H NMR (400 MHz, CDCl3, 294 K): δ 2.52 (dd, J = 9.5, 16.5 Hz, 1H), 2.73 (dd, J = 5.3, 16.5 Hz, 1H), 3.72 (s, 3H), 3.84–3.91 (m, 1H), 4.18 (dd, J = 6.3, 9.2 Hz, 1H), 4.68 (t, J = 9.0 Hz, 1H), 6.80 (d, J = 7.7 Hz, 1H), 6.86 (t, J = 8.3 Hz, 1H), 7.08–7.17 (m, 2H). 13C NMR (75 MHz, CDCl3, 295 K): δ 38.7, 39.7, 52.3, 110.2, 114.1, 121.0, 124.6, 129.1, 129.5, 160.2, 172.7. LR-ESIMS: m/z 215 [M + Na]+. HR-ESIMS: m/z 215.0674 (calcd for C11H12O3Na, 215.0679). Photoisomerization of 8a to its Z isomer was observed during the course of this experiment. 1H NMR (400 MHz, CD3CN, 298 K): δ 7.48 (dd, J = 7.6, 1.8 Hz, 1H), 7.31 (t, J = 7.8 Hz, 1H), 7.19 (d, J = 12.5 Hz, 1H), 6.97 (t, J = 7.6 Hz, 1H), 6.88 (d, J = 8.3 Hz, 1H), 6.01 (d, J = 12.3 Hz, 1H), 4.69 (s, 2H), 3.63 (s, 3H).
2-(2,3-Dihydrobenzofuran-3-yl)acetonitrile (10b)
A solution of 8b (0.050 g, 0.246 mmol) in MeCN (15 mL) in a TiO2 sol–gel-coated tube was reacted according to the general procedure. The solvent was removed under reduced pressure, and the crude residue was purified by column chromatography (CH2Cl2) to yield 10b as a colorless powder (0.007 g, 28%). 1H NMR (400 MHz, CDCl3, 296 K): δ 2.58–2.71 (m, 2H), 3.74–3.82 (m, 1H), 4.34 (dd, J = 4.8, 9.6 Hz, 1H), 4.69 (dd, J = 8.7, 9.6 Hz, 1H). 6.84 (d, J = 8.1 Hz, 1H), 6.93 (td, J = 1.0, 7.5 Hz, 1H), 7.22 (m, 1H), 7.32 (d, J = 7.5 Hz, 1H). 13C NMR (75 MHz, CDCl3, 295 K): δ 22.8, 38.8, 75.4, 110.3, 117.7, 121.2, 124.4, 127.1, 129.7, 159.7. LR-ESIMS: m/z 159 [M]+. HR-ESIMS: m/z 159.0676 (calcd for C10H9NO, 159.0679). Photoisomerization of 8b to its Z isomer was observed during the course of this experiment. 1H NMR (400 MHz, CD3CN, 298 K): δ 8.01 (dd, J = 7.6, 1.6 Hz, 1H), 7.66 (d, J = 12.5 Hz, 1H), 7.44 (t, J = 8.0 Hz, 1H), 7.11 (t, J = 7.5 Hz, 1H), 6.97 (d, J = 8.3 Hz, 1H), 5.64 (d, J = 12.3 Hz, 1H), 4.74 (s, 2H).
Photocatalyzed Reaction of 2-((4′-(Trifluoromethyl)-[1,1′-biphenyl]-2-yl)oxy)acetic Acid (11c)
A suspension of 11c (59 mg, 0.20 mmol, 1.0 equiv) and TiO2 (P25, 0.024 g, 0.30 mmol, 1.5 equiv) in MeCN (20 mL) was reacted in accordance with the general procedure for 18 h. Following removal of the solvent under reduced pressure, 1H NMR analysis of the reaction mixture showed mainly unreacted acid (79%) together with a small amount of the cyclized product 12c (0.01 mmol, 5% wrt CH2Br2). 1H NMR (400 MHz, CDCl3, 296 K): δ 5.07 (s, 2H), 6.93–6.96 (m, 2H), 6.98–7.04 (m; obscured by SM), 7.72–7.65 (m, 3H). GC–MS analysis of the reaction mixture showed 12c as the main product: tR = 11.76 min; m/z (%) 250 (58), 249 (100), 231 (6), 201 (10), 181 (10), 152 (16). Also observed was a trace of what was probably the direct reduction product 2-methoxy-4′-(trifluoromethyl)-1,1′-biphenyl: tR = 11.84 min; m/z (%) 252 (82), 251 (48), 250 (61), 249 (100), 237 (20), 201 (16), 183 (26), 152 (32). Attempts to isolate and thoroughly characterize 12c were unsuccessful.
Photocatalyzed Reaction of 2-(2-((Methoxyimino)methyl)phenoxy)acetic Acid (13a)
A suspension of 13a (51 mg, 0.24 mmol, 1.0 equiv) and TiO2 (P25, 0.029 g, 0.36 mmol, 1.5 equiv) in MeCN (29 mL) was reacted in accordance with the general procedure for 24 h. Following removal of the solvent under reduced pressure, 1H NMR analysis of the reaction mixture showed that 13a had been entirely consumed and that a complex mixture of products had been formed. GC–MS analysis of the mixture revealed that 14a had been formed: tR = 8.15 min; m/z (%) 165 (29), 133 (12), 119 (100), 91 (77) 77 (26). Also observed were benzofuran-3(2H)-one O-methyl oxime [tR = 7.93 min; m/z (%) 163 (100), 118 (91), 91 (52)], 2,3-dihydrobenzofuran [tR = 7.87 min; m/z (%) 120 (27), 119 (100), 91 (57)], 2,3-dihydrobenzofuran-3-amine [tR = 5.06 min; m/z (%) 135 (7), 134 (74), 119 (100), 117 (11)], and benzofuran [tR = 3.43 min; m/z (%) 118 (100), 90 (22), 84 (30)]. Attempts to isolate and thoroughly characterize 14a, or any other products, were unsuccessful.
Photocatalyzed Reaction of 2-(2-(((Benzyloxy)imino)methyl)phenoxy)acetic Acid (13b)
A suspension of 13b (62 mg, 0.22 mmol, 1.0 equiv) and TiO2 (P25, 0.026 g, 0.33 mmol, 1.5 equiv) in MeCN (26 mL) was reacted in accordance with the general procedure for 24 h. Following removal of solvent under reduced pressure, 1H NMR analysis of the reaction mixture showed that 13b had been entirely consumed and that a complex mixture of products had been formed. GC–MS analysis of the mixture revealed that 14b had been formed: tR = 15.53 min; m/z (%) 241 (9), 210 (9), 133 (6), 91 (100), 77 (14). Also observed were benzofuran-3(2H)-one O-benzyl oxime [tR = 15.37 min; m/z (%) 239 (37), 222 (4), 148 (3), 119 (7), 91 (100), 77 (12)], 2,3-dihydrobenzofuran-3-amine [tR = 5.04 min; m/z (%) 135 (7), 134 (74), 119 (100), 117 (11)], benzyl alcohol [tR = 3.77 min; m/z (%) 108 (100), 107 (73), 79 (76)], benzofuran [tR = 3.41 min; m/z (%) 118 (100), 90 (22), 84 (30)], and benzaldehyde [tR = 2.90 min; m/z (%) 106 (87), 105 (100), 77 (66)]. Attempts to isolate and thoroughly characterize 14b or any other product were unsuccessful.
SCPC Reaction of Phenoxyacetic Acid with Acrylamide and P25 Vacuum-Dried at 150 °C
TiO2 (12 mg, 0.15 mmol) was dried overnight at 150 °C under a continuous vacuum in an oven-dried Schlenk tube. Phenoxyacetic acid (15.2 mg, 0.1 mmol) and acrylamide (14.2 mg, 0.2 mmol) in MeCN (12 mL) were added via syringe, and the resultant mixture was irradiated for 18 h. 1H NMR analysis of the resultant mixture revealed 19H (39% by NMR).
SCPC Reaction in Deuterated Acetonitrile (d3-MeCN)
Phenoxyacetic acid (15.2 mg, 0.1 mmol), acrylamide (14.2 mg, 0.2 mmol), and TiO2 (12 mg, 0.15 mmol) in d3-MeCN (12 mL) were irradiated for 18 h. 1H NMR analysis of the resultant mixture revealed 19H (55% by NMR). Any deuterium incorporation was below the NMR and MS detection limits.
SCPC Reaction with Deuterated Phenoxyacetic Acid
d1-Phenoxyacetic acid (15.3 mg, 0.1 mmol), acrylamide (14.2 mg, 0.2 mmol), and TiO2 (12 mg, 0.15 mmol) in MeCN (12 mL) were irradiated for 19 h. 1H NMR analysis (wrt CH2Br2 standard) of the resultant mixture revealed 19H (49% by NMR). Any deuterium incorporation was below the NMR and MS detection limits.
SCPC Reaction with Deuterated P25
An oven-dried Schlenk tube, complete with magnetic stirrer, was connected to a vacuum line, evacuated, and then back-filled with argon while still hot from the oven. In a glovebox, TiO2 (P25, 0.15 mmol, 12 mg) was added, and the tube was then heated at 150 °C for 3 h under vacuum. In a separate oven-dried vessel, 10 mL of D2O (freshly opened) was degassed by three freeze–pump–thaw cycles before being transferred by cannula to the tube containing the TiO2 once it had cooled to ambient temperature. The resultant mixture was allowed to stir overnight before the D2O was removed under reduced pressure. At this point the TiO2 was again dried for 3 h at 150 °C under vacuum. In a separate oven-dried vessel, phenoxyacetic acid (0.1 mmol, 15.2 mg) and acrylamide (0.2 mmol, 14.2 mg) were dissolved in MeCN (12 mL) that had been freshly collected after distillation over calcium hydride. The resultant mixture was degassed for 15 min by bubbling with argon and then transferred via cannula to the tube containing the TiO2 once it had cooled to ambient temperature. The resultant mixture was then irradiated in accordance with the general procedure for 19 h. 1H NMR analysis of the resultant mixture revealed 19H (58% by NMR). Any deuterium incorporation was below the NMR and MS detection limits.
SCPC Reaction with Deuterated Phenoxyacetic Acid and Deuterated P25
This reaction was performed as above using d1-phenoxyacetic acid (15.3 mg, 0.1 mmol) for 19 h. 1H NMR analysis of the resultant mixture revealed no clear signals indicating the formation of 19H or 19D.
Acknowledgments
We thank the EPSRC for funding (Grants EP/I003479/1 and EP/I003800/2), the EPSRC National Mass Spectrometry Service, Swansea, and Prof. Russell Howe and Shona Rhydderch for help with deuterium labeling of P25.
Supporting Information Available
General procedures, EPR spectra, NMR monitoring of coated tube reactions, and 1H and 13C NMR spectra for new compounds. This material is available free of charge via the Internet at http://pubs.acs.org.
The authors declare no competing financial interest.
Supplementary Material
References
- For a review, see:Calvert J. G.; Pitts J. N.. Photochemistry; Wiley: New York, 1966; pp 427–434. [Google Scholar]
- Budac D.; Wan P. J. Photochem. Photobiol., A 1992, 67, 135–166. [Google Scholar]
- For a review, see:Nonhebel D. C.; Walton J. C. In Free Radical Chemistry; Cambridge University Press: Cambridge, U.K., 1974; pp 99–127. [Google Scholar]
- a Barton D. H. R.; Crich D.; Motherwell W. B. J. Chem. Soc., Chem. Commun. 1983, 939–941. [Google Scholar]; b Motherwell W. B.; Crich D.. Free Radical Chain Reactions in Organic Synthesis; Academic Press: London, 1992. [Google Scholar]
- a Johnson R. G.; Ingham R. K. Chem. Rev. 1956, 56, 219–269. [Google Scholar]; b Baker F. W.; Holtz H. D.; Stock L. M. J. Org. Chem. 1963, 28, 514–516. [Google Scholar]
- Magdziak D.; Lalic G.; Lee H. M.; Fortner K. C.; Aloise A. D.; Shair M. D. J. Am. Chem. Soc. 2005, 127, 7284–7285. [DOI] [PubMed] [Google Scholar]
- Yin L.; Kanai M.; Shibasaki M. J. Am. Chem. Soc. 2009, 131, 9610–9611. [DOI] [PubMed] [Google Scholar]
- Bi H.-P.; Zhao L.; Liang Y.-M.; Li C.-J. Angew. Chem., Int. Ed. 2009, 48, 792–795. [DOI] [PubMed] [Google Scholar]
- a Yoshimi Y.; Itou T.; Hatanaka M. Chem. Commun. 2007, 5244–5246. [DOI] [PubMed] [Google Scholar]; b Leung J. C. T.; Chatalova-Sazepin C.; West J. G.; Rueda-Becerril M.; Paquin J.-F.; Sammis G. M. Angew. Chem., Int. Ed. 2012, 51, 10804–10807. [DOI] [PubMed] [Google Scholar]
- a Kraeutler B.; Bard A. J. J. Am. Chem. Soc. 1978, 100, 2239–2240. [Google Scholar]; b Kraeutler B.; Jaeger C. D.; Bard A. J. J. Am. Chem. Soc. 1978, 100, 4903–4905. [Google Scholar]
- a Higashida S.; Harada A.; Kawakatsu R.; Fujiwara N.; Matsumura M. Chem. Commun. 2006, 2804–2806. [DOI] [PubMed] [Google Scholar]; b Mills A. J. Photochem. Photobiol., A 2012, 233, 34–39. [Google Scholar]
- a Linsebigler A. L.; Lu G.; Yates J. T. Jr. Chem. Rev. 1995, 95, 735–758. [Google Scholar]; b Ravelli D.; Dondi D.; Fagnoni M.; Albini A. Chem. Soc. Rev. 2009, 38, 1999–2011. [DOI] [PubMed] [Google Scholar]; c Li W.; Li D.; Lin Y.; Wang P.; Chen W.; Fu X.; Shao Y. J. Phys. Chem. C 2012, 116, 3552–3560. [Google Scholar]
- a Ohtani B.; Osaki H.; Nishimoto S.-I.; Kagiya T. J. Am. Chem. Soc. 1986, 108, 308–310. [Google Scholar]; b Wang C. M.; Mallouk T. E. J. Am. Chem. Soc. 1990, 112, 2016–2018. [Google Scholar]; c Ohtani B.; Goto Y.; Nishimoto S.-I.; Inui T. J. Chem. Soc., Faraday Trans. 1996, 92, 4291–4295. [Google Scholar]; d Cermenati L.; Mella M.; Albini A. Tetrahedron 1998, 54, 2575–2582. [Google Scholar]
- a Künneth R.; Feldmer C.; Knoch F.; Kisch H. Chem.—Eur. J. 1995, 1, 441–448. [Google Scholar]; b Keck H.; Schindler W.; Knoch F.; Kisch H. Chem.—Eur. J. 1997, 3, 1638–1645. [Google Scholar]
- a Marinković S.; Hoffmann N. Chem. Commun. 2001, 1576–1578. [DOI] [PubMed] [Google Scholar]; b Marinković S.; Hoffmann N. Int. J. Photoenergy 2003, 5, 175–182. [Google Scholar]; c Marinković S.; Hoffmann N. Eur. J. Org. Chem. 2004, 3102–3107. [DOI] [PubMed] [Google Scholar]
- Manley D. W.; McBurney R. T.; Miller P.; Howe R. F.; Rhydderch S.; Walton J. C. J. Am. Chem. Soc. 2012, 134, 13580–13583. [DOI] [PubMed] [Google Scholar]
- Noorjahan M.; Reddy M. P.; Kumari V. D.; Lavédrine B.; Boule P.; Subrahmanyam M. J. Photochem. Photobiol., A 2003, 156, 179–187. [Google Scholar]
- http://microspheretechnology.com/wordpress/index.php/titanium-dioxide-coating-technology/ (accessed Nov 19, 2013).
- Roy R. B.; Swan G. A. J. Chem. Soc. C 1969, 1886–1891. [Google Scholar]
- Mikami T.; Harada M.; Narasaka K. Chem. Lett. 1999, 425–426. [Google Scholar]
- For example, see:; a Moshkin V. S.; Sosnovskikh V. Ya.; Roschenthaler G.-V. Tetrahedron 2013, 69, 5884–5892. [Google Scholar]; b Dubuffet T.; Newman-Tancredi A.; Cussac D.; Audinot V.; Loutz A.; Millan M. J.; Lavielle G. Bioorg. Med. Chem. Lett. 1999, 9, 2059–2064. [DOI] [PubMed] [Google Scholar]
- For example, see:; a Hata K.; He Z.; Daniliuc C. G.; Itami K.; Studer A. Chem. Commun. 2014, 50, 463–465. [DOI] [PubMed] [Google Scholar]; b Sheppard T. D. J. Chem. Res. 2011, 35, 377–385. [Google Scholar]; c Bertolini F.; Pineschi M. Org. Prep. Proced. Int. 2009, 41, 385–418. [Google Scholar]
- Millenium PC-500 TiO2 was used in EPR experiments because its finer mesh meant it stayed in suspension without stirring during EPR spectral scans.
- a Tedder J. M.; Walton J. C. Acc. Chem. Res. 1976, 9, 183–191. [Google Scholar]; b Fischer H.; Radom L. Angew. Chem., Int. Ed. 2001, 1340–1371. [DOI] [PubMed] [Google Scholar]; c Poutsma M. L. J. Phys. Org. Chem. 2008, 21, 758–782. [Google Scholar]
- Kraeutler B.; Bard A. J. J. Am. Chem. Soc. 1978, 100, 5985–5992. [Google Scholar]
- a Heiba E.-A. I.; Dessau R. M.; Koehl W. J. Jr. J. Am. Chem. Soc. 1968, 90, 1082–1084. [Google Scholar]; b Heiba E.-A. I.; Dessau R. M.; Koehl W. J. Jr. J. Am. Chem. Soc. 1969, 91, 138–145. [Google Scholar]; c Kurz M. E.; Baru V.; Nguyen P. N. J. Org. Chem. 1984, 49, 1603–1607. [Google Scholar]; d Citterio A.; Sebastiano R.; Maronati A.; Santi R.; Bergamini F. Chem. Commun. 1994, 1517–1518. [Google Scholar]
- Henderson M. A. Surf. Sci. Rep. 2011, 66, 185–297. [Google Scholar]
- a Deiana C.; Fois E.; Coluccia S.; Martra G. J. Phys. Chem. C 2010, 114, 21531–21538. [Google Scholar]; b Salazar C.; Nanny M. A. J. Catal. 2010, 269, 404–410. [Google Scholar]
- Szczepankiewicz S. H.; Colussi A. J.; Hoffmann M. R. J. Phys. Chem. B 2000, 104, 9842–9850. [Google Scholar]
- Ballini R.; Bosica G.; Cioci G.; Fiorini D.; Petrini M. Tetrahedron 2003, 59, 3603–3608. [Google Scholar]
- Bouillon J.-P.; Brule C.; Portella C. Tetrahedron 2004, 60, 9849–9855. [Google Scholar]
- Chen P.-S.; Chou C.-H. Tetrahedron 1996, 52, 13615–13622. [Google Scholar]
- Montanaro S.; Ravelli D.; Merli D.; Fagnoni M.; Albini A. Org. Lett. 2012, 14, 4218–4221. [DOI] [PubMed] [Google Scholar]
- Felpin F.-X.; Coste J.; Zakri C.; Fouquet E. Chem.—Eur. J. 2009, 15, 7238–7245. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.