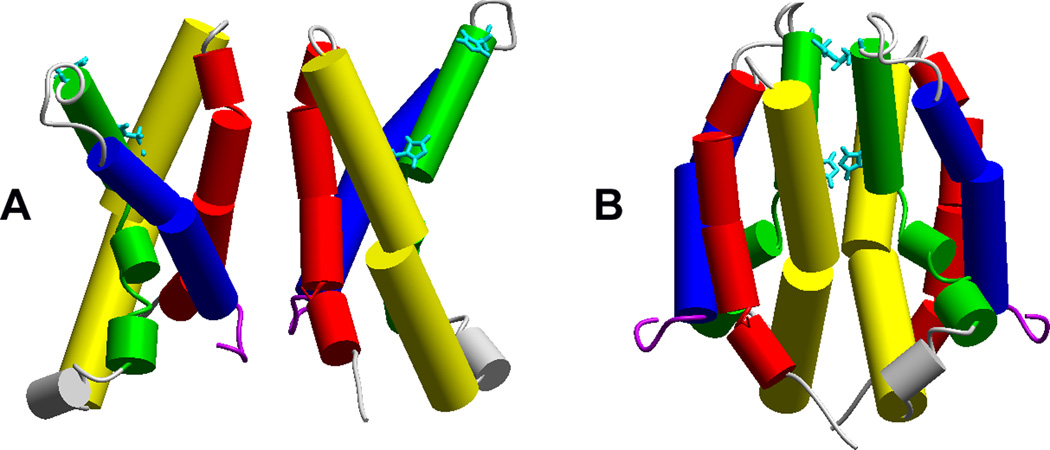
Figure 4.
Two proposed dimer interfaces. Crosslinking studies indicate several points of attachment at the top (outer) end of S1 (red) [4]. The binding of Zn2+ to various hHV1 mutants with His140 or His193 (aqua) replaced by Ala suggested that high-affinity bidentate Zn2+ binding occurs at the dimer interface [21]. Reprinted with permission from: Musset B, Smith SME, Rajan S, Cherny VV, Sujai S, Morgan D, and DeCoursey TE. Zinc inhibition of monomeric and dimeric proton channels suggests cooperative gating. Journal of Physiology 588, 1435–1449, copyright 2010.